Abstract
Background
The objective of the study was to explore the potential biomarkers and risk factors in children with immunoglobulin A nephropathy (IgAN).
Methods
Untargeted metabolomics analysis was performed on children with IgAN before and after treatment. Subsequently, a retrospective study involving the past 15 years and a follow-up study were performed to verify the role of hyperuricemia in IgAN children.
Results
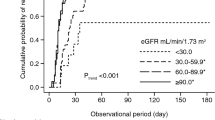
Serum metabolomics analyses showed that levels of serum xanthosine were closely related to the outcome of IgAN, and KEGG analyses showed that differential metabolites were significantly enriched in purine metabolism. Furthermore, retrospectively analyses of 252 children with IgAN showed that hyperuricemia was associated with poorer renal outcome. Logistic regression analysis showed that BMI, serum creatinine, eGFR, Lee’s grade III, and crescents were risk factors of hyperuricemia in children with IgAN. Kaplan–Meier analysis revealed that kidney progression-free survival in IgAN children with hyperuricemia was lower than that without hyperuricemia, especially in females.
Conclusions
We first performed a dynamic metabolomics study to reveal that hyperuricemia is closely related to the progression of IgAN in children. Then retrospective and follow-up studies confirmed that hyperuricemia is an important risk factor for poor renal outcomes. We need to pay more attention to the hyperuricemia in children with IgAN.
Impact
-
We first performed a dynamic metabolomics study to reveal that hyperuricemia was closely related to the progression of IgAN in children.
-
Retrospective analyses in past 15 years confirmed that IgAN children with hyperuricemia had poorer renal function and worse renal pathology.
-
The BMI, Scr, eGFR, Lee’s grade III, and crescents were risk factors of hyperuricemia in children with IgAN.
-
The long-term follow-up study showed that hyperuricemia was an important risk factor for poor renal outcome in children with IgAN.
-
We need to pay more attention to hyperuricemia in children with IgAN, especially in females.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets used and analyzed in this study are available from the corresponding author upon reasonable request.
References
Hassler, J. R. IgA nephropathy: a brief review. Semin. Diagn. Pathol. 37, 143–147 (2020).
Wyatt, R. J. & Hogg, R. J. Evidence-based assessment of treatment options for children with IgA nephropathies. Pediatr. Nephrol. 16, 156–167 (2001).
Wu, H. et al. Long-term renal survival and undetected risk factors of IgA nephropathy in Chinese children-a retrospective 1243 cases analysis from single centre experience. J. Nephrol. 33, 1263–1273 (2020).
Taherkhani, A., Farrokhi, Y. R., Mohseni, M., Saidijam, M. & Arefi, O. A. Chronic kidney disease: a review of proteomic and metabolomic approaches to membranous glomerulonephritis, focal segmental glomerulosclerosis, and IgA nephropathy biomarkers. Proteome Sci. 17, 7 (2019).
Rinschen, M. M., Ivanisevic, J., Giera, M. & Siuzdak, G. Identification of bioactive metabolites using activity metabolomics. Nat. Rev. Mol. Cell Biol. 20, 353–367 (2019).
Noone, D. G. & Marks, S. D. Hyperuricemia is associated with hypertension, obesity, and albuminuria in children with chronic kidney disease. J. Pediatr. 162, 128–132 (2013).
Mian, A. N. & Schwartz, G. J. Measurement and estimation of glomerular filtration rate in children. Adv. Chronic Kidney Dis. 24, 348–356 (2017).
Trimarchi, H. et al. Oxford Classification of IgA nephropathy 2016: an update from the IgA Nephropathy Classification Working Group. Kidney Int. 91, 1014–1021 (2017).
Xia, X. et al. Serum uric acid and mortality in chronic kidney disease: a systematic review and meta-analysis. Metabolism 65, 1326–1341 (2016).
Ponticelli, C., Podestà, M. A. & Moroni, G. Hyperuricemia as a trigger of immune response in hypertension and chronic kidney disease. Kidney Int. 98, 1149–1159 (2020).
Moriyama, T. et al. High uric acid level is a risk factor for progression of IgA nephropathy with chronic kidney disease stage G3a. J. Nephrol. 28, 451–456 (2015).
Shu, D. et al. Risk factors of progressive IgA nephropathy which progress to end stage renal disease within ten years: a case-control study. BMC Nephrol. 18, 11 (2017).
Molfino, T. et al. Hyperuricemia and obesity in North American and European Children with IgA nephropathy. Int. J. Nephrol. Kidney Fail. https://doi.org/10.16966/2380-5498.126 (2016).
Chen, T. et al. Prediction and risk stratification of kidney outcomes in IgA nephropathy. Am. J. Kidney Dis. 74, 300–309 (2019).
