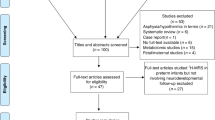
Abstract
White matter (WM) injury is the most common type of brain injury in preterm infants and is associated with impaired neurodevelopmental outcome (NDO). Currently, there are no treatments for WM injury, but optimal nutrition during early preterm life may support WM development. The main aim of this scoping review was to assess the influence of early postnatal nutrition on WM development in preterm infants. Searches were performed in PubMed, EMBASE, and COCHRANE on September 2022. Inclusion criteria were assessment of preterm infants, nutritional intake before 1 month corrected age, and WM outcome. Methods were congruent with the PRISMA-ScR checklist. Thirty-two articles were included. Negative associations were found between longer parenteral feeding duration and WM development, although likely confounded by illness. Positive associations between macronutrient, energy, and human milk intake and WM development were common, especially when fed enterally. Results on fatty acid and glutamine supplementation remained inconclusive. Significant associations were most often detected at the microstructural level using diffusion magnetic resonance imaging. Optimizing postnatal nutrition can positively influence WM development and subsequent NDO in preterm infants, but more controlled intervention studies using quantitative neuroimaging are needed.
Impact
-
White matter brain injury is common in preterm infants and associated with impaired neurodevelopmental outcome.
-
Optimizing postnatal nutrition can positively influence white matter development and subsequent neurodevelopmental outcome in preterm infants.
-
More studies are needed, using quantitative neuroimaging techniques and interventional designs controlling for confounders, to define optimal nutritional intakes in preterm infants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Ortinau, C. & Neil, J. The neuroanatomy of prematurity: normal brain development and the impact of preterm birth. Clin. Anat. 28, 168–183 (2015).
Pascal, A. et al. Neurodevelopmental outcome in very preterm and very-low-birthweight infants born over the past decade: a meta-analytic review. Dev. Med. Child Neurol. 60, 342–355 (2018).
Wilson, S. et al. Development of human white matter pathways in utero over the second and third trimester. Proc. Natl Acad. Sci. USA 118, 1–7 (2021).
van Tilborg, E. et al. Origin and dynamics of oligodendrocytes in the developing brain: Implications for perinatal white matter injury. Glia 66, 221–238 (2018).
Back, S. A. White matter injury in the preterm infant: pathology and mechanisms. Acta Neuropathol 134, 331–349 (2017).
Volpe, J. J. Brain injury in premature infants: a complex amalgam of destructive and developmental disturbances. Lancet Neurol. 8, 110–124 (2009).
Cayam-Rand, D. et al. Predicting developmental outcomes in preterm infants: a simple white matter injury imaging rule. Neurology 93, E1231–E1240 (2019).
Back, S. A. & Miller, S. P. Brain injury in premature neonates: a primary cerebral dysmaturation disorder? Ann. Neurol. 75, 469–486 (2014).
Elitt, C. M. & Rosenberg, P. A. The challenge of understanding cerebral white matter injury in the premature infant. Neuroscience 276, 216–238 (2014).
Cusick, S. E. & Georgieff, M. K. The role of nutrition in brain development: the golden opportunity of the “First 1000 Days”. J. Pediatr. 175, 16–21 (2016).
Keunen, K., Van Elburg, R. M., Van Bel, F. & Benders, M. J. N. L. Impact of nutrition on brain development and its neuroprotective implications following preterm birth. Pediatr. Res. 77, 148–155 (2015).
Volpe, J. J. Dysmaturation of premature brain: importance, cellular mechanisms, and potential interventions. Pediatr. Neurol. 95, 42–66 (2019).
Fink, N. H., Collins, C. T., Gibson, R. A., Makrides, M. & Penttila, I. A. Targeting inflammation in the preterm infant: the role of the omega-3 fatty acid docosahexaenoic acid. J. Nutr. Intermed. Metab. 5, 55–60 (2016).
Garofalo, R. Cytokines in human milk. J. Pediatr. 156, S36–S40 (2010).
Eyles, D., Burne, T. & Mcgrath, J. Vitamin D in fetal brain development. Semin. Cell Dev. Biol. 22, 629–636 (2011).
Jarosz, M., Olbert, M., Wyszogrodzka, G., Młyniec, K. & Librowski, T. Antioxidant and anti-inflammatory effects of zinc. Zinc-dependent NF-κB signaling. Inflammopharmacology 25, 11–24 (2017).
