Abstract
Background
Allopurinol reduces oxidative stress and may thus have an anti-inflammatory effect. Previous studies suggest that allopurinol use might decrease the risk of prostate cancer (PCa) among gout patients. We studied the association between allopurinol use and PCa incidence.
Methods
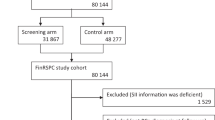
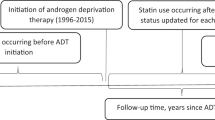
The cohort consists of 76,874 men without prevalent PCa, originally identified for the Finnish Randomized Study of Screening for Prostate Cancer (FinRSPC). The follow-up started at entry to the trial. We excluded men using allopurinol in the year before entry (wash-out). PCa cases detected during 1996–2015 were identified from the Finnish Cancer Registry. Information on tumor Gleason score and TNM stage were obtained from medical files. Information on PSA level was obtained from screening samples for men in the FinRSPC screening arm and from laboratory databases for men in the control arm. Information on BMI was based on a questionnaire sent to men in the FinRSPC screening arm in 2004–2008. Drug purchase information were obtained from the national prescription database. We used Cox regression (adjusted for age, FinRSPC trial arm, PCa family history and use of other medication) to calculate hazard ratios (HRs) and 95% confidence intervals (CIs) of PCa risk by allopurinol use. We analyzed medication as a time-dependent variable to minimize immortal time bias.
Results
There were 9062 new PCa diagnoses in the cohort. Follow-up time did not differ by allopurinol use (median 17 yr; IQR 11–19 vs median 17 yr; IQR 12.33–19). The risk of PCa did not differ by allopurinol use (multivariable adjusted HR 1.03; 95% CI 0.92–1.16). Allopurinol use did not associate with the risk of high-grade or metastatic cancer. Cumulative duration or average yearly dose of allopurinol use showed no association with PCa risk. No delayed risk associations were observed in the lag-time analyses.
Conclusions
We observed no difference in the PCa risk by allopurinol use.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Kienhorst LBE, van Lochem E, Kievit W, Dalbeth N, Merriman ME, Phipps-Green A, et al. Gout is a chronic inflammatory disease in which high levels of interleukin-8 (CXCL8), myeloid-related protein 8/myeloid-related protein 14 complex, and an altered proteome are associated with diabetes mellitus and cardiovascular disease. Arthritis Rheumatol. 2015;67:3303–13.
So AK, Martinon F. Inflammation in gout: mechanisms and therapeutic targets. Nat Rev Rheumatol. 2017;13:639–47.
Chang S-J, Tsai P-C, Chen C-J, Lai H-M, Ko Y-C. The polymorphism-863C/A in tumour necrosis factor-alpha gene contributes an independent association to gout. Rheumatology. 2007;46:1662–6.
Tsai P-C, Chen C-J, Lai H-M, Chang S-J. Analysis of polymorphisms in the promoter region and protein levels of interleukin-6 gene among gout patients. Clin Exp Rheumatol. 2008;26:841–7.
Liu S, Yin C, Chu N, Han L, Li C. IL-8 -251T/A and IL-12B 1188A/C polymorphisms are associated with gout in a Chinese male population. Scand J Rheumatol. 2013;42:150–8.
Platz EA, Kulac I, Barber JR, Drake CG, Joshu CE, Nelson WG, et al. A prospective study of chronic inflammation in benign prostate tissue and risk of prostate cancer: Linked PCPT and SELECT Cohorts. Cancer Epidemiol Biomark Prev. 2017;26:1549–57.
Tewari AK, Stockert JA, Yadav SS, Yadav KK, Khan I. Inflammation and prostate cancer. Adv Exp Med Biol. 2018;1095:41–65.
Wang W, Xu D, Wang B, Yan S, Wang X, Yin Y, et al. Increased risk of cancer in relation to gout: a review of three prospective cohort studies with 50,358 subjects. Mediat Inflamm. 2015;2015:680853.
Chen C-J, Yen J-H, Chang S-J. Gout patients have an increased risk of developing most cancers, especially urological cancers. Scand J Rheumatol. 2014;43:385–90.
Kuo C-F, Luo S-F, See L-C, Chou I-J, Fang Y-F, Yu K-H. Increased risk of cancer among gout patients: a nationwide population study. Jt Bone Spine. 2012;79:375–8.
