Abstract
Background
We implemented sophisticated color M-mode analysis to assess age-dependent progression of left ventricular (LV) diastolic function.
Methods
Normal infants were prospectively enrolled for serial echocardiograms at 1 week, 1 month, 6 months, 1 year, and 2 years. From color M-mode scans, propagation velocity (VP), strength of filling (VS), and intraventricular pressure difference (IVPD) in 3 segments along apex-to-mitral valve scan line were measured.
Results
Age-wise comparisons of diastolic filling from 121 echocardiograms in 31 infants showed VP (cm/s), VS (cm2/s), and E-wave IVPD (mmHg) at 1 week to be 66.2 ± 11.9, 75.3 ± 19.9, and 1.5 ± 0.4, respectively, while VP, VS, and E-wave IVPD at 1 month were 80.3 ± 14.4, 101.2 ± 28.3, and 2.42 ± 1.1, respectively. There were significant differences in VP and segmental IVPD between first week and first month (p < 0.005) and IVPD between the age groups (p < 0.001).
Conclusions
Comprehensive analysis of transmitral color M-mode data is feasible in infants, enabling calculation of pressure drop between the LV base and apex and strength of propagation from two distinct slopes. Profound changes very early followed by relatively constant filling mechanics in later infancy indicate significant LV maturation occurring during the first month of life.
Impact
-
We implemented sophisticated analytic methods for color M-mode echocardiography in infants to assess age- and dimension-dependent changes in left ventricular diastolic function.
-
Comprehensive characterization of transmitral color M-mode flow was feasible, enabling calculation of pressure drop between left ventricular base and apex and strength of propagation.
-
Left ventricular diastolic filling function has predictable maturational progression, with significant differences in the intraventricular pressure between infants from birth to 2 years.
-
This study forms the basis for future studies to examine alteration of early diastolic filling in congenital heart disease.
Similar content being viewed by others
Introduction
Quantitative evaluation of left ventricular (LV) diastolic performance carries important prognostic implications in many conditions in which myocardial function is depressed.1,2,3,4 Advances in echocardiography and image analysis algorithms have enabled development of new modalities and tools for diastolic functional assessment.5,6,7,8 Color M-mode (CMM) echocardiography is a pulsed-wave Doppler technique that provides a spatiotemporal view of intraventricular blood velocity shifts. Postprocessing algorithms enable users to analyze properties of ventricular diastology from CMM images. These algorithms have proven valid and reliable against invasive micromanometer methodology in studies of adult human and porcine subjects.9,10,11 This work has allowed normal values for propagation velocities (VPs) and intraventricular pressure differences (IVPDs) to be established for adults. Although several groups have studied diastolic dysfunction in fetal life and in children,12,13,14 diastology data are sparse in normal, healthy infants. Evaluation of infants with diastolic dysfunction therefore lacks the context that an extensive experience with normal infants could provide. CMM could provide insight into developmental changes to be expected in the diastolic behavior of the infant LV.
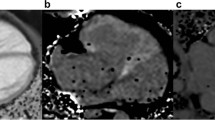
While VP was introduced as a potential single measurement for assessing diastolic function using CMM, it is not broadly utilized clinically. Current guidelines acknowledge the potential utility of VP, but it is not yet included in the routine assessment of LV diastolic dysfunction in pediatric or adult populations.15,16 Our software improves the methodology by separating each beat for automated filling function analysis and pressure reconstruction, with the objective of decreasing interobserver variability with increased automation (Fig. 1). The purpose of the present study was to implement this sophisticated CMM analysis for the assessment of age- and dimensional-dependent changes in LV diastolic function in normal infants. Accordingly, CMM assessment of diastolic function was undertaken serially in healthy children from infancy to early childhood for correlation with age and specified LV dimensions.
a A CMM scan is loaded into the in-house software where several beats along with the EKG signal, from T-wave to T-wave, are selected; b the software separates each beat and creates an individual set-up file. For a single beat (b; outlined in red), the signal undergoes c automated filling function analysis and d pressure reconstruction. The filling analysis computes c-1 the conventional propagation velocity (VP) and c-2 filling strength (VS) based on ensemble contour analysis. Pressure reconstruction computes d-1 the spatial gradient pressure field based on Euler’s equation, d-2 integrates with respect to time to reconstruction pressure, and returns d-3 the intraventricular pressure difference (IVPD) between the mitral valve and apex, where the inertial (red) curve corresponds to \(\mathop {\int }\nolimits_{x_{{\rm{MV}}}}^{x_{{\rm{apex}}}} - \rho \frac{{\partial U(x, \, t)}}{{\partial t}}{\rm{d}}x\), the convective (green) curve corresponds to \(\mathop {\int }\nolimits_{x_{{\rm{MV}}}}^{x_{{\rm{apex}}}} - \rho \frac{{\partial U(x, \, t)}}{{\partial x}}{\rm{d}}x\), and the total pressure difference (blue) curve corresponds to their sum. \(x_{{\rm{apex}}}\) and xMV are the positions of the apex and mitral valve, respectively.
