Abstract
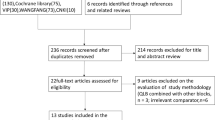
Pediatric analgesic trials are challenging, especially in newborns and infants. Following an FDA-academic consensus meeting, we analyzed pragmatic rescue designs in postoperative trials of local anesthetics, acetaminophen, opioids, and NSAIDs involving children ages 0–2 years and assessed surgical volumes to provide trial design recommendations. Searches of PubMed, Embase, CINAHL, The Cochrane Library, and Web of Science were conducted. A scoping approach identified trends in analgesic trials with an emphasis on randomized controlled trials (RCTs) utilizing immediate rescue designs. Age-specific surgical volumes were estimated from French national databases. Of 3563 studies identified, 23 RCTs used study medication(s) of interest and immediate rescue paradigms in children ages 0–2 years. A total of 270 studies met at least one of these criteria. Add-on and head-to-head designs were common and often used sparing of non-opioid or opioid rescue medication as a primary outcome measure. According to French national data, inguinal and penile surgeries were most frequent in ages 1 month to 2 years; abdominal and thoracic surgeries comprise approximately 75% of newborn surgeries. Analgesic trials with rescue sparing paradigm are currently sparse among children ages 0–2 years. Future trials could consider age-specific surgical procedures and use of add-on or head-to-head designs.
Impact
-
Clinical trials of analgesic medications have been challenging in pediatrics, especially in the group from newborns to 2 years of age. Following an FDA-academic workshop, we analyzed features of completed analgesic trials in this age group. Studies using immediate rescue in placebo control, add-on, and head-to-head trial designs are pragmatic approaches that can provide important information regarding clinical effectiveness, side effects, and safety. Using a French national dataset with a granular profile of inpatient, outpatient, and short-stay surgeries, we provide information to future investigators on relative frequencies of different operations in neonates and through the first 2 years of life.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Vickers, A. Why use placebos in clinical trials? A narrative review of the methodological literature. J. Clin. Epidemiol. 53, 157–161 (2000).
Berde, C. B. et al. Pediatric analgesic clinical trial designs, measures, and extrapolation: report of an FDA Scientific Workshop. Pediatrics 129, 354–364 (2012).
Kossowsky, J., Donado, C. & Berde, C. B. Immediate rescue designs in pediatric analgesic trials: a systematic review and meta-analysis. Anesthesiology 122, 150–171 (2015).
Eerdekens, M., Beuter, C., Lefeber, C. & van den Anker, J. The challenge of developing pain medications for children: therapeutic needs and future perspectives. J. Pain. Res. 12, 1649–1664 (2019).
Kaye, D. K. The ethical justification for inclusion of neonates in pragmatic randomized clinical trials for emergency newborn care. BMC Pediatr. 19, 218 (2019).
Walco, G. A. et al. Clinical trial designs and models for analgesic medications for acute pain in neonates, infants, toddlers, children, and adolescents: ACTTION recommendations. Pain 159, 193–205 (2018).
Baer, G. R. & Nelson, R. M. Ethical challenges in neonatal research: summary report of the ethics group of the newborn drug development initiative. Clin. Ther. 28, 1399–1407 (2006).
US Food and Drug Administration. E11 clinical investigation of medicinal products in the pediatric population. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/e11-clinical-investigation-medicinal-products-pediatric-population (2000).
FDA-M CERSI. Analgesic Clinical Trial Designs, Extrapolation, and Endpoints in Patients from Birth to Less Than Two Years of Age Public Workshop October 13, 2021–October 14, 2021. FDA. https://www.fda.gov/drugs/news-events-human-drugs/fda-m-cersi-analgesic-clinical-trial-designs-extrapolation-and-endpoints-patients-birth-less-two (2021).
De Lima, J. & Carmo, K. B. Practical pain management in the neonate. Best. Pract. Res. Clin. Anaesthesiol. 24, 291–307 (2010).
Perry, M. et al. Neonatal pain: perceptions and current practice. Crit. Care Nurs. Clin. North Am. 30, 549–561 (2018).
Ryan, R., Davoren, J., Grant, H. & Delbridge, L. A 23-hour care centre model for the management of surgical patients. ANZ J. Surg. 74, 754–759 (2004).
