Abstract
Background
Preterm birth predisposes infants to adverse outcomes that, without early intervention, impacts their long-term health. To assist bedside monitoring, we developed a tool to track the autonomic maturation of the preterm by assessing heart rate variability (HRV) changes during intensive care.
Methods
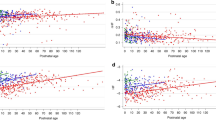
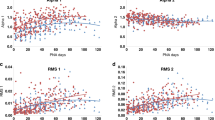
Electrocardiogram (ECG) recordings were longitudinally recorded in 67 infants (26–38 weeks postmenstrual age (PMA)). Supervised machine learning was used to generate a functional autonomic age (FAA), by combining 50 computed HRV features from successive 5-minute ECG epochs (median of 23 epochs per infant). Performance of the FAA was assessed by correlation to PMA, clinical outcomes and the infant’s functional brain age (FBA), an index of maturation derived from the electroencephalogram.
Results
The FAA was strongly correlated to PMA (r = 0.86, 95% CI: 0.83–0.93) with a mean absolute error (MAE) of 1.66 weeks and also accurately estimated FBA (MAE = 1.58 weeks, n = 54 infants). The relationship between PMA and FAA was not confounded by neurodevelopmental outcome (p = 0.18, n = 45), sex (p = 0.88, n = 56), patent ductus arteriosus (p = 0.08, n = 56), IVH (p = 0.63, n = 56) or body weight at birth (p = 0.95, n = 56).
Conclusions
The FAA, an index derived from the ubiquitous ECG signal, offers direct avenues towards estimating autonomic maturation at the bedside during intensive care monitoring.
Impact
-
The development of a tool to track functional autonomic age in preterm infants based on heart rate variability features in the electrocardiogram provides a rapid and specialized view of autonomic maturation at the bedside.
-
Functional autonomic age is linked closely to postmenstrual age and central nervous system function response, as determined by its relationship to functional brain age from the electroencephalogram.
-
Tracking functional autonomic age during neonatal intensive care unit monitoring offers a unique insight into cardiovascular health in infants born extremely preterm and their maturational trajectories to term age.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
Code for processing an ECG epoch to estimate FAA, along with the trained data model, can be found here: https://github.com/brain-modelling-group/Estimate-FAA.
References
Patel, R. M. Short-and long-term outcomes for extremely preterm infants. Am. J. Perinatol. 33, 318–328 (2016).
Blencowe, H. et al. Born too soon: the global epidemiology of 15 million preterm births. Reprod. Health 10, 1–14 (2013).
Allen, M. C., Cristofalo, E. A. & Kim, C. Outcomes of preterm infants: morbidity replaces mortality. Clin. Perinatol. 38, 441–454 (2011).
Symington, A. J. & Pinelli, J. Developmental care for promoting development and preventing morbidity in preterm infants. Cochrane Database Syst. Rev. 2006, CD001814 (2006).
Bonifacio, S. L., Glass, H. C., Peloquin, S. & Ferriero, D. M. A new neurological focus in neonatal intensive care. Nat. Rev. Neurol. 7, 485–494 (2011).
Bonifacio, S. L. & Van Meurs, K. Neonatal neurocritical care: providing brain-focused care for all at risk neonates. Semin. Pediatr. Neurol. 32, 100774 (2019).
World Health Organization. Who Child Growth Standards: Growth Velocity Based on Weight, Length and Head Circumference: Methods and Development (World Health Organization, 2009).
Cole, T. J. The development of growth references and growth charts. Ann. Hum. Biol. 39, 382–394 (2012).
Jones, M. J., Goodman, S. J. & Kobor, M. S. DNA methylation and healthy human aging. Aging Cell 14, 924–932 (2015).
Stevenson, N. J. et al. Automated cot‐side tracking of functional brain age in preterm infants. Ann. Clin. Transl. Neurol. 7, 891–902 (2020).
Dubois, J. et al. Mapping the early cortical folding process in the preterm newborn brain. Cereb. Cortex 18, 1444–1454 (2008).
Smyser, C. D. et al. Prediction of brain maturity in infants using machine-learning algorithms. Neuroimage 136, 1–9 (2016).
Barlow, J., Herath, N. I., Torrance, C. B., Bennett, C. & Wei, Y. The Neonatal Behavioral Assessment Scale (NBAS) and Newborn Behavioral Observations (NBO) system for supporting caregivers and improving outcomes in caregivers and their infants. Cochrane Database Syst. Rev. 3, CD011754 (2018).
Novak, I. et al. Early, accurate diagnosis and early intervention in cerebral palsy: advances in diagnosis and treatment. JAMA Pediatr. 171, 897–907 (2017).
Einspieler, C., Bos, A. F., Libertus, M. E. & Marschik, P. B. The general movement assessment helps us to identify preterm infants at risk for cognitive dysfunction. Front. Psychol. 7, 406 (2016).
Craciunoiu, O. & Holsti, L. A systematic review of the predictive validity of neurobehavioral assessments during the preterm period. Phys. Occup. Ther. Pediatr. 37, 292–307 (2017).
Lonsdale, H., Jalali, A., Ahumada, L. & Matava, C. Machine learning and artificial intelligence in pediatric research: current state, future prospects, and examples in perioperative and critical care. J. Pediatr. 221, S3–S10 (2020).
Stevenson, N. et al. Functional maturation in preterm infants measured by serial recording of cortical activity. Sci. Rep. 7, 1–7 (2017).
Stevenson, N. J. et al. Reliability and accuracy of EEG interpretation for estimating age in preterm infants. Ann. Clin. Transl. Neurol. 7, 1564–1573 (2020).
Kamath, M. V., Watanabe, M. & Upton, A. Heart Rate Variability (HRV) Signal Analysis: Clinical Applications (CRC Press, 2012).