Methods
Study population
This was a prospective observational study of healthy infants conducted at the University of Nebraska Medical Center in Omaha, NE. A total of 121 unsedated examinations were performed on 31 infants. All infants included in this study were normal and term-born without complications.
All subjects were serially followed through 2 years of age, and images were collected at five specific ages. Twenty-five (25/31) subjects were scanned at 1 week, 25/31 at 1 month, 29/31 at 6 months, 31/31 at 1 year, and 11/31 at 2 years of age. All 11 subjects assessed at 2 years were assessed at all previous time points, and each subject in the cohort underwent at least three evaluations. Figure 2 summarizes the specifics of the cohort. Different numbers of subjects in different time periods reflect changes in recruitment and loss to follow-up. Age, weight, body surface area (BSA), and heart rate were recorded for each examination. Exclusion criteria consisted of: prematurity (<37 weeks gestational age (GA) at birth), intrauterine growth restriction, perinatal acidosis, suspected chromosomal anomalies, neuro-muscular disorders, cardiac arrhythmias, patent ductus arteriosus, and heart failure. Infants readmitted to the hospital within the first 2 years of age were also excluded. Maternal exclusion criteria included: multiple gestation pregnancy, maternal diabetes, maternal self-reported use of illegal drugs, maternal smoking, antepartum hemorrhage, pre-eclampsia, and/or proven chorioamnionitis. The Institutional Review Board for Human Studies approved the study. Written informed consents were obtained from the parents or guardians of the study subjects.
Echocardiography
Echocardiograms were performed on each infant by using commercially available ultrasound equipment (Vivid E9; General Electric Medical Systems, Milwaukee, WI). One designated trained cardiac sonographer obtained all the echocardiographic images using a GE phased-array-transducer (4.5–12 MHz). The echocardiographic images were collected by using the recommendations from the American Society of Echocardiography.15 The four-chamber and parasternal long- and short-axis (at the level of the LV base) views of the heart were visualized and recorded. From the four-chamber view, CMM images were collected along the scan line between the mitral valve (MV) and apex. Images were stored in Digital Imaging and Communications in Medicine (DICOM) format for postprocessing.
CMM scans were analyzed on a computer using a MATLAB algorithm for CMM analysis. Dimensional measurements were also collected with the GE EchoPAC software from the apical four-chamber view scans. LV parasternal short axis at the LV base and parasternal long axis lengths, end-diastolic volume (EDV), stroke volume (SV), and ejection fraction were recorded.
Description of the analysis software
The automated CMM analysis algorithm, described by Stewart et al.,1 was used to analyze single-beat recordings for diastolic measurements as shown in Fig. 1. Briefly, each scan is segmented into separate beat images from R-wave to R-wave using the electrocardiogram (Fig. 1a), and each image is converted from a color pixel (RGB) value to a velocity value (Fig. 1b). Reconstructed velocity images are subsequently analyzed to compute filling function measurements such as VP, filling strength (VS), and intraventricular relative pressure.
The filling function analysis is performed in four key steps to automatically determine MV position, filling deceleration position (L) from the MV, VP, and VS. VS correlates with strength of early diastolic filling as it accounts for both VP and the deceleration point at the MV annulus. Analysis begins by segmenting the early filling (E-wave) phase from the rest of the scan automatically using image-processing methods. A series of 11 isovelocity contours between 45% and 55% of the peak E-wave transmitral velocity are determined. A smoothing spline is fit to the ensemble of these contours to produce a representative isovelocity contour.
The leading edge of the ensemble isovelocity contour is cropped from its earliest time point to the time when the furthest position of the isovelocity contour has penetrated into the LV. Two slopes are fit along the edge, one from the earliest time point and along the edge into the LV and the other from the earliest time point and along the edge into the LA. The intersection of these two lines provides an automated measurement of the MV position.
