Abstract
Introduction
Epidemiological evidence suggests an association between CS and offspring metabolic syndrome (MetS), but whether a causal relationship exists is unknown.
Methods
In this study, timed-mated Wistar rat dams were randomly assigned to cesarean section (CS), vaginal delivery (VD), and surrogate groups. The offspring from both CS and VD groups were reared by surrogate dams until weaning, and weaned male offspring from both groups were randomly assigned to receive normal diet (ND) or high-fat/high-fructose diet (HFF) ad libitum for 39 weeks.
Results
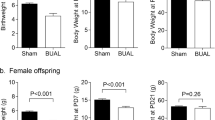
By the end of study, CS-ND offspring gained 17.8% more weight than VD-ND offspring, while CS-HFF offspring gained 36.4% more weight than VD-HFF offspring. Compared with VD-ND offspring, CS-ND offspring tended to have increased triglycerides (0.27 mmol/l, 95% CI, 0.05 to 0.50), total cholesterol (0.30 mmol/l, −0.08 to 0.68), and fasting plasma glucose (FPG) (0.30 mmol/l, −0.01 to 0.60); more pronounced differences were observed between CS-HFF and VD-HFF offspring in these indicators (triglyceride, 0.66 mmol/l, 0.35 to 0.97; total cholesterol, 0.46 mmol/l, 0.13 to 0.79; and FPG, 0.55 mmol/l, 0.13 to 0.98).
Conclusions
CS offspring were more prone to adverse metabolic profile and HFF might exacerbate this condition, indicating the association between CS and MetS is likely to be causal.
Impact
-
Whether the observed associations between CS and MetS in non-randomized human studies are causally relevant remains undetermined.
-
Compared with vaginally born offspring rats, CS born offspring gained more body weight and tended to have compromised lipid profiles and abnormal insulin sensitivity, suggesting a causal relationship between CS and MetS that may be further amplified by a high-fat/high-fructose diet.
-
Due to the high prevalence of CS births globally, greater clinical consideration must be given to the potential adverse effects of CS, and whether these risks should be made known to patients in clinical practice merits evaluation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
References
DeclercqE, Y., Cabral, H. & Ecker, J. Is a rising cesarean delivery rate inevitable? Trends in industrialized countries, 1987 to 2007. Birth 38, 99–104 (2011).
Boerma, T. et al. Global epidemiology of use of and disparities in caesarean sections. Lancet 392, 1341–1348 (2018).
Betran, A. P., Ye, J., Moller, A. B., Souza, J. P. & Zhang, J. Trends and projections of caesarean section rates: global and regional estimates. BMJ Glob. Health 6, e005671 (2021).
Li, H. T., Hellerstein, S., Zhou, Y. B., Liu, J. M. & Blustein, J. Trends in cesarean delivery rates in China, 2008-2018. JAMA 323, 89–91 (2020).
Sandall, J. et al. Short-term and long-term effects of caesarean section on the health of women and children. Lancet 392, 1349–1357 (2018).
Li, H. T., Zhou, Y. B. & Liu, J. M. The impact of cesarean section on offspring overweight and obesity: a systematic review and meta-analysis. Int. J. Obes. 37, 893–899 (2013).
Darmasseelane, K., Hyde, M. J., Santhakumaran, S., Gale, C. & Modi, N. Mode of delivery and offspring body mass index, overweight and obesity in adult life: a systematic review and meta-analysis. PLoS ONE 9, e87896 (2014).
Kuhle, S., Tong, O. S. & Woolcott, C. G. Association between caesarean section and childhood obesity: a systematic review and meta-analysis. Obes. Rev. 16, 295–303 (2015).
Kassi, E., Pervanidou, P., Kaltsas, G. & Chrousos, G. Metabolic syndrome: definitions and controversies. BMC Med. 9, 48 (2011).
Eckel, R. H., Grundy, S. M. & Zimmet, P. Z. The metabolic syndrome. Lancet 365, 1415–1428 (2005).
Zhou, Y. B. et al. Association of elective cesarean delivery with metabolic measures in childhood: a prospective cohort study in China. Nutr. Metab. Cardiovasc. Dis. 29, 775–782 (2019).
Bouhanick, B. et al. Mode of delivery at birth and the metabolic syndrome in midlife: the role of the birth environment in a prospective birth cohort study. BMJ Open 4, e005031 (2014).
