Abstract
Purpose
The objective of this study was to test a low-carbohydrate diet (LCD) plus walking to reduce androgen deprivation therapy (ADT)-induced metabolic disturbances.
Materials and methods
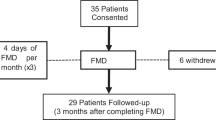
This randomized multi-center trial of prostate cancer (PCa) patients initiating ADT was designed to compare an LCD (≤20g carbohydrate/day) plus walking (≥30 min for ≥5 days/week) intervention vs. control advised to maintain usual diet and exercise patterns. Primary outcome was change in insulin resistance by homeostatic model assessment at 6 months. To detect 20% reduction in insulin resistance, 100 men were required. The study was stopped early after randomizing 42 men due to slow accrual. Secondary outcomes included weight, body composition, lipids, and prostate-specific antigen (PSA). Changes from baseline were compared between arms using rank-sum tests.
Results
At 6 months, LCD/walking reduced insulin resistance by 4% vs. 36% increase in control (p = 0.13). At 3 months, vs. control, LCD/walking arm significantly lost weight (7.8kg; p<0.001), improved insulin resistance (↑36%; p = 0.015), hemoglobin A1c (↓3.3%; p = 0.01), high-density lipoprotein (HDL) (↑13%; p = 0.004), and triglyceride (↓37%; p = 0.036). At 6 months, weight loss (10.6kg; p<0.001) and HDL (↑27%; p = 0.003) remained significant. LCD/walking preserved total body bone mineral count (p = 0.025), reduced fat mass (p = 0.002), lean mass (p = 0.036), and percent body fat (p = 0.004). There were no differences in PSA. Limitations include the effect of LCD, weight loss vs. walking instruction are indistinguishable, and small sample size.
Conclusions
In an underpowered study, LCD/walking did not improve insulin sensitivity at 6 months. Given most secondary outcomes were improved at 3 months with some remaining improved at 6 months and a secondary analysis showed that LCD/walking reduced insulin resistance over the study, supporting future larger studies of LCD/walking intervention to reduce ADT-induced disturbances.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Liede A, Hallett DC, Hope K, Graham A, Arellano J, Shahinian VB. International survey of androgen deprivation therapy (ADT) for non-metastatic prostate cancer in 19 countries. ESMO Open. 2016;1:e000040.
Gilbert SM, Kuo YF, Shahinian VB. Prevalent and incident use of androgen deprivation therapy among men with prostate cancer in the United States. Urol Oncol. 2011;29:647–53.
Ahmadi H, Daneshmand S. Androgen deprivation therapy for prostate cancer: long-term safety and patient outcomes. Patient Relat Outcome Meas. 2014;5:63–70.
Davis MK, Rajala JL, Tyldesley S, Pickles T, Virani SA. The prevalence of cardiac risk factors in men with localized prostate cancer undergoing androgen deprivation therapy in British Columbia, Canada. J Oncol. 2015;2015:820403.
Smith MR, Lee H, Nathan DM. Insulin sensitivity during combined androgen blockade for prostate cancer. J Clin Endocrinol Metab. 2006;91:1305–8.
Keating NL, O'Malley AJ, Smith MR. Diabetes and cardiovascular disease during androgen deprivation therapy for prostate cancer. J Clin Oncol. 2006;24:4448–56.
Keating NL, Liu PH, O'Malley AJ, Freedland SJ, Smith MR. Androgen-deprivation therapy and diabetes control among diabetic men with prostate cancer. Eur Urol. 2014;65:816–24.
Crawley D, Garmo H, Rudman S, Stattin P, Haggstrom C, Zethelius B, et al. Association between duration and type of androgen deprivation therapy and risk of diabetes in men with prostate cancer. Int J Cancer. 2016;139:2698–704.
Gardner CD, Kiazand A, Alhassan S, Kim S, Stafford RS, Balise RR, et al. Comparison of the Atkins, Zone, Ornish, and LEARN diets for change in weight and related risk factors among overweight premenopausal women: the A TO Z Weight Loss Study: a randomized trial. JAMA. 2007;297:969–77.
Finlay A, Wittert G, Short CE. A systematic review of physical activity-based behaviour change interventions reaching men with prostate cancer. J Cancer Surviv. 2018;12:571–91.
ATKINS 20®: An effective diet for weight loss: Atkins Foundation. https://www.atkins.com/how-it-works/atkins-20.
Thompson FE, Kirkpatrick SI, Subar AF, Reedy J, Schap TE, Wilson MM, et al. The National Cancer Institute's Dietary Assessment Primer: a resource for diet research. J Acad Nutr Diet. 2015;115:1986–95.
Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346:393–403.
Amireault S, Godin G, Lacombe J, Sabiston CM. Validation of the Godin–Shephard Leisure-Time Physical Activity Questionnaire classification coding system using accelerometer assessment among breast cancer survivors. J Cancer Surviv. 2015;9:532–40.
Hagell P, Westergren A, Arestedt K. Beware of the origin of numbers: standard scoring of the SF-12 and SF-36 summary measures distorts measurement and score interpretations. Res Nurs Health. 2017;40:378–86.
Morote J, Gomez-Caamano A, Alvarez-Ossorio JL, Pesqueira D, Tabernero A, Gomez Veiga F, et al. The metabolic syndrome and its components in patients with prostate cancer on androgen deprivation therapy. J Urol. 2015;193:1963–9.
Gandaglia G, Sun M, Popa I, Schiffmann J, Trudeau V, Shariat SF, et al. Cardiovascular mortality in patients with metastatic prostate cancer exposed to androgen deprivation therapy: a population-based study. Clin Genitourin Cancer. 2015;13:e123–30.