Alexander, S. et al. Epidemiology, baseline characteristics and risk of progression in the first South-Asian prospective longitudinal observational IgA nephropathy cohort. Kidney Int. Rep. 6, 414–428 (2021).
Russo, E. et al. Long-term blood pressure behavior and progression to end-stage renal disease in patients with immunoglobulin A nephropathy: a single-center observational study in Italy. J. Hypertens. 38, 925–935 (2020).
Choi, W. J. et al. The serum uric acid level is related to the more severe renal histopathology of female IgA nephropathy patients. J. Clin. Med. 10, 1885 (2021).
Myers, G. L. et al. Recommendations for improving serum creatinine measurement: a report from the Laboratory Working Group of the National Kidney Disease Education Program. Clin. Chem. 52, 5–18 (2006).
Chertow, G. M., Burdick, E., Honour, M., Bonventre, J. V. & Bates, D. W. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J. Am. Soc. Nephrol. 16, 3365–3370 (2005).
Uwaezuoke, S. N., Ayuk, A. C., Muoneke, V. U. & Mbanefo, N. R. Chronic kidney disease in children: using novel biomarkers as predictors of disease. Saudi J. Kidney Dis. Transpl. 29, 775–784 (2018).
Zhu, B. et al. Uric acid as a predictor of immunoglobulin A nephropathy progression: a cohort study of 1965 cases. Am. J. Nephrol. 48, 127–136 (2018).
Liu, R. et al. Prevalence of hyperuricemia and gout in Mainland China from 2000 to 2014: a systematic review and meta-analysis. Biomed. Res. Int. 2015, 762820 (2015).
Jutabha, P. et al. Apical voltage-driven urate efflux transporter NPT4 in renal proximal tubule. Nucleosides Nucleotides Nucleic Acids 30, 1302–1311 (2011).
Xu, Y. et al. Association between dyslipidaemia and the risk of hyperuricaemia: a six-year longitudinal cohort study of elderly individuals in China. Ann. Med. 54, 2402–2410 (2022).
Sun, J. et al. Mediation effect of obesity on the association between triglyceride-glucose index and hyperuricemia in Chinese hypertension adults. J. Clin. Hypertens. 24, 47–57 (2022).
Zha, D., Wu, S., Gao, P. & Wu, X. Telmisartan attenuates uric acid-induced epithelial-mesenchymal transition in renal tubular cells. Biomed. Res. Int. 2019, 3851718 (2019).
Kovács, T. et al. Metabolic syndrome and other cardiovascular risk factors associated with the progression of IgA nephropathy. Clin. Kidney J. 6, 395–401 (2013).
Kataoka, H. et al. Overweight and obesity accelerate the progression of IgA nephropathy: prognostic utility of a combination of BMI and histopathological parameters. Clin. Exp. Nephrol. 16, 706–712 (2012).
Wu, C., Wang, A. Y., Li, G. & Wang, L. Association of high body mass index with development of interstitial fibrosis in patients with IgA nephropathy. BMC Nephrol. 19, 381 (2018).
Lu, P. et al. Serum uric acid level is correlated with the clinical, pathological progression and prognosis of IgA nephropathy: an observational retrospective pilot-study. Peer J. 8, e10130 (2020).
Zhou, J. et al. Plasma uric acid level indicates tubular interstitial leisions at early stage of IgA nephropathy. BMC Nephrol. 15, 11 (2014).
Haas, M. et al. A multicenter study of the predictive value of crescents in IgA nephropathy. J. Am. Soc. Nephrol. 28, 691–701 (2017).
Funding
This work was supported by the Sun Yat-sen University Basic Scientific Research Young Teacher Training Project (grant number: 19ykpy65); Guangdong Basic and Applied Basic Research Foundation (grant number: 2019A1515010694); Science and Technology Planning Project of Guangzhou, China (grant number: 202201011400); College Student Innovation and Entrepreneurship Program of Sun Yat-sen University (grant number: 202210651).
Author information
Authors and Affiliations
Contributions
Y.X., Y.M., X.J., and W.L. contributed to the study design. Y.X., H.Z., and H.L. contributed to the serum sample and data collections. Y.X., X.Z., and H.Z. contributed to the data analysis and writing of the manuscript. Y.M., X.J., and W.L. contributed to the project supervision and review and editing of the manuscript, and all authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by the ethics committee of the First Affiliated Hospital of Sun Yat-sen University (No. [2016] 040 and [2020] 499). All the children and their guardians were informed and signed consent forms before the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, Y., Zheng, X., Zhuang, H. et al. Hyperuricemia is associated with the progression of IgA nephropathy in children. Pediatr Res 94, 1057–1066 (2023). https://doi.org/10.1038/s41390-023-02538-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02538-w