Georgieff, M. K. Nutrition and the developing brain: nutrient priorities and measurement. Am. J. Clin. Nutr. 85, 614–620 (2007).
Strømmen, K. et al. Enhanced nutrient supply to very low birth weight infants is associated with improved white matter maturation and head growth. Neonatology 107, 68–75 (2015).
Schneider, J. et al. Nutrient intake in the first two weeks of life and brain growth in preterm neonates. Pediatrics 141, e20172169 (2018).
Coviello, C. et al. Effects of early nutrition and growth on brain volumes, white matter microstructure, and neurodevelopmental outcome in preterm newborns. Pediatr. Res. 83, 102–110 (2018).
Mudd, A. T. & Dilger, R. N. Early-life nutrition and neurodevelopment: use of the piglet as a translational model. Adv. Nutr. 8, 92–104 (2017).
Chan, S. H. T., Johnson, M. J., Leaf, A. A. & Vollmer, B. Nutrition and neurodevelopmental outcomes in preterm infants: a systematic review. Acta Paediatr. 105, 587–599 (2016).
Belfort, M. B. & Ehrenkranz, R. A. Neurodevelopmental outcomes and nutritional strategies in very low birth weight infants. Semin. Fetal Neonatal Med. 22, 42–48 (2017).
Ottolini, K. M., Andescavage, N., Keller, S. & Limperopoulos, C. Nutrition and the developing brain: the road to optimizing early neurodevelopment: a systematic review. Pediat. Res. 87, 194–201 (2020)
Hortensius, L. M., Van Elburg, R. M., Nijboer, C. H., Benders, M. J. N. L. & De Theije, C. G. M. Postnatal nutrition to improve brain development in the preterm infant: A systematic review from bench to bedside. Front. Physiol. 10, 1–18 (2019).
Peters, M.D.J. et al. Chapter 11: Scoping Reviews (2020 version). In: Aromataris E, Munn Z Editors. JBI Manual for Evidence Synthesis, JBI, 2020
Tricco, A. C. et al. PRISMA extension for scoping reviews (PRISMA-ScR): checklist and explanation. Ann. Intern. Med. 169, 467–473 (2018).
Mueen Ahmed, K. K. & Al Dhubaib, B. E. Zotero: a bibliographic assistant to researcher. J. Pharm. Pharmacother. 2, 303–305 (2011).
Ouzzani, M., Hammady, H., Fedorowicz, Z. & Elmagarmid, A. Rayyan-a web and mobile app for systematic reviews. Syst. Rev. 5, 1–10 (2016).
Brouwer, M. J. et al. Preterm brain injury on term-equivalent age MRI in relation to perinatal factors and neurodevelopmental outcome at two years. PLoS One 12, 1–13 (2017).
Barnett, M. L. et al. Exploring the multiple-hit hypothesis of preterm white matter damage using diffusion MRI. Neuroimage Clin. 17, 596–606 (2018).
Parikh, N. A., Lasky, R. E., Kennedy, K. A., McDavid, G. & Tyson, J. E. Perinatal factors and regional brain volume abnormalities at term in a cohort of extremely low birth weight infants. PLoS One 8, e62804 (2013).
Parikh, N. A., He, L., Li, H., Priyanka Illapani, V. S. & Klebanoff, M. A. Antecedents of objectively diagnosed diffuse white matter abnormality in very preterm infants. Pediatr. Neurol. 106, 56–62 (2020).
Parikh, N. A. et al. Perinatal risk and protective factors in the development of diffuse white matter abnormality on term-equivalent age magnetic resonance imaging in infants born very preterm. J. Pediatr. 233, 58–65.e3 (2021).
Rogers, CE., Smyser, T., Smyser, CD., Shimony, J., Inder, TE. & Neil, JJ. Regional white matter development in very preterm infants: perinatal predictors and early developmental outcomes. Physiol. Behav. 176, 100–106 (2016).
Blesa, M. et al. Early breast milk exposure modifies brain connectivity in preterm infants. Neuroimage 184, 431–439 (2019).
Belfort, M. B. et al. Breast milk feeding, brain development, and neurocognitive outcomes: a 7-year longitudinal study in infants born at less than 30 weeks’ gestation. J. Pediatr. 177, 133–139.e1 (2016).