Wang A, Karunasinghe N, Plank L, Zhu S, Osborne S, Bishop K, et al. Effect of androgen deprivation therapy on bone mineral density in a Prostate Cancer Cohort in New Zealand: A Pilot Study. Clin Med Insights Oncol. 2017;11:1179554917733449.
Teoh JY, Chiu PK, Chan SY, Poon DM, Cheung HY, Hou SS, et al. Risk of new-onset diabetes after androgen deprivation therapy for prostate cancer in the Asian population. J Diabetes. 2015;7:672–80.
Derweesh IH, Diblasio CJ, Kincade MC, Malcolm JB, Lamar KD, Patterson AL, et al. Risk of new-onset diabetes mellitus and worsening glycaemic variables for established diabetes in men undergoing androgen-deprivation therapy for prostate cancer. BJU Int. 2007;100:1060–5.
Mitsuzuka K, Kyan A, Sato T, Orikasa K, Miyazato M, Aoki H, et al. Influence of 1 year of androgen deprivation therapy on lipid and glucose metabolism and fat accumulation in Japanese patients with prostate cancer. Prostate Cancer Prostatic Dis. 2016;19:57–62.
Smith MR, Lee H, McGovern F, Fallon MA, Goode M, Zietman AL, et al. Metabolic changes during gonadotropin-releasing hormone agonist therapy for prostate cancer: differences from the classic metabolic syndrome. Cancer . 2008;112:2188–94.
Oka R, Utsumi T, Endo T, Yano M, Kamijima S, Kamiya N, et al. Effect of androgen deprivation therapy on arterial stiffness and serum lipid profile changes in patients with prostate cancer: a prospective study of initial 6-month follow-up. Int J Clin Oncol. 2016;21:389–96.
Nguyen PL, Jarolim P, Basaria S, Zuflacht JP, Milian J, Kadivar S, et al. Androgen deprivation therapy reversibly increases endothelium-dependent vasodilation in men with prostate cancer. J Am Heart Assoc. 2015;4:e001914.
Dockery F, Bulpitt CJ, Agarwal S, Donaldson M, Rajkumar C. Testosterone suppression in men with prostate cancer leads to an increase in arterial stiffness and hyperinsulinaemia. Clin Sci (Lond). 2003;104:195–201.
Ziaran S, Goncalves FM, Breza J Sr. Patients with prostate cancer treated by ADT have significantly higher fibrinogenemia than healthy control. World J Urol. 2013;31:289–92.
Torimoto K, Samma S, Kagebayashi Y, Chihara Y, Tanaka N, Hirayama A, et al. The effects of androgen deprivation therapy on lipid metabolism and body composition in Japanese patients with prostate cancer. Jpn J Clin Oncol. 2011;41:577–81.
Saglam HS, Kose O, Kumsar S, Budak S, Adsan O. Fasting blood glucose and lipid profile alterations following twelve-month androgen deprivation therapy in men with prostate cancer. Scientific World J. 2012;2012:696329.
Salvador C, Planas J, Agreda F, Placer J, Trilla E, Lopez MA, et al. Analysis of the lipid profile and atherogenic risk during androgen deprivation therapy in prostate cancer patients. Urol Int. 2013;90:41–4.
Eri LM, Haug E, Tveter KJ. Effects on the endocrine system of long-term treatment with the non-steroidal anti-androgen Casodex in patients with benign prostatic hyperplasia. Br J Urol. 1995;75:335–40.
Smith MR, Finkelstein JS, McGovern FJ, Zietman AL, Fallon MA, Schoenfeld DA, et al. Changes in body composition during androgen deprivation therapy for prostate cancer. J Clin Endocrinol Metab. 2002;87:599–603.
Weinheimer EM, Sands LP, Campbell WW. A systematic review of the separate and combined effects of energy restriction and exercise on fat-free mass in middle-aged and older adults: implications for sarcopenic obesity. Nutr Rev. 2010;68:375–88.
Cava E, Yeat NC, Mittendorfer B. Preserving healthy muscle during weight loss. Adv Nutr. 2017;8:511–9.
Cormie P, Galvao DA, Spry N, Joseph D, Chee R, Taaffe DR, et al. Can supervised exercise prevent treatment toxicity in patients with prostate cancer initiating androgen-deprivation therapy: a randomised controlled trial. BJU Int. 2015;115:256–66.
Nobes JP, Langley SE, Klopper T, Russell-Jones D, Laing RW. A prospective, randomized pilot study evaluating the effects of metformin and lifestyle intervention on patients with prostate cancer receiving androgen deprivation therapy. BJU Int. 2012;109:1495–502.
Yunfeng G, Weiyang H, Xueyang H, Yilong H, Xin G. Exercise overcome adverse effects among prostate cancer patients receiving androgen deprivation therapy: an update meta-analysis. Medicine (Baltimore). 2017;96:e7368.
Acknowledgements
This study was funded by the Atkins Foundation, AUA, NIH K24 CA160653.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Freedland, S.J., Howard, L., Allen, J. et al. A lifestyle intervention of weight loss via a low-carbohydrate diet plus walking to reduce metabolic disturbances caused by androgen deprivation therapy among prostate cancer patients: carbohydrate and prostate study 1 (CAPS1) randomized controlled trial. Prostate Cancer Prostatic Dis 22, 428–437 (2019). https://doi.org/10.1038/s41391-019-0126-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-019-0126-5
This article is cited by
-
Plant-based diets to reduce prostate cancer risk and improve prostate cancer outcomes—ready for prime time?
Prostate Cancer and Prostatic Diseases (2023)
-
Dietary interventions to improve body composition in men treated with androgen deprivation therapy for prostate cancer: a solution for the growing problem?
Prostate Cancer and Prostatic Diseases (2022)