The edge of the ensemble isovelocity contour from the updated MV position to the furthest position into the LV is evaluated using statistical changepoint analysis.17,18 The identified changepoint corresponds to the abrupt deceleration during filling and corresponds to filling wave-penetration depth or filling deceleration position L. This edge is then segmented at L to allow for two additional line fits to determine modified VP values. The early VP (VP,Early) corresponds to a portion of the curve from the MV to L. VP,Early and deceleration position are used to determine VS, computed as follows:
An example of the VP,Early, L, and VS, as they are obtained from the CMM scan, is provided in Fig. 1c-2. The final step of the filling function analysis pulls the 50% isovelocity contour for the segmentation series. The leading edge of the 50% contour, from the MV to either a 4-cm distance or the furthest position into the LV, is fit with a line to provide the VP (Fig. 1c-1). This measurement is based on the conventional definition by Brun et al.19
Reconstructed relative pressure was determined from each beat velocity field using the one-dimensional incompressible Euler equation. This equation relates the CMM velocities U to pressure values P along the scan line for given spatiotemporal dimensions x and t and density ρ.
In order to determine pressure P using Eq. 2, two steps are performed. First, spatial and temporal derivatives of the velocity U are computed using a noise-optimized differentiation function,20 then summed to produce the pressure gradient field. Second, the gradient field is integrated at each time step along the spatial direction to yield the relative pressure field. In order to compute the IVPD, the pressure at the MV position is subtracted from the pressure at the apex position. This approach is similar to calculations performed in literature5 and has been validated by direct comparison to pressure catheter readings.5,7,21,22 An example output IVPD is provided in Fig. 1d-3, where the total IVPD, along with the inertial and convective terms, are included. The inertial and convective terms are the integrated, MV-to-apex difference results for each term on the right-hand side of Eq. 1. The inertial term (Fig. 1d, red line) is the integrated temporal derivative of U and the convective term (Fig. 1d, green line) is the integrated spatial derivative of U. These terms, summed together, make up the total IVPD (Fig. 1d, blue line).
Reproducibility
We randomly selected 15 examinations from patients at the 1-year time point for assessing the reproducibility of the algorithm. The primary observer (C.T.E.) repeated the analysis after a 3-month interval for examination of intraobserver agreement. A blinded secondary observer (L.L.) performed analysis for examining interobserver agreement. Bland–Altman plots were generated to display agreements.
Statistical analysis
Continuous variables were described with means and standard deviations; categorical variables as percentages. Data were organized into five groups by age of the subject at examination. Normality was assessed using Shapiro–Wilk test. Differences in diastolic function parameters among the five groups were sought using one-way analysis of variance or Kruskal–Wallis W test, when appropriate. If differences were suggested, pairwise comparisons were made using Bonferroni correction for multiple comparisons. When it was observed that changes in diastolic function parameters early in infancy were rapid relative to the more gradual changes later in infancy, linear modeling of these parameters versus age, weight, BSA, and heart rate was abandoned in favor of modeling based on the natural logarithmic transformation of the independent variables. Correlations of diastolic function parameters with log-transformed age, weight, BSA, and heart rate were calculated and compared. Association of diastolic function parameters with logarithmically transformed age, weight, height, BSA, and LV dimensions and volumes were assessed by calculating the Pearson product moment correlation coefficient and testing the null hypothesis that this coefficient = 0. Multivariate regression analysis was performed to address for collinearity of heart rate and age when examining correlations between CMM echocardiographic parameters and LV volumes. Furthermore, reproducibility analyses, namely, intraobserver and interobserver reproducibility, was assessed utilizing the intraclass correlation coefficient (ICC). Linear regression of dependent variables (diastolic function parameters) versus independent variable (natural logarithm of subject’s age in days) produced graphs of age-related expected values of these parameters and their 95% confidence intervals. The standard of statistical significance is p < 0.05 for all analyses.
Results
Demographic features of the study subjects are shown in Table 1. The LV short- and long-axis diastolic dimensions, EDV, and SV increased among the subjects from age 1 week to 2 years (Table 2). This closely matches with expectations for normal growth and development of a child.23,24 All the diastolic function parameters also varied depending on the age group of the subject (Figs. 3 and 4, Table 2). Pairwise comparisons of the values of diastolic function parameters among the age categories revealed that all had significant (ps corrected for multiple comparisons) changes between 1 week and 1 month in VP (p = 0.03), VS (p = 0.04), breakpoint (p < 0.01), E-wave IVPD (p = 0.02), and A-wave IVPD (p = 0.01). Of all these parameters, however, only E-wave IVPD had demonstrable change between 1 month and 6 months (p = 0.03). Pairwise comparisons among the age categories did not reveal statistically significant change in any of the parameters beyond 6 months of age (p > 0.05).
