Abstract
Background
Autonomic dysfunction is a key trait in the development of obstructive sleep apnea syndrome (OSAS) in adults. However, few similar studies focused on children and adolescents. We investigated if there was any association between heart rate recovery (HRR) and the severity of OSAS in children and adolescents.
Methods
A total of 161 subjects were included: 87 healthy controls, 35 mild OSAS patients (M-OSAS) and 39 moderate-severe OSAS (M-S-OSAS) patients. Clinical parameters, cardiopulmonary exercise test (CPET) indexes including HRR and polysomnographic records including apnea−hypopnea index (AHI) were compared among the three groups. Pearson correlation analysis and multivariable linear regression analysis were used to detect the relationship between HRR and polysomnographic parameters.
Results
HRR values in either the OSAS group were lower than those of the control group, and the values in the M-S OSAS group were even lower than those of the M-OSAS group (P < 0.05). Correlation analysis showed that HRR was inversely correlated with AHI (r = −0.190, P < 0.01). Moreover, multivariable linear regression analyses showed the association between HRR and AHI was significant (β = 0.174, P < 0.01).
Conclusions
HRR was closely associated with OSAS severity, which suggested that HRR might be a promising index for risk stratification and clinical management in children and adolescents with OSAS.
Impact
-
Heart rate recovery (HRR), a noninvasive and reliable index of automatic nervous system, is closely associated with the severity of obstructive sleep apnea syndrome (OSAS) in children and adolescents.
-
To our knowledge, our study was the first study in China focusing on evaluating the role of HRR in children with OSAS.
-
We found HRR was closely associated with OSAS severity in children and adolescents, and it suggested that HRR might be a promising index for risk stratification and clinical management in children and adolescents with OSAS.
Similar content being viewed by others
Introduction
Obstructive sleep apnea syndrome (OSAS) is defined as recurrent occurrence of upper airway obstruction, characterized by hypoxia and intermittent hypercapnia. Symptoms and signs of OSAS are classified into those directly related to the intermittent pharyngeal airway obstruction (e.g., parental report of snoring, apneic events) and into morbidity resulting from the upper airway obstruction (e.g., increased daytime sleepiness, poor school performance, inadequate somatic growth rate).1 It is a common disease in children and adolescents, and epidemiological data show the prevalence is about 1.2–5.7% in them.2 Similar to adults, metabolic disorder and cardiovascular diseases, such as hyperglycemia, hypertension and cardiac dysfunction, can be found in children with OSAS.3 Autonomic dysfunction has been identified as a key trait of the above-mentioned complications in both adult and pediatric OSAS.4,5,6 Some studies began to assess the effect of adenotonsillectomy, a classic and effective therapy for OSAS, on children’s autonomic nervous system.7,8,9
Heart rate recovery (HRR) is calculated as the difference between heart rate (HR) at peak exercise and HR 1 min after exercise. It reflects immediate alterations in autonomic nervous function after the cessation of exercise.10 Meta-analysis has proved slow HRR having the prognostic value of predicting cardiovascular diseases and mortality in adults.10 Meanwhile, this noninvasive assessment has been studied in children, such as metabolically unhealthy obese children,11 children with sickle cell anemia,12 and in children following pediatric heart transplantation13 in different regions of the world, including Chile,14 India15 and China.16 The significance of HRR was confirmed in those children’s investigations; thus, we hypothesized that an evaluation of HRR in children with OSAS would be of clinical interest, and it would be helpful for exploring underlying pathways and serial stratified management. Polysomnography (PSG) measurement is the gold standard test for diagnosing OSAS, which calculates the apnea−hypopnea index (AHI), the number of apnea-plus-hypopnea events per sleeping hour. The severity of the OSAS depends on the magnitude of the AHI value: 1 ≤ AHI < 5 events/h, mild OSAS; 5 ≤ AHI ≤ 10 events/h, moderate OSAS; >10 events/h, severe OSAS.17 Thus we designed this study to observe if there is any association between HRR and the severity of OSAS or any other index of PSG.