Boney, C. M., Verma, A., Tucker, R. & Vohr, B. R. Metabolic syndrome in childhood: association with birth weight, maternal obesity, and gestational diabetes mellitus. Pediatrics 115, e290–e296 (2005).
Hrudey, E. J., Reynolds, R. M., Oostvogels, A. J., Brouwer, I. A. & Vrijkotte, T. G. The association between maternal 25-hydroxyvitamin D concentration during gestation and early childhood cardio-metabolic outcomes: is there interaction with pre-pregnancy BMI? PLoS ONE 10, e0133313 (2015).
Fraser, A. et al. Association of maternal weight gain in pregnancy with offspring obesity and metabolic and vascular traits in childhood. Circulation 121, 2557–2564 (2010).
Koskinen, J. et al. Childhood age and associations between childhood metabolic syndrome and adult risk for metabolic syndrome, type 2 diabetes mellitus and carotid intima media thickness: the International Childhood Cardiovascular Cohort Consortium. J. Am. Heart Assoc. 6, e005632 (2017).
Agodi, A. et al. Association of dietary patterns with metabolic syndrome: results from the Kardiovize Brno 2030 Study. Nutrients 10, 898 (2018).
Weiss, R., Bremer, A. A. & Lustig, R. H. What is metabolic syndrome, and why are children getting it? Ann. N. Y. Acad. Sci. 1281, 123–140 (2013).
García-Montero, C. et al. Nutritional components in Western diet Versus Mediterranean diet at the gut microbiota-immune system interplay. Implications for health and disease. Nutrients 13, 699 (2021).
Boulangé, C. L., Neves, A. L., Chilloux, J., Nicholson, J. K. & Dumas, M. E. Impact of the gut microbiota on inflammation, obesity, and metabolic disease. Genome Med. 8, 42 (2016).
MacLachlan, J., Wotherspoon, A. T., Ansell, R. O. & Brooks, C. J. Cholesterol oxidase: sources, physical properties and analytical applications. J. Steroid Biochem. Mol. Biol. 72, 169–195 (2000).
Sugiuchi, H. et al. Direct measurement of high-density lipoprotein cholesterol in serum with polyethylene glycol-modified enzymes and sulfated alpha-cyclodextrin. Clin. Chem. 41, 717–723 (1995).
Rifai, N., Iannotti, E., DeAngelis, K. & Law, T. Analytical and clinical performance of a homogeneous enzymatic Ldl-cholesterol assay compared with the ultracentrifugation-dextran sulfate-Mg2+ method. Clin. Chem. 44, 1242–1250 (1998).
Kagan, A. Radioimmunoassay of insulin. Semin. Nucl. Med. 5, 183–188 (1975).
Khan, A. I., Vasquez, Y., Gray, J., Wians, F. H. Jr & Kroll, M. H. The variability of results between point-of-care testing glucose meters and the central laboratory analyzer. Arch. Pathol. Lab. Med. 130, 1527–1532 (2006).
Matthews, D. R. et al. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28, 412–419 (1985).
UCLA Statistical Consulting Group. Repeated Measures Analysis Using SAS. https://stats.oarc.ucla.edu/sas/seminars/sas-repeatedmeasures/. (2011)
Martinez, K. A. 2nd et al. Increased weight gain by C-section: functional significance of the primordial microbiome. Sci. Adv. 3, eaao1874 (2017).
Castillo-Ruiz, A., Mosley, M., Jacobs, A. J., Hoffiz, Y. C. & Forger, N. G. Birth delivery mode alters perinatal cell death in the mouse brain. Proc. Natl Acad. Sci. USA 115, 11826–11831 (2018).
Rader, D. J. & Hovingh, G. K. Hdl and cardiovascular disease. Lancet 384, 618–625 (2014).
Li, J., Wu, H., Liu, Y. & Yang, L. High fat diet induced obesity model using four strainsof mice: kunming, C57bl/6, Balb/C and Icr. Exp. Anim. 69, 326–335 (2020).
Blázquez, E. & Quijada, C. L. The effect of a high-fat diet on glucose, insulin sensitivity and plasma insulin in rats. J. Endocrinol. 42, 489–494 (1968).