The highly nonlinear relations between age and diastolic function parameters with major changes early in infancy which diminished rapidly suggested that nonlinear (logarithmic) modeling would provide better description of these changes than linear modeling. Table 3 shows correlation of the diastolic function parameters with independent variables (age, weight, BSA, and heart rate after logarithmic transformation). Although significant correlations of diastolic filling parameters with age, weight, BSA, and heart rate were discovered (Table 3), the strongest correlations were with age. Comparison of diastolic filling parameters with left diastolic ventricular dimensions and volumes, whether log transformed or not, did not yield consistently stronger correlations than logarithm of age (Table 4).
Multiple linear regression was utilized to account for collinearity between diastolic parameters, age, and heart rate. These results are summarized in Supplementary Table 1. Breakpoint was not found to be correlated with heart rate even with multivariate regression analysis and was only found to be significantly correlated with EDV.
Reproducibility results
Figure 5 represents Bland–Altman plots of agreement between two separate measurements done by the first observer 3 months apart (intraobserver) and between the measurements made by two observers (interobserver). The Bland–Altman method as well as ICCs with 95% confidence intervals showed good agreement for both intraobserver and interobserver comparisons (Table 5). Briefly, ICC for intraobserved and interobserved variability was >0.87 for all parameters, indicating reasonable reproducibility compared to similar methods.
Discussion
Principle findings
This study utilized CMM with more sophisticated analytic techniques to evaluate diastolic function longitudinally in normal infant subjects. While recent works have performed CMM for fetal arrhythmia evaluation and LV diastolic function in adults,1,25 few studies exist assessing pediatric LV diastolic function utilizing CMM. The investigation confirmed previous findings that there are temporal changes in diastolic function in infants. There were several observations of interest: (1) diastolic function in the young ventricle seems to change more in the first month of life than later in infancy, as previously reported;26,27 (2) there are strong correlations between LV dimensions and the IVPDs in each segment; and (3) CMM is a feasible and reproducible method for evaluation of intraventricular diastolic flow patterns in infants.
Color M-mode
CMM allows for acquisition of spatiotemporal velocity fields across the left ventricle and assessment of both systolic and diastole ventricular hemodynamics. Furthermore, utilizing simple fluid dynamics and assumptions of Bernoulli flow, pressure gradients can be estimated. Traditional assessment of diastolic function involves assessment of E-wave–A-wave ratio, which is limited. Furthermore, compared to conventional transmitral Doppler parameters and other mitral annular tissue Doppler velocities, CMM allows for direct assessment of LV relaxation and diastolic function and allows for spatial and temporal velocity fields. Sophisticated analytic methods, as those presented here and previously in the literature,5,7,21,22 allow for assessment of diastolic hemodynamics even in situations where separation of E-wave and A-wave is not possible. Furthermore, clinical assessment of VP involves measurement of the initial linear slope of the isovelocity contour. However, this fails to take into consideration patients with normal filling, but deceleration points further from the annulus, likely due to different ventricular localization of dysfunction. Thus our methods, while resulting in different VPs from clinically utilized methods, allow for a more inclusive definition for diastolic dysfunction. Utilization of CMM for assessment of LV diastolic function utilizing sophisticated computational methods may allow for determination of spatiotemporal maps of diastolic function. While theoretically VP, is entirely preload dependent, the interaction between VP, filling pressures and ventricular compliance remains unclear.28,29 Future studies should aim to clarify this relationship.
Diastolic physiology
After ejection of blood from the LV and the closure of the aortic valve, the ventricle starts the phase of isovolumetric ventricular relaxation that results in chamber expansion. Owing to the inverse pressure–volume relationship, volumetric expansion will decrease pressure within the chamber, generating suction at the MV.21,30 The strength of the suction promotes both MV opening and prograde blood flow into the LV. This mass flow across the valve is manifested in both the VP and VS, which measure the rate at which mass penetrates into the LV. Their magnitudes are dependent on the degree of myocardial relaxation.1 Our results further demonstrated that breakpoint was found to be constant during the first month of life despite further increases in LV length throughout the remaining 2 years assessed in this study. This reflects the observation that diastolic function seems to change most in the first month of life.