Verma, P. K., Panerai, R. B., Rennie, J. M. & Evans, D. H. Grading of cerebral autoregulation in preterm and term neonates. Pediatr. Neurol. 23, 236–242 (2000).
Tan, C. M. J. & Lewandowski, A. J. The transitional heart: from early embryonic and fetal development to neonatal life. Fetal Diagnosis Ther. 47, 373–386 (2020).
Longin, E., Gerstner, T., Schaible, T., Lenz, T. & Koenig, S. Maturation of the autonomic nervous system: differences in heart rate variability in premature vs. term infants. J Perinat. Med. 34, 303–308 (2006).
Hoyer, D. et al. Fetal functional brain age assessed from universal developmental indices obtained from neuro-vegetative activity patterns. PLoS ONE 8, e74431 (2013).
Lavanga, M. et al. Maturation of the autonomic nervous system in premature infants: estimating development based on heart-rate variability analysis. Front. Physiol. 11, 581250 (2021).
Clairambault, J., Curzi-Dascalova, L., Kauffmann, F., Médigue, C. & Leffler, C. Heart rate variability in normal sleeping full-term and preterm neonates. Early Hum. Dev. 28, 169–183 (1992).
Massin, M. & Von Bernuth, G. Normal ranges of heart rate variability during infancy and childhood. Pediatr. Cardiol. 18, 297–302 (1997).
Rosenstock, E., Cassuto, Y. & Zmora, E. Heart rate variability in the neonate and infant: analytical methods, physiological and clinical observations. Acta Paediatr. 88, 477–482 (1999).
Nakamura, T., Horio, H., Miyashita, S., Chiba, Y. & Sato, S. Identification of development and autonomic nerve activity from heart rate variability in preterm infants. Biosystems 79, 117–124 (2005).
Cardoso, S., Silva, M. J. & Guimarães, H. Autonomic nervous system in newborns: a review based on heart rate variability. Childs Nerv. Syst. 33, 1053–1063 (2017).
Golder, V., Hepponstall, M., Yiallourou, S. R., Odoi, A. & Horne, R. S. autonomic cardiovascular control in hypotensive critically ill preterm infants is impaired during the first days of life. Early Hum. Dev. 89, 419–423 (2013).
Stevenson, N. J. et al. Automated cot‐side tracking of functional brain age in preterm infants. Ann. Clin. Transl. Neurol. 7, 891–902 (2020).
Hoshi, R. A., Pastre, C. M., Vanderlei, L. C. M. & Godoy, M. F. Poincaré plot indexes of heart rate variability: relationships with other nonlinear variables. Auton. Neurosci. 177, 271–274 (2013).
Doyle, O. et al. Heart rate variability during sleep in healthy term newborns in the early postnatal period. Physiol. Meas. 30, 847 (2009).
Le, T. T. et al. A nonlinear simulation framework supports adjusting for age when analyzing brainage. Front. Aging Neurosci. 10, 317 (2018).
Papile, L.-A., Burstein, J., Burstein, R. & Koffler, H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J. Pediatr. 92, 529–534 (1978).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B (Methodol.) 57, 289–300 (1995).
Silvani, A., Calandra-Buonaura, G., Dampney, R. A. L. & Cortelli, P. Brain–heart interactions: physiology and clinical implications. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 374, 20150181 (2016).
Shaffer, F. & Ginsberg, J. P. An overview of heart rate variability metrics and norms. Front. Public Health 5, 258 (2017).
Watanabe, K., Hayakawa, F. & Okumura, A. Neonatal EEG: a powerful tool in the assessment of brain damage in preterm infants. Brain Dev. 21, 361–372 (1999).
Javorka, K. et al. Determinants of heart rate in newborns. Acta Med. Martiniana https://doi.org/10.2478/v10201-011-0012-x (2011).
Ulanovsky, I., Haleluya, N., Blazer, S. & Weissman, A. The effects of caffeine on heart rate variability in newborns with apnea of prematurity. J. Perinatol. 34, 620–623 (2014).
Goudjil, S. et al. Patent ductus arteriosus in preterm infants is associated with cardiac autonomic alteration and predominant parasympathetic stimulation. Early Hum. Dev. 89, 631–634 (2013).
Patural, H. et al. Autonomic maturation from birth to 2 years: normative values. Heliyon 5, e01300 (2019).
Schlatterer, S. D. et al. Autonomic development in preterm infants is associated with morbidity of prematurity. Pediatr. Res. 91, 171–177 (2022).
Jary, S., Whitelaw, A., Walløe, L. & Thoresen, M. Comparison of Bayley‐2 and Bayley‐3 scores at 18 months in term infants following neonatal encephalopathy and therapeutic hypothermia. Dev. Med. Child Neurol. 55, 1053–1059 (2013).
Funding
This study was supported by an NHMRC Grant no. 2002135.
Author information
Authors and Affiliations
Contributions
K.K.I., N.J.S., V.G., S.V., and K.K.-S. conceptualized the design of the study and overall motivations of the study. V.G., U.L., and K.K.-S. assisted with data collection, curation, and resources necessary for investigation. K.K.I. and N.J.S. processed, analyzed, and interpreted the data. S.V., K.K.-S., and V.G. further assisted with the clinical interpretations. K.K.I., N.J.S., and S.V. wrote and edited the draft of the manuscript. K.K.-S., J.A.R., V.G., and U.L. contributed substantially to the manuscript and provided critical revisions.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Informed parental consent was obtained for all infants included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Iyer, K.K., Leitner, U., Giordano, V. et al. Bedside tracking of functional autonomic age in preterm infants. Pediatr Res 94, 206–212 (2023). https://doi.org/10.1038/s41390-022-02376-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-022-02376-2