Abstract
Background
Perinatal brain injury is multifactorial and primarily associated with brain prematurity, inflammation, and hypoxia–ischemia. Although recent advances in perinatal medicine have improved the survival rates of preterm infants, neurodevelopmental disorders remain a significant complication. We tested whether the intravenous infusion of mesenchymal stem cells (MSCs) had therapeutic efficacy against perinatal brain injury in rats.
Methods
Pregnant rats at embryonic day (E) 18 received lipopolysaccharide and the pups were born at E21. On postnatal day (PND) 7, the left common carotid artery of each pup was ligated, and they were exposed to 8% oxygen for 2 h. They were randomized on PND10, and MSCs or vehicle were intravenously infused. We performed behavioral assessments, measured brain volume using MRI, and performed histological analyses on PND49.
Results
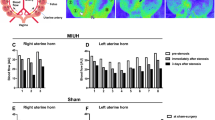
Infused MSCs showed functional improvements in our model. In vivo MRI revealed that MSC infusion increased non-ischemic brain volume compared to the vehicle group. Histological analyses showed that cortical thickness, the number of NeuN+ and GAD67+ cells, and synaptophysin density in the non-ischemic hemisphere in the MSC group were greater than the vehicle group, but less than the control group.
Conclusions
Infused MSCs improve sensorimotor and cognitive functions in perinatal brain injury and enhance neuronal growth.
Impact
-
Intravenous infusion of MSCs improved neurological function in rats with perinatal brain injury, including motor, sensorimotor, cognitive, spatial, and learning memory.
-
Infused MSCs increased residual (non-ischemic) tissue volume, number of neuronal cells, GABAergic cells, and cortical synapses in the contralesional (right) hemisphere.
-
Intravenous administration of MSC might be suitable for the treatment of perinatal brain injury.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author on reasonable request.
References
du Plessis, A. J. & Volpe, J. J. Perinatal brain injury in the preterm and term newborn. Curr. Opin. Neurol. 15, 151–157 (2002).
Jain, V. G., Willis, K. A., Jobe, A. & Ambalavanan, N. Chorioamnionitis and neonatal outcomes. Pediatr. Res. 91, 289–296 (2022).
Wu, Y. W. et al. Chorioamnionitis and cerebral palsy in term and near-term infants. JAMA 290, 2677–2684 (2003).
Novak, C. M., Ozen, M. & Burd, I. Perinatal brain injury: mechanisms, prevention, and outcomes. Clin. Perinatol. 45, 357–375 (2018).
Hagberg, H. et al. The role of inflammation in perinatal brain injury. Nat. Rev. Neurol. 11, 192–208 (2015).
Sakai, T. et al. Functional recovery after the systemic administration of mesenchymal stem cells in a rat model of neonatal hypoxia-ischemia. J. Neurosurg. Pediatr. 22, 513–522 (2018).
Zhao, Y. et al. Lipopolysaccharides (LPSs) as potent neurotoxic glycolipids in Alzheimer’s disease (AD). Int. J. Mol. Sci. 23, 12671 (2022).
Sisa, C. et al. Properdin: a novel target for neuroprotection in neonatal hypoxic-ischemic brain injury. Front. Immunol. 10, 2610 (2019).
Hirota, R. et al. Enhanced network in corticospinal tracts after infused mesenchymal stem cells in spinal cord injury. J. Neurotrauma 39, 1665–1677 (2022).
Morita, T. et al. Intravenous infusion of mesenchymal stem cells promotes functional recovery in a model of chronic spinal cord injury. Neuroscience 335, 221–231 (2016).
Kim, S. et al. Neural differentiation potential of peripheral blood- and bone-marrow-derived precursor cells. Brain Res. 1123, 27–33 (2006).
Takemura, M. et al. Repeated intravenous infusion of mesenchymal stem cells for enhanced functional recovery in a rat model of chronic cerebral ischemia. J. Neurosurg. https://doi.org/10.3171/2021.8.Jns21687 (2021).
Leem, Y.-H., Park, J.-S., Park, J.-E., Kim, D.-Y. & Kim, H.-S. Neurogenic effects of rotarod walking exercise in subventricular zone, subgranular zone, and substantia nigra in MPTP-induced Parkinson’s disease mice. Sci. Rep. 12, 10544 (2022).
Lehnerer, V. et al. Mesenchymal stem cell therapy in perinatal arterial ischemic stroke: systematic review of preclinical studies. Pediatr. Res. https://doi.org/10.1038/s41390-022-02208-3 (2022).
Cai, Z., Pan, Z. L., Pang, Y., Evans, O. B. & Rhodes, P. G. Cytokine induction in fetal rat brains and brain injury in neonatal rats after maternal lipopolysaccharide administration. Pediatr. Res. 47, 64–72 (2000).
Nakazaki, M. et al. Prevention of neointimal hyperplasia induced by an endovascular stent via intravenous infusion of mesenchymal stem cells. J. Neurosurg. 133, 1–13 (2019).
Naiki, Y. et al. TGF-β1 inhibits the production of IFN in response to CpG DNA via ubiquitination of TNF receptor-associated factor (TRAF) 6. Innate Immun. 21, 770–777 (2015).
Acknowledgements
We are thankful to the National BioResource Project - Rat (http://www.anim.med.kyoto-u.ac.jp/NBR/) for providing this rat strain (W-Tg (CAG-GFP) 184Ys).
Funding
This work was supported in part by the JSPS KAKENHI grant (grant number 20K08163) and the RR&D Service of the US Department of Veterans Affairs (grants B7335R and B9260L).
Author information
Authors and Affiliations
Contributions
Each author has met the Pediatric Research authorship requirements. Each author completed the following criteria: (1) substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; (2) drafting the article or revising it critically for important intellectual content; and (3) final approval of the version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Terada, K., Sasaki, M., Nagahama, H. et al. Therapeutic efficacy of intravenous infusion of mesenchymal stem cells in rat perinatal brain injury. Pediatr Res 94, 1921–1928 (2023). https://doi.org/10.1038/s41390-023-02717-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02717-9