Covidence – Better systematic review management. Covidence. https://www.covidence.org/.
Sunil, R., Jacob, M. & Lakshmi, K. Effect of bilateral infraorbital nerve block on intraoperative anesthetic requirements, hemodynamics, glycemic levels, and extubation in infants undergoing cheiloplasty under general anesthesia. J. Dent. Anesth. Pain. Med 21, 129 (2021).
Nour, C. et al. Analgesic effectiveness of acetaminophen for primary cleft palate repair in young children: a randomized placebo controlled trial. Pediatr. Anesth. 24, 574–581 (2014).
Penk, J. S. et al. Intermittent versus continuous and intermittent medications for pain and sedation after pediatric cardiothoracic surgery; a randomized controlled trial. Crit. Care Med. 46, 123–129 (2018).
Chiono, J. & Bigorre, M. Bilateral suprazygomatic maxillary nerve block for cleft palate repair in children. Anesth 120, 1362–1369 (2014).
Olischar, M. et al. The addition of tramadol to the standard of IV acetaminophen and morphine infusion for postoperative analgesia in neonates offers no clinical benefit: a randomized placebo-controlled trial. Pediatr. Anesth. 24, 1149–1157 (2014).
Hammer, G. B. et al. Randomized population pharmacokinetic analysis and safety of intravenous acetaminophen for acute postoperative pain in neonates and infants. J. Clin. Pharmacol. 60, 16–27 (2020).
Ceelie, I. et al. Effect of intravenous paracetamol on postoperative morphine requirements in neonates and infants undergoing major noncardiac surgery: a randomized controlled trial. JAMA 309, 149–154 (2013).
Rabie, A., Ghoneim, T. & El-Rouby, A. Paravertebral versus single shot epidural blockade for neonates undergoing thoracotomy. Egypt J. Anaesth. 36, 147–152 (2020).
Lorenzo, A. J., Lynch, J., Matava, C., El-Beheiry, H. & Hayes, J. Ultrasound guided transversus abdominis plane vs surgeon administered intraoperative regional field infiltration with bupivacaine for early postoperative pain control in children undergoing open pyeloplasty. J. Urol. 192, 207–213 (2014).
Abu Elyazed, M. M. & Mostafa, S. F. Bilateral suprazygomatic maxillary nerve block versus palatal block for cleft palate repair in children: a randomized controlled trial. Egypt J. Anaesth. 34, 83–88 (2018).
Dehghan, K., Ghalizadeh, M., Macooie, A. & Foroutan, H. The comparison of the analgesic effect of intravenous acetaminophen with fentanyl in thoracic and abdominal surgeries of newborns. Int. J. Pediatr. 7, 9773–9781 (2019).
Sethi, S., Ghai, B., Sen, I., Ram, J. & Wig, J. Efficacy of subtenon block in infants – a comparison with intravenous fentanyl for perioperative analgesia in infantile cataract surgery. Pediatr. Anesth. 23, 1015–1020 (2013).
Singh, S., Singh, V., Goel, P., Maitra, S. & Rawat, J. Randomised controlled trial to compare the efficacy of epidural analgesia versus intravenous analgesia during thoracotomy for repair of oesophageal atresia. J. Clin. Diagn. Res. 13, UC01–UC05 (2019).
Rabie, A. & Ghoneim, T. Ultrasound guided continuous caudal epidural analgesia in neonates undergoing thoracotomy. Egypt J. Anaesth. 37, 208–213 (2021).
Rabie, A., Ghoneim, T. & Saad El-Rouby, A. Single-shot thoracic epidural analgesia for neonates undergoing thoracotomy. Egypt J. Anaesth. 36, 282–287 (2020).
Yildirim, S. A., Ozcan, H. & Turkoz, A. Ultrasound-guided penile block for circumcision instead of anatomical landmark method in newborn babies. Ain-Shams J. Anesthesiol. 13, 39 (2021).
Xiang, K., Cai, H. & Song, Z. Comparison of analgesic effects of remifentanil and fentanyl NCA after pediatric cardiac surgery. J. Invest. Surg. 27, 214–218 (2014).