Isaacs, E. B. et al. Impact of breast milk on intelligence quotient, brain size, and white matter development. Pediatr. Res. 67, 357–362 (2010).
Bell, K. A. et al. Associations of macronutrient intake determined by point-of-care human milk analysis with brain development among very preterm infants. Children (Basel) 9, 969 (2022).
Ottolini, K. M., Andescavage, N., Kapse, K., Jacobs, M. & Limperopoulos, C. Improved brain growth and microstructural development in breast milk–fed very low birth weight premature infants. Acta Paediatr. 109, 1580–1587 (2020).
Pogribna, U. et al. Perinatal clinical antecedents of white matter microstructural abnormalities on diffusion tensor imaging in extremely preterm infants. PLoS One 8, e72974 (2013).
Sato, J. et al. Early nutrition and white matter microstructure in children born very low birth weight. Brain Commun. 3, 1–12 (2021).
Keller, T. et al. Intranasal breast milk for premature infants with severe intraventricular hemorrhage—an observation. Eur. J. Pediatr. 178, 199–206. https://doi.org/10.1007/s00431-018-3279-7 (2019).
Terrin, G. et al. Early protein intake influences neonatal brain measurements in preterms: an observational study. Front. Neurol. 11, 885 (2020).
Boscarino, G. et al. Effects of early energy intake on neonatal cerebral growth of preterm newborn: an observational study. Sci. Rep. 11, 1–7 (2021).
Ottolini, K. M. et al. Early lipid intake improves cerebellar growth in very low-birth-weight preterm infants. J. Parenter. Enter. Nutr. 45, 587–595 (2021).
Power, V. A. et al. Nutrition, growth, brain volume, and neurodevelopment in very preterm children. J. Pediatr. 215, 50–55.e3 (2019).
Hansen-Pupp, I. et al. Postnatal decrease in circulating insulin-like growth factor-I and low brain volumes in very preterm infants. J. Clin. Endocrinol. Metab. 96, 1129–1135 (2011).
van Beek, P. E. et al. Increase in brain volumes after implementation of a nutrition regimen in infants born extremely preterm. J. Pediatr. 223, 57–63.e5 (2020).
Rozé, J. C. et al. Association between early amino acid intake and full-scale IQ at age 5 years among infants born at less than 30 weeks’ gestation. JAMA Netw. Open 4, e2135452. https://doi.org/10.1001/jamanetworkopen.2021.35452 (2021).
Hortensius, L. M. et al. Nutritional intake, white matter integrity, and neurodevelopment in extremely preterm born infants. Nutrients 13, 1–14 (2021).
Beauport, L. et al. Impact of early nutritional intake on preterm brain: a magnetic resonance imaging study. J. Pediatr. 181, 29–36.e1 (2017).
Tan, M., Abernethy, L. & Cooke, R. Improving head growth in preterm infants – a randomised controlled trial II: MRI and developmental outcomes in the first year. Arch. Dis. Child Fetal Neonatal Ed. 93, 342–346 (2008).
Isaacs, E. B. et al. The effect of early human diet on caudate volumes and IQ. Pediatr. Res. 63, 308–314 (2008).
Van Wezel-Meijler, G. et al. Dietary supplementation of long-chain polyunsaturated fatty acids in preterm infants: effects on cerebral maturation. Acta Paediatr. 91, 942–950 (2002).
Almaas, A. N. et al. Long-chain polyunsaturated fatty acids and cognition in VLBW infants at 8 years: An RCT. Pediatrics 135, 972–980 (2015).
Almaas, A. N. et al. Diffusion tensor imaging and behavior in premature infants at 8 years of age, a randomized controlled trial with long-chain polyunsaturated fatty acids. Early Hum. Dev. 95, 41–46 (2016).
De Kieviet, J. F. et al. Effects of glutamine on brain development in very preterm children at school age. Pediatrics 130, e1121–e1127 (2012).
Ehrenkranz, R. A. et al. Early nutrition mediates the influence of severity of illness on extremely LBW infants. Pediatr. Res. 69, 522–529 (2011).
Moltu, S. J. et al. Nutritional management of the critically ill neonate: a position paper of the ESPGHAN Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 73, 274–289 (2021).