Age-related myocardial transition
Diastolic function in the very young should be understood not only on the basis of the mechanical aspects of filling (loading and compliance) but also in terms of the underlying biological adaptations that may be responsible for normal changes in LV filling characteristics. The young myocardium has not developed its full compliance and functional potential, so it will not provide as much diastolic suction as would a fully developed myocardium. As the myocardium matures and remodels, it develops a higher content in its extracellular matrix, and the ratio of collagen type I (rigidity) to type III (elasticity) decreases, allowing for a more compliant muscle.31,32,33 Thus the myofibers in the septal and lateral wall are making internal adaptations for the applied forces of the ventricle’s new postnatal circulatory environment. This insight into the mechanical nature of diastolic function provides a basis for understanding the developmental changes in flow characteristics across the MV and deeper into the chamber. From our data, we observed that both the VP and VS increase significantly between the first week and first month of life. We believe that these changes are indicative that immediately after birth the ventricle needs to adapt to the postnatal circulatory conditions.34,35 Other groups have shown that fetal VP is lower than those of adults, most likely due to the continual development and structural programming of the myocardium.13,36,37 As the infantile myocardium transitions out of fetal circulation and into postnatal, independent circulation, loading conditions will change and so will the mechanical compliance of the myocardium.31,38,39
Greater myocardial compliance produces greater diastolic ventricular suction strength resulting in a higher VP, and thus higher VS. While theoretically appealing, and initial reports of its use for detection of diastolic dysfunction have been promising,1 more validation of VS is required before adopting it into standard clinical practice.
As the suction within the ventricle expands in the initial stages of diastole to pull open the MV and draw the blood apically, the data reported here demonstrate that the apical region of the chamber reaches the highest pressure along the scan line. We speculate the high pressure generated to force the blood out of the apical portion of the ventricle and into the outflow tract persists transiently in the apex after the closure of the aortic valve. Greater changes in pressure were, therefore, observed at the apex than at the base during the transition from systole to diastole.
As expected in a growing infant, the long- and short-axis LV dimensions increase with age, and this also allows for higher pressure gradient in the ventricle. It is important to note, however, that the methodology in our study does not assess absolute pressures but rather pressure differences. Nonetheless, it is not surprising then to find that each of the E-wave IVPDs also strongly and positively correlated with age. In a larger infant, previous studies have demonstrated that a wider MV orifice will be less resistant to inflow, and we hypothesize pressure will be lower at the MV during the passive filling process.26 These results suggest that the longer ventricle allows a higher pressure difference between the apical and basal regions of the chamber. The shape of the chamber differs over the length of the long axis, where along that axis the cross-sectional area changes from apex to base, in a fashion like that of a prolate spheroid.40 If the shape of the chamber changes, there could be variation in the spatial pressure distributions around the chamber during diastole.40,41 Consequently, we hypothesize that pressure at the MV will be decreased during passive filling, resulting in a pressure gradient between the left atrium and ventricle. In older infants, the decrease in MV pressure during passive filling is suggestive of a relatively increased MV orifice area. Fetal and early postnatal LV loading conditions drastically change in the early part of life due to decreasing pulmonary vascular resistance.42,43 While diastolic function from CMM was found to change most during the first month of life, it is unknown whether those were due to changing hemodynamic loading conditions or maturation of fetal cardiomyocytes.
Our data demonstrate that there are significant changes in CMM parameters between 1 week and 1 month of life, indicating rapid development during this time period. Though the infant experiences a significant change in ventricular dimensions in the first month that correlate with the changes in diastolic functional parameters, it is also likely that, in addition to changes in dimensions, biological tissue composition and the transition to postnatal circulation contribute to the alterations in the circulation.
Clinical implications
Age-based normal values for echocardiographic indices of diastolic function are needed for comparative interpretation and assessment of diastolic functional abnormalities due to congenital defects.14,44 In adults, it has been shown that in conditions that limit diastolic function, such as cardiomyopathies and heart failure, the degree of function based on VP and IVPDs in the ventricles are reduced.19 Establishing normal ranges and correlations of function in the infant will aid in future diagnosis and prognosis. Because of the observed reproducibility of this data as assessed by the ICC, there is potential to clinically use this application for the assessment of diastolic function in the young. The program demonstrates efficiency and allows the user to operate the interface at ease, which would allow for the future implementation of the program into the patient care setting. While the small sample size presented prevents determination of age-based normal values of echocardiographic indices of diastolic function, we have demonstrated that the computational methods utilized allow for feasible application, and future work should validate this methodology in larger cohorts. More conventional measures of LV diastolic function were not assessed, and future studies should aim to examine CMM parameters along with traditional diastolic parameters in infants. Furthermore, future work should include pediatric pathologies in which diastolic function is impaired, including congenital heart disease, pediatric obesity, and dysglycemia.