Boffetta P, Nordenvall C, Nyrén O, Ye W. A prospective study of gout and cancer. Eur J Cancer Prev. 2009;18:127–32.
Sangkop F, Singh G, Rodrigues E, Gold E, Bahn A. Uric acid: a modulator of prostate cells and activin sensitivity. Mol Cell Biochem. 2016;414:187–99.
Castro GD, Costantini MH, Castro JA. Rat ventral prostate xanthine oxidase-mediated metabolism of acetaldehyde to acetyl radical. Hum Exp Toxicol. 2009;28:203–8.
Yasuda T, Yoshida T, Goda AE, Horinaka M, Yano K, Shiraishi T, et al. Anti-gout agent allopurinol exerts cytotoxicity to human hormone-refractory prostate cancer cells in combination with tumor necrosis factor-related apoptosis-inducing ligand. Mol Cancer Res. 2008;6:1852–60.
Shih H-J, Kao M-C, Tsai P-S, Fan Y-C, Huang C-J. Long-term allopurinol use decreases the risk of prostate cancer in patients with gout: a population-based study. Prostate Cancer Prostatic Dis. 2017;20:328–33.
Kukko V, Kaipia A, Talala K, Taari K, Tammela TLJ, Auvinen A, et al. Allopurinol and risk of benign prostatic hyperplasia in a Finnish population-based cohort. Prostate Cancer Prostatic Dis. 2018;21:373–8.
Kilpeläinen TP, Tammela TL, Malila N, Hakama M, Santti H, Määttänen L, et al. Prostate cancer mortality in the Finnish randomized screening trial. J Natl Cancer Inst. 2013;105:719–25.
Sarre S, Määttänen L, Tammela TLJ, Auvinen A, Murtola TJ. Postscreening follow-up of the Finnish Prostate Cancer Screening Trial on putative prostate cancer risk factors: vitamin and mineral use, male pattern baldness, pubertal development and non-steroidal anti-inflammatory drug use. Scand. J Urol. 2016;50:267–73.
Rates of reimbursement for medicines. https://www.kela.fi/web/en/reimbursements-for-medicine-expences-amount. Accessed 30 July 2018.
WHOCC—ATC/DDD Index. https://www.whocc.no/atc_ddd_index/. Accessed 30 July 2018.
Buschemeyer WC, Freedland SJ. Obesity and prostate cancer: epidemiology and clinical implications. Eur Urol. 2007;52:331–43.
Murtola TJ, Tammela TLJ, Määttänen L, Ala-Opas M, Stenman UH, Auvinen A. Prostate cancer incidence among finasteride and alpha-blocker users in the Finnish Prostate Cancer Screening Trial. Br J Cancer. 2009;101:843–8.
Murtola TJ, Vettenranta AM, Talala K, Taari K, Stenman U-H, Tammela TLJ, et al. Outcomes of prostate-specific antigen-based prostate cancer screening among men using nonsteroidal anti-inflammatory drugs. Eur Urol Focus. 2018;4:851–7.
Bardin T, Richette P. Definition of hyperuricemia and gouty conditions. Curr Opin Rheumatol. 2014;26:186–91.
Singh JA. Gout: will the “King of Diseases” be the first rheumatic disease to be cured? BMC Med. 2016;14:180.
Thottam GE, Krasnokutsky S, Pillinger MH. Gout and metabolic syndrome: a tangled web. Curr Rheumatol Rep. 2017;19:60.
Klaukka T. The Finnish database on drug utilisation. Nor Epidemiol. 2001;11:19–22 19.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
TLJT: consultant fees from Amgen, Astellas, Bayer, Ferring and Roche. TJM: consultant fees from Astellas and Janssen Cilag. Lecture fees from Astellas, GSK and Janssen Cilag. KT: Research funding from Medivation/Astellas/Pfizer, travel support from Astellas. The remaining authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Kukko, V., Kaipia, A., Talala, K. et al. Allopurinol and the risk of prostate cancer in a Finnish population-based cohort. Prostate Cancer Prostatic Dis 22, 483–490 (2019). https://doi.org/10.1038/s41391-019-0129-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-019-0129-2
This article is cited by
-
Allopurinol and prostate cancer survival in a Finnish population-based cohort
Prostate Cancer and Prostatic Diseases (2022)