Stensvold, H. J. et al. Early enhanced parenteral nutrition, hyperglycemia, and death among extremely low-birth-weight infants. JAMA Pediatr. 169, 1003–1010 (2015).
Nilsson, A. K. et al. Serum choline in extremely preterm infants declines with increasing parenteral nutrition. Eur. J. Nutr. 60, 1081–1089 (2021).
Asztalos, E. V., Church, P. T., Riley, P., Fajardo, C. & Shah, P. S. Neonatal factors associated with a good neurodevelopmental outcome in very preterm infants. Am. J. Perinatol. 34, 388–396 (2017).
Wojnar, M. M., Fan, J., Li, Y. H. & Lang, C. H. Endotoxin-induced changes in IGF-I differ in rats provided enteral vs. parenteral nutrition. Am. J. Physiol. 276, E455–E464 (1999).
Marcobal, A. et al. Consumption of human milk oligosaccharides by gut-related microbes. J. Agric. Food Chem. 58, 5334–5340 (2010).
Ayechu-Muruzabal, V. et al. Diversity of human milk oligosaccharides and effects on early life immune development. Front. Pediatr. 6, 1–9 (2018).
Kotey, F. O. & Spatz, D. L. White matter injury in preterm infants: could human milk play a role in its prevention? Adv. Neonatal Care 13, 89–94 (2013).
Quigley, M., Embleton, N. D. & McGuire, W. Formula versus maternal breast milk for feeding preterm or low birth weight infants. Cochrane Database Syst. Rev. 8, CD002972 (2019).
Chetta, K. E., Schulz, E. V. & Wagner, C. L. Outcomes improved with human milk intake in preterm and full-term infants. Semin. Perinatol. 45, 151384 (2021).
Dueñas-Espín, I. et al. Breastfeeding education, early skin-to-skin contact and other strong determinants of exclusive breastfeeding in an urban population: a prospective study. BMJ Open 11, 1–8 (2021).
Walfisch, A., Sermer, C., Cressman, A. & Koren, G. Breast milk and cognitive development-the role of confounders: a systematic review. BMJ Open 3, e003259 (2013).
Yumani, D. F. J., Calor, A. K. & van Weissenbruch, M. M. The course of IGF-1 levels and nutrient intake in extremely and very preterm infants during hospitalisation. Nutrients 12, 1–13 (2020).
Okuma, C. et al. Microstructural brain and multivoxel spectroscopy in very low birth weight infants related to insulin-like growth factor concentration and early growth. Horm. Res. Paediatr. 79, 197–207 (2013).
Hay, W. W. Aggressive nutrition of the preterm infant. Curr. Pediatr. Rep. 1. https://doi.org/10.1007/s40124-013-0026-4 (2013).
Cormack, B. E., Harding, J. E., Miller, S. P. & Bloomfield, F. H. The influence of early nutrition on brain growth and neurodevelopment in extremely preterm babies: a narrative review. Nutrients 11, 2029 (2019).
Balakrishnan, M. et al. Growth and neurodevelopmental outcomes of early high‐dose parenteral amino acid intake in very low birth weight infants: a randomized controlled trial. JPEN J Parenter Enteral Nutr. 42, 597–606 (2017)
Lapillonne, A. & Moltu, S. J. Long-chain polyunsaturated fatty acids and clinical outcomes of preterm infants. Ann. Nutr. Metab. 69, 36–44 (2016).
Sakamoto, T., Cansev, M. & Wurtman, R. J. Oral supplementation with docosahexaenoic acid and uridine-5′-monophosphate increases dendritic spine density in adult gerbil hippocampus. Brain Res. 1182, 50–59 (2007).
Farquharson, J., Cockburn, F., Patrick, W. A., Jamieson, E. C. & Logan, R. W. Infant cerebral cortex phospholipid fatty-acid composition and diet. Lancet 340, 810–813 (1992).
Makrides, M., Neumann, M. A., Byard, R. W., Simmer, K. & Gibson, R. A. Fatty acid composition of brain, retina, and erythrocytes in breast-and formula-fed infants. Am. J. Clin. Nutr. 60, 189–194 (1994).
Lapillonne, A., Groh-Wargo, S., Lozano Gonzalez, C. H. & Uauy, R. Lipid needs of preterm infants: updated recommendations. J. Pediatr. 162, S37–S47 (2013).