Limitations
This study analyzed only the diastology of the LV, without assessing its relation to other important aspects of myocardial function (i.e., strain, strain rate, annular systolic excursion, etc.). Future investigations will need to address how diastolic function relates to strain rate, a well-accepted indicator of contractility,45,46 and other measures of systolic function. The indices measured in this study relate to the rate of LV relaxation and do not examine passive diastolic properties. Because neonatal circulatory transition is anticipated to have profound effect on the right ventricle, and this was unaddressed in the current investigation, we propose to study developmental changes in diastolic function in the normal right ventricle.
Another limitation of this study is the differences in cohort size for the age ranges. As a result, conclusions regarding serial changes in CMM parameters should be interpreted with caution. The variability in measurements is likely to be higher in younger patients due to higher heart rates. The noninvasive methodology of this study makes the inferences about diastolic function somewhat indirect. More invasive techniques, such as the use of micromanometers or catheterization, may give slightly more accurate values for pressure differences; however, the use of CMM for assessing ventricular pressure difference has been validated as an accurate method by several other groups.7,11,36 Images collected in echocardiography are dependent on the angle of the scan line. If there is error in the scan line, it is possible that the Euler relationship’s results between VPs and pressure differences may be skewed.9,21,47 With this data being collected only in M-mode, there is only one line of dimension that is being represented—blood that is on the edge of the flow will have reduced velocity due to frictional energy losses in its contact with the valve and myocardium.48 Error may also occur while the algorithm is in the process of dealiasing, if it misinterprets the color scale or misidentifies aliased regions. These errors may account for large variation in VP and VS within each age group. Finally, while measurements of diastolic flow velocities under resting conditions was performed in the study, perturbations that challenge diastolic function such as volume load was not examined.
Conclusion
Transitional changes in normal LV diastolic function in the healthy infant are largely complete within the first month after birth. Because of the rapid change and development of the ventricle in the first 2 years of life, we were able to show that changes in CMM parameters, namely, VP, VS, and IVDP, were significantly different between 1 week and 1 month of life but not thereafter. While these results suggest that diastolic indices correlate with dimensional aspects of the ventricle and follow distinct patterns over neonatal life and into early childhood, future work is required to assert changes in diastology in early life. Future work will expand observations to the right ventricle, explore inter-relation with indices of systolic function, and serially monitor patients with disease states that are known to cause diastolic LV dysfunction using comparisons made to the normal standards reported here.
References
Stewart, K. C. et al. Evaluation of LV diastolic function from color M-mode echocardiography. JACC Cardiovasc. Imaging 4, 37–46 (2011).
Lo Iudice, F. et al. Left ventricular diastolic dysfunction in type I Gaucher disease: an Echo Doppler study. Echocardiography 32, 890–895 (2015).
Hui, W. et al. Diastolic asynchrony and myocardial dysfunction in patients with univentricular heart after Fontan operation. J. Echocardiogr. 11, 130–137 (2013).
Agha, H. M. et al. Forgotten right ventricle in pediatric dilated cardiomyopathy. Pediatr. Cardiol. 38, 819–827 (2017).
Greenberg, N. L., Vandervoort, P. M., Firstenberg, M. S., Garcia, M. J. & Thomas, J. D. Estimation of diastolic intraventricular pressure gradients by Doppler M-mode echocardiography. Am. J. Physiol. Circ. Physiol. 280, H2507–H2515 (2001).
Thomas, J. D., Greenberg, N. L., Vandervoort, P. M., Aghassi, D. S. & Hunt, B. F. Digital analysis of transmitral color Doppler M-mode data: a potential new approach to the noninvasive assessment of diastolic function. In Proc. Computers in Cardiology 631–734 (IEEE, 1992).
Yotti, R. et al. Noninvasive assessment of ejection intraventricular pressure gradients. J. Am. Coll. Cardiol. 43, 1654–1662 (2004).
Garcia, M. J. et al. An index of early left ventricular filling that combined with pulsed Doppler peak E velocity may estimate capillary wedge pressure. J. Am. Coll. Cardiol. 29, 448–454 (1997).
Yotti, R. et al. Doppler-derived ejection intraventricular pressure gradients provide a reliable assessment of left ventricular contracility. Circulation 112, 1771–1779 (2005).
Takatsuji, H. et al. A new approach for evaluation of left ventricular diastolic function: spatial and temporal analysis of left ventricular filling flow propagation by color M-mode Doppler echocardiography. J. Am. Coll. Cardiol. 27, 365–371 (1996).