Solanki, N., Engineer, S. & Vecham, P. Comparison of epidural versus systemic analgesia for major surgeries in neonates and infants. J. Clin. Neonatol. 6, 23 (2017).
Abdellatif, A. A., Elagamy, A. E. & Elgazzar, K. Ultrasound-guided infraorbital nerve block for cleft lip repair in pediatrics: a new technique for an old block. Ain-Shams J. Anesthesiol. 10, 3 (2018).
Ponde, V., Desai, A. P. & Shah, D. Comparison of success rate of ultrasound-guided sciatic and femoral nerve block and neurostimulation in children with arthrogryposis multiplex congenita: a randomized clinical trial. Pediatr. Anesth. 23, 74–78 (2013).
Anell-Olofsson, M. et al. Plasma concentrations of levobupivacaine associated with two different intermittent wound infusion regimens following surgical ductus ligation in preterm infants. Pediatr. Anesth. 25, 711–718 (2015).
El-Emam, E. S., & El motlb, E. A. A. Ultrasound-guided erector spinae versus ilioinguinal/iliohypogastric block for postoperative analgesia in children undergoing inguinal surgeries. Anesth. Essays Res. 13, 274 (2019).
Ozer-Cinar, A. S., Isil, C. T., Hekimoglu-Sahin, S. & Paksoy, İ. Caudal ropivacaine and bupivacaine for postoperative analgesia in infants undergoing lower abdominal surgery. Pak. J. Med. Sci. 31, 903–908 (2015).
Eissa, A. et al. Tapentadol for the treatment of moderate-to-severe acute pain in children under the age of two years. J. Pain. Res. 14, 229–248 (2021).
Lynn, A. M. et al. Postoperative ketorolac tromethamine use in infants aged 6–18 months: the effect on morphine usage, safety assessment, and stereo-specific pharmacokinetics. Anesth. Analg. 104, 1040–1051 (2007).
Moffett, B. S., Wann, T. I., Carberry, K. E. & Mott, A. R. Safety of ketorolac in neonates and infants after cardiac surgery. Pediatr. Anesth. 16, 424–428 (2006).
Ziesenitz, V. C. et al. Efficacy and safety of NSAIDs in infants: a comprehensive review of the literature of the past 20 years. Pediatr. Drugs 24, 603–655 (2022).
Aldrink, J. H. et al. Safety of ketorolac in surgical neonates and infants 0 to 3 months old. J. Pediatr. Surg. 46, 1081–1085 (2011).
Eerdekens, M. et al. Outcomes of the pediatric development plan of tapentadol. J. Pain. Res. 14, 249–261 (2021).
Beverly, A., Kaye, A. D., Ljungqvist, O. & Urman, R. D. Essential elements of multimodal analgesia in Enhanced Recovery After Surgery (ERAS) Guidelines. Anesthesiol. Clin. 35, 115–143 (2017).
Bednarski, B. K. et al. Randomized clinical trial of accelerated enhanced recovery after minimally invasive colorectal cancer surgery (RecoverMI trial). Br. J. Surg. 106, 1311–1318 (2019).
Acknowledgements
We are thankful for Claire McEwen, Jillian Rhodes, and Jacob Zhang for their significant contributions to study screening and data extraction. We would also like to acknowledge Dr Sophie Tezenas du Montcel and Dr Aurélia Retbi from the Department of Medical Information at Saint Antoine Hospital in Paris for their retrieval and organization of the surgical data from the PMSI. Supported in part by the Sara Page Mayo Endowment for Pediatric Pain Research.
Author information
Authors and Affiliations
Contributions
Substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data: K.H., C.D., R.C., J.K., C.B. Drafting the article or revising it critically for important intellectual content: K.H., C.D., R.C., J.K., C.B. Final approval of the version to be published: K.H., C.D., R.C., J.K., C.B.
Corresponding author
Ethics declarations
Competing interests
All authors declare: no support from any organization for the submitted work; no financial relationships with any organizations that might have an interest in the submitted work; no other relationships or activities that could appear to have influenced the submitted work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Haskes, K., Donado, C., Carbajal, R. et al. Rescue designs in analgesic trials from 0 to 2 years of age: scoping review. Pediatr Res 95, 1237–1245 (2024). https://doi.org/10.1038/s41390-023-02897-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02897-4