Hortensius, L. M. et al. Serum docosahexaenoic acid levels are associated with brain volumes in extremely preterm born infants. Pediatr. Res. 90, 1177–1185. https://doi.org/10.1038/s41390-021-01645-w (2021).
Tam, E. W. Y. et al. Early postnatal docosahexaenoic acid levels and improved preterm brain development. Pediatr. Res. 79, 723–730 (2016).
Fang, P. C. et al. The effect of supplementation of docosahexaenoic acid and arachidonic acid on visual acuity and neurodevelopment in larger preterm infants. Chang Gung Med. J. 28, 708–715 (2005).
Clandinin, M. T. et al. Growth and development of preterm infants fed infant formulas containing docosahexaenoic acid and arachidonic acid. J. Pediatr. 146, 461–468 (2005).
Makrides, M. et al. Neurodevelopmental outcomes of preterm infants fed high-dose docosahexaenoic acid – a randomized controlled. Trial 301, 175–182 (2015).
Sugasini, D., Yalagala, P. C. R., Goggin, A., Leon M. Tai, P. & Subbaiah, P. V. Enrichment of brain docosahexaenoic acid (DHA) is highly dependent upon the molecular carrier of dietary DHA: lysophosphatidylcholine is more efficient than either phosphatidylcholine or triacylglycerol. J. Nutr. Biochem. 74, 108231. https://doi.org/10.1159/000444169.Carotid (2019).
Klevebro, S., Juul, S. E. & Wood, T. R. A more comprehensive approach to the neuroprotective potential of long-chain polyunsaturated fatty acids in preterm infants is needed—should we consider maternal diet and the n-6:n-3 fatty acid ratio? Front. Pediatr. 7, 533 (2020).
Alshweki, A. et al. Effects of different arachidonic acid supplementation on psychomotor development in very preterm infants; a randomized controlled trial. Nutr. J. 14, 101 (2015).
Kamino, D. et al. Postnatal polyunsaturated fatty acids associated with larger preterm brain tissue volumes and better outcomes. Pediatr. Res. 83, 93–101 (2018).
Van Den Berg, A., Van Elburg, R. M., Westerbeek, E. A. M., Twisk, J. W. R. & Fetter, W. P. F. Glutamine-enriched enteral nutrition in very-low-birth-weight infants and effects on feeding tolerance and infectious morbidity: a randomized controlled trial. Am. J. Clin. Nutr. 81, 1397–1404 (2005).
Mok, E. & Hankard, R. Glutamine supplementation in sick children: is it beneficial? J. Nutr. Metab. 2011, 617597 (2011).
De Kieviet, J. F. et al. Glutamine effects on brain growth in very preterm children in the first year of life. Clin. Nutr. 33, 69–74 (2014).
van Zwol, A. et al. Neurodevelopmental outcomes of very low‐birth‐weight infants after enteral glutamine. Acta Paediatr. 97, 562–7 (2008)
De Kieviet, J. F. et al. Effects of neonatal enteral glutamine supplementation on cognitive, motor and behavioural outcomes in very preterm and/or very low birth weight children at school age. Br. J. Nutr. 108, 2215–2220 (2012).
Moe-Byrne, T., Brown, J. V. E. & Mcguire, W. Glutamine supplementation to prevent morbidity and mortality in preterm infants. Cochrane Database Syst. Rev. 2016, CD001457 (2016).
Rochow, N. et al. Individualized target fortification of breast milk with protein, carbohydrates, and fat for preterm infants: a double-blind randomized controlled trial. Clin. Nutr. 40, 54–63 (2021).
Fusch, S. et al. Individualized target fortification of breast milk: optimizing macronutrient content using different fortifiers and approaches. Front. Nutr. 8, 652641 (2021).
Cormack, B. E., Embleton, N. D., Van Goudoever, J. B., Hay, W. W. & Bloomfield, F. H. Comparing apples with apples: it is time for standardized reporting of neonatal nutrition and growth studies. Pediatr. Res. 79, 810–820 (2016).
Hauser, J., Sultan, S., Rytz, A., Steiner, P. & Schneider, N. A blend containing docosahexaenoic acid, arachidonic acid, vitamin B12, vitamin B9, iron and sphingomyelin promotes myelination in an in vitro model. Nutr. Neurosci. 23, 931–945 (2020).