Gonzalez-Vilchez, F., Ares, M., Ayuela, J. & Alonso, L. Combined use of pulsed and color M-mode Doppler echocardiography for the estimation of pulmonary capillary wedge pressure: an empirical approach based on an analytical relation. J. Am. Coll. Cardiol. 34, 515–523 (1999).
Hashimoto, I. et al. Assessment of variables affecting flow propagation velocity of the left ventricle in healthy children. Pediatr. Int. 49, 305–309 (2007).
Moon‐Grady, A. J., Taylor, D., Bennett, S. H., Hornberger, L. K. & Tacy, T. A. Color M‐mode propagation velocity, but not its ratio to early diastolic inflow velocity, changes throughout gestation in normal human fetuses. Ultrasound Obstet. Gynecol. 31, 535–541 (2008).
Border, W. L. et al. Color M-mode and Doppler tissue evaluation of diastolic function in children: simultaneous correlation with invasive indices. J. Am. Soc. Echocardiogr. 16, 988–994 (2003).
Lopez, L. et al. Recommendations for quantification methods during the performance of a pediatric echocardiogram: a report from the Pediatric Measurements Writing Group of the American Society of Echocardiography Pediatric and Congenital Heart Disease Council. J. Am. Soc. Echocardiogr. 23, 465–495 (2010).
Nagueh, S. F. et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur. J. Echocardiogr. 17, 1321–1360 (2016).
Hinkley, D. V. Inference about the change-point from cumulative sum tests. Biometrika 58, 509–523 (1971).
Taylor, W. A. Change-point analysis: a powerful new tool for detecting changes. https://pdfs.semanticscholar.org/c723/b683b231b1917804f217d186cfbe5be25079.pdf (2000).
Brun, P. et al. Left ventricular flow propagation during early filling is related to wall relaxation: a color M-mode Doppler analysis. J. Am. Coll. Cardiol. 20, 420–432 (1992).
Etebari, A. & Vlachos, P. P. Improvements on the accuracy of derivative estimation from DPIV velocity measurements. Exp. Fluids 39, 1040–1050 (2005).
Yotti, R. et al. A noninvasive method for assessing impaired diastolic suction in patients with dilated cardiomyopathy. Circulation 112, 2921–2929 (2005).
Rovner, A. et al. Improvement in diastolic intraventricular pressure gradients in patients with HOCM after ethanol septal reduction. Am. J. Physiol. Circ. Physiol. 285, H2492–H2499 (2003).
Pearlman, J. D., Triulzi, M. O., King, M. E., Newell, J. & Weyman, A. E. Limits of normal left ventricular dimensions in growth and development: analysis of dimensions and variance in the two-dimensional echocardiograms of 268 normal healthy subjects. J. Am. Coll. Cardiol. 12, 1432–1441 (1988).
Huwez, F. U., Houston, A. B., Watson, J., McLaughlin, S. & Macfarlane, P. W. Age and body surface area related normal upper and lower limits of M mode echocardiographic measurements and left ventricular volume and mass from infancy to early adulthood. Heart 72, 276–280 (1994).
Detterich, J. A., Pruetz, J. & Sklansky, M. S. Color M-mode sonography for evaluation of fetal arrhythmias. J. Ultrasound Med. 31, 1681–1688 (2012).
Schmitz, L., Koch, H., Bein, G. & Brockmeier, K. Left ventricular diastolic function in infants, children, and adolescents. Reference values and analysis of morphologic and physiologic determinants of echocardiographic Doppler flow signals during growth and maturation. J. Am. Coll. Cardiol. 32, 1441–1448 (1998).
Kozák-Bárány, A., Jokinen, E., Saraste, M., Tuominen, J. & Välimäki, I. Development of left ventricular systolic and diastolic function in preterm infants during the first month of life: a prospective follow-up study. J. Pediatr. 139, 539–545 (2001).
Seo, Y. et al. Preload-dependent variation of the propagation velocity in patients with congestive heart failure. J. Am. Soc. Echocardiogr. 17, 432–438 (2004).
Tsujita, Y., Kato, T. & Sussman, M. A. Evaluation of left ventricular function in cardiomyopathic mice by tissue Doppler and color M‐mode Doppler echocardiography. Echocardiography 22, 245–253 (2005).
Tanaka, M. et al. Physiological basis and clinical significance of left ventricular suction studied using echo-dynamography. J. Cardiol. 58, 232–244 (2011).