Minguez-Alarcon, L. et al. Cesarean delivery and metabolic health and inflammation biomarkers during mid-childhood and early adolescence. Pediatr. Res. 91, 672–680 (2022).
Ikeda, K., Onimaru, H., Matsuura, T. & Kawakami, K. Different impacts on brain function depending on the mode of delivery. Brain Res. 1720, 146289 (2019).
Pei, H., Sutton, A. K., Burnett, K. H., Fuller, P. M. & Olson, D. P. Avp neurons in the paraventricular nucleus of the hypothalamus regulate feeding. Mol. Metab. 3, 209–215 (2014).
Koenig, J. E. et al. Succession of microbial consortia in the developing infant gut microbiome. Proc. Natl Acad. Sci. USA 108, 4578–4585 (2011).
Penders, J. et al. Factors influencing the composition of the intestinal microbiota in early infancy. Pediatrics 118, 511–521 (2006).
Jakobsson, H. E. et al. Decreased gut microbiota diversity, delayed bacteroidetes colonisation and reduced Th1 responses in infants delivered by caesarean section. Gut 63, 559–566 (2014).
Ridaura, V. K. et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 341, 1241214 (2013).
Cox, L. M. et al. Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences. Cell 158, 705–721 (2014).
Zanardo, V. et al. Elective cesarean delivery: does it have a negative effect on breastfeeding? Birth 37, 275–279 (2010).
Li, L., Wan, W. & Zhu, C. Breastfeeding after a cesarean section: a literature review. Midwifery 103, 103117 (2021).
Wisnieski, L., Kerver, J., Holzman, C., Todem, D. & Margerison-Zilko, C. Breastfeeding and risk of metabolic syndrome in children and adolescents: a systematic review. J. Hum. Lact 34, 515–525 (2018).
O’Neill, S. & O’Driscoll, L. Metabolic syndrome: a closer look at the growing epidemic and its associated pathologies. Obes. Rev. 16, 1–12 (2015).
Lurie, S. et al. Different degrees of fetal oxidative stress in elective and emergent cesarean section. Neonatology 92, 111–115 (2007).
Hsu, C. N., Hou, C. Y., Hsu, W. H. & Tain, Y. L. Early-life origins of metabolic syndrome: mechanisms and preventive aspects. Int. J. Mol. Sci. 22, 11872 (2021).
Stinson, L. F., Payne, M. S. & Keelan, J. A. A critical review of the bacterial baptism hypothesis and the impact of cesarean delivery on the infant microbiome. Front. Med. 5, 135 (2018).
Boyle, E. M. et al. Effects of gestational age at birth on health outcomes at 3 and 5 years of age: population based cohort study. BMJ 344, e896 (2012).
Li, H. et al. Caesarean delivery, caesarean delivery on maternal request and childhood overweight: a Chinese birth cohort study of 181 380 children. Pediatr. Obes. 9, 10–16 (2014).
Chiesa, M. et al. Term or preterm cesarean section delivery does not lead to long-term detrimental consequences in mice. Cereb. Cortex 29, 2424–2436 (2019).
Chiesa, M., Ferrari, D. C. & Ben-Ari, Y. Alteration in the time and/or mode of delivery differentially modulates early development in mice. Mol. Brain 13, 34 (2020).
Hansen, C. H. et al. Mode of delivery shapes gut colonization pattern and modulates regulatory immunity in mice. J. Immunol. 193, 1213–1222 (2014).
Funding
This research was funded by the National Natural Science Foundation of China (grant number 81273163).
Author information
Authors and Affiliations
Contributions
HT Li and JM Liu conceived the study; HT Li, YB Zhou, MJ Liu and JM Liu proposed the initial research plan; MJ Liu and L Zhang conducted the animal experiment; MX Zhang and HT Li analyzed all data, performed all statistical analyses, and wrote the manuscript; all authors critically reviewed the manuscript; HT Li and YB Zhou had primary responsibility for the final content; all authors have read and agreed to the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The animal study protocol was approved by the animal Care and Use Committee of Peking University Health Science Center (SYXK (jing) 2011-0039).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, M., Liu, M., Zhang, L. et al. Impact of cesarean section on metabolic syndrome components in offspring rats. Pediatr Res (2024). https://doi.org/10.1038/s41390-024-03079-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41390-024-03079-6