Holguin, S., Martinez, J., Chow, C. & Wurtman, R. Dietary uridine enhances the improvement in learning and memory produced by administering DHA to gerbils. FASEB J. 22, 3938–3946 (2008).
Schneider, N. et al. A nutrient formulation affects developmental myelination in term infants: a randomized clinical trial. Front. Nutr. 9, 1–12 (2022).
Andrew, M. J. et al. Neurodevelopmental outcome of nutritional intervention in newborn infants at risk of neurodevelopmental impairment: the Dolphin neonatal double-blind randomized controlled trial. Dev. Med. Child Neurol. 60, 897–905 (2018).
Andrew, M. J. et al. Nutritional intervention and neurodevelopmental outcome in infants with suspected cerebral palsy: the Dolphin infant double-blind randomized controlled trial. Dev. Med. Child Neurol. 60, 906–913 (2018).
Hortensius, L. M. et al. NutriBrain: protocol for a randomised, double-blind, controlled trial to evaluate the effects of a nutritional product on brain integrity in preterm infants. BMC Pediatr. 21, 1–10 (2021).
He, Y., Zhang, Y., Li, F. & Shi, Y. White matter injury in preterm infants: pathogenesis and potential therapy from the aspect of the gut–brain axis. Front. Neurosci. 16, 849372 (2022).
Kramer, M. S. et al. Breastfeeding and child cognitive development: new evidence from a large randomized. Trial 65, 578–584 (2008).
Sullivan, G. et al. Breast milk exposure is associated with cortical maturation in preterm infants Gemma. Ann. Neurol. 93, 591–603. https://doi.org/10.1002/ana.26559 (2023).
Tottman, A. C., Oliver, C. J., Alsweiler, J. M. & Cormack, B. E. Do preterm girls need different nutrition to preterm boys? Sex-specific nutrition for the preterm infant. Pediatr. Res. 89, 313–317 (2021).
Reiss, A. L. et al. Sex differences in cerebral volumes of 8-year-olds born preterm. J. Pediatr. 145, 242–249 (2004).
Schneider, N. & Garcia-Rodenas, C. L. Early nutritional interventions for brain and cognitive development in preterm infants: a review of the literature. Nutrients 9, 187 (2017).
Munn, Z. et al. Systematic review or scoping review? Guidance for authors when choosing between a systematic or scoping review approach. BMC Med. Res. Methodol. 18, 1–7 (2018).
Hopewell, S., Loudon, K., Clarke, M. J., Oxman, A. D. & Dickersin, K. Publication bias in clinical trials due to statistical significance or direction of trial results. Cochrane Database Syst. Rev. 2009, MR000006. https://doi.org/10.1002/14651858.MR000006.pub3 (2009).
Acknowledgements
We thank the following staff at the University Library Utrecht for their help in drafting the initial search strategy and assistance in updating the initial search: Paulien Wiersma and Dr Marie-Louise S. Goudeau.
Funding
E.J. and M.J.N.L.B. are funded by the Athena Grant “Utrecht Center for Food and Health – research program specialized nutrition,” subsidy from the Dutch Ministry of Economic Affairs, Utrecht Province and the municipality of Utrecht. E.J., N.E. and M.J.N.L.B. are also funded by a Health-Holland-TKI grant, LSHM19087.
Author information
Authors and Affiliations
Consortia
Contributions
E.J., M.F.W., P.E.v.B., J.D., R.M.v.E., and N.E.v.d.A. made substantial contributions to the conception and design, acquisition of data, or analysis and interpretation of data. E.J., P.E.v.B., J.D., R.M.v.E., L.M.H., E.W.Y.T., M.S.d.P., A.L., C.G.M.d.T., and N.E.v.d.A. contributed to drafting the article or revising it critically for important intellectual content. E.J., M.F.W., P.E.v.B., J.D., R.M.v.E., L.M.H., E.W.Y.T., M.S.d.P., A.L., C.G.M.d.T., M.J.N.L.B., N.E.v.d.A., S.J.M., G.Z., M.J.J. C.F., and S.I. contributed to final approval of the version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Janson, E., Willemsen, M.F., Van Beek, P.E. et al. The influence of nutrition on white matter development in preterm infants: a scoping review. Pediatr Res (2023). https://doi.org/10.1038/s41390-023-02622-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41390-023-02622-1