Marijianowski, M. M. H., van der Loos, C. M., Mohrschladt, M. F. & Becker, A. E. The neonatal heart has a relatively high content of total collagen and type I collagen, a condition that may explain the less compliant state. J. Am. Coll. Cardiol. 23, 1204–1208 (1994).
Hutchinson, K. R., Stewart, J. A. Jr & Lucchesi, P. A. Extracellular matrix remodeling during the progression of volume overload-induced heart failure. J. Mol. Cell. Cardiol. 48, 564–569 (2010).
Leite-Moreira, A. F. Current perspectives in diastolic dysfunction and diastolic heart failure. Heart 92, 712–718 (2006).
Romero, T., Covell, J. & Friedman, W. F. A comparison of pressure-volume relations of the fetal, newborn, and adult heart. Am. J. Physiol. 222, 1285–1290 (1972).
Davis, P. J., Cladis, F. P. & Motoyama, E. K. Smith’s Anesthesia for Infants and Children (Mosby, Philadelphia, PA, 2011).
Garcia, M. J. et al. Color M-mode Doppler flow propagation velocity is a preload insensitive index of left ventricular relaxation: animal and human validation. J. Am. Coll. Cardiol. 35, 201–208 (2000).
Legato, M. J. Cellular mechanisms of normal growth in the mammalian heart. I. Qualitative and quantitative features of ventricular architecture in the dog from birth to five months of age. Circ. Res. 44, 250–262 (1979).
Friedman, W. F. The intrinsic physiologic properties of the developing heart. Prog. Cardiovasc. Dis. 15, 87–111 (1972).
Price, J. F. in Heart Failure in Congenital Heart Disease 21–42 (Springer, 2010).
Moulton, M. J., Hong, B. D. & Secomb, T. W. Simulation of left ventricular dynamics using a low-order mathematical model. Cardiovasc. Eng. Technol. 8, 480–494 (2017).
Feit, T. S. Diastolic pressure-volume relations and distribution of pressure and fiber extension across the wall of a model left ventricle. Biophys. J. 28, 143–166 (1979).
Patey, O., Gatzoulis, M. A., Thilaganathan, B. & Carvalho, J. S. Perinatal changes in fetal ventricular geometry, myocardial performance, and cardiac function in normal term pregnancies. J. Am. Soc. Echocardiogr. 30, 485–492 (2017).
Patey, O., Carvalho, J. S. & Thilaganathan, B. Perinatal changes in fetal cardiac geometry and function in diabetic pregnancy at term. Ultrasound Obstet. Gynecol. 54, 634–642 (2019).
Mottram, P. M. & Marwick, T. H. Assessment of diastolic function: what the general cardiologist needs to know. Heart 91, 681–695 (2005).
Ferferieva, V. et al. The relative value of strain and strain rate for defining intrinsic myocardial function. Am. J. Physiol. Circ. Physiol. 302, H188–H195 (2011).
Alvarez, S. V. et al. Strain rate in children and young piglets mirrors changes in contractility and demonstrates a force-frequency relationship. J. Am. Soc. Echocardiogr. 30, 797–806 (2017).
Londono-Hoyos, F. J. et al. Assessment of methodologies to calculate intraventricular pressure differences in computational models and patients. Med. Biol. Eng. Comput. 56, 469–481 (2018).
Akins, C. W., Travis, B. & Yoganathan, A. P. Energy loss for evaluating heart valve performance. J. Thorac. Cardiovasc. Surg. 136, 820–833 (2008).
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The authors acknowledge the support of the Children’s Hospital and Medical Center Foundation.
Author information
Authors and Affiliations
Contributions
C.T.E., B.M., P.V., and S.K. conceived and designed the study. C.T.E., B.M., L.L., V.J., and M.C. performed measurements. K.S.B.M., J.B., and D.A.D. performed statistical analysis. C.T.E., B.M., V.J., J.B., and S.K. wrote the manuscript. All authors revised the manuscript critically for important intellectual content, and all authors read and approved the final version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Patient consent
Written informed consents were obtained from the parents or guardians of the study subjects.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Erickson, C.T., Meyers, B., Li, L. et al. Progression of left ventricular diastolic function in the neonate and early childhood from transmitral color M-mode filling analysis. Pediatr Res 89, 987–995 (2021). https://doi.org/10.1038/s41390-020-1011-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-020-1011-6
This article is cited by
-
Heart Failure with Preserved Ejection Fraction in Children
Pediatric Cardiology (2023)