Participants and methods
Study subjects
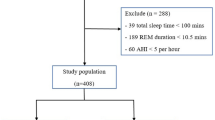
Initially, we enrolled 168 newly diagnosed OSAS patients who had not received any related treatment before. They visited the Department of Children Health Check-up Center and Otorhinolaryngology in Minhang Hospital for snoring evaluation from January 2016 to September 2019. All participants underwent an overnight PSG examination. Our exclusion criteria were as follows: children who were known to having defined genetic syndrome, congenital heart disease, systemic or pulmonary hypertension, hyperlipidemia, endocrine diseases (i.e., diabetes mellitus, thyroid disorder), neuromuscular diseases, severe orthopedic problems, craniofacial anomalies, acute illness or taking chronotropic drugs (i.e., β-blockers, amiodarone, or calcium-channel-blockers), other sympathomimetric drugs or bronchodilators were excluded. Then 74 patients were included into our research. Meanwhile, 87 healthy age-matched children from the Health Check-up Center in Shanghai Minhang Hospital were enrolled as controls.
Our study was approved by Shanghai Minhang Hospital affiliated with Fudan University research ethics committee. Written informed consents were provided by parents or children (whose age was more than 8 years old).
Clinical data collection
All children’s body mass index (BMI), systolic blood pressure (SBP), diastolic blood pressure (DBP) and HR at rest (HRrest) were recorded. BMI was calculated as the body weight divided by the square of the body height (kg/m2).
PSG assessment
Every participant underwent 7 h of sleep monitoring with PSG (Philips, Respironics, Amsterdam, Netherlands). Tea, coffee, cola or tranquilizers were not allowed before going to bed. Two experienced pediatric PSG technicians assessed the PSG data manually, according to American Academy of Sleep Medicine (AASM) Scoring Manual.18 AHI was the number of apnea-plus-hypopnea incidents per sleeping hour. Oxygen desaturation index (ODI) was the hourly incidents number of the blood’s oxygen level declining by 3% (or 4%) and more from baseline. The average oxyhemoglobin saturation (AverSpO2), the minimum oxyhemoglobin saturation (MinSpO2), arousal index, and Time/percentage SpO2 below 90% were recorded. Seventy-four OSAS participants were divided into two subgroups, 35 into mild OSAS (M-OSAS) group and 39 into moderate-severe OSAS (M-S-OSAS) group according to their AHI values.
Cardiopulmonary exercise test
Every participant conducted a cardiopulmonary exercise test (CPET) according to a symptom-limited Bruce’s protocol. The participant was requested to sit still on the bicycle ergometer for about 5 min before exercise to stabilize respiratory exchange. Continuous electrocardiographic monitoring was with the participant during the whole test, and blood pressure was measured regularly. The test was terminated for any of the following conditions: the actual HR exceeding 90% of the age-predicted maximum HR; the exertion score greater than 17 (Borg scale); SBP more than 200 mmHg; the participant was so exhausted that he/she was unable to complete the CPET safely; significant chest pain; ST segment depression more than 1 mm; severe arrhythmia. After attaining the maximal workload of exercise, the participant was asked to stop immediately, and then the data were recorded while the participant was sitting on the bicycle. Hence the subjects in our CPET had no “cool-down” phase. It was reported that small exercise load in the “cool-down” period might influence HRR value.19,20 Respiratory metabolic indexes were measured automatically through gas exchange monitoring (Innocor 6.0, Innovision Cor., Denmark). VO2peak was the average VO2 value in the last 20 s of the test. Powermax was the maximum exercise load in the test. The anerobic threshold (AT) and VO2 at AT (VO2AT) was detected by the computer software. The whole CPET test was instructed by a cardiologist and a rehabilitation physician.
Statistical analysis
Continuous variables were given in terms of mean ± standard deviation (SD) or interquartile ranges; categorical variables were given in terms of counts and percentages. Log-transformed method was used for variables with non-normal distributions to achieve normal distribution. Differences among healthy controls, M-OSAS group and M-S-OSAS group were assessed with variance analysis. Tukey’s post hoc test was used for continuous variables, and chi-squared test was used for categorical variables, respectively. Pearson correlation analysis was applied to determine the correlation of HRR and AHI with other clinical parameters. Multivariable linear regression analysis was applied to assess the association between HRR and PSG indexes after adjusting the well-known factors. P value < 0.05 was considered statistically significant. All statistical analyses were completed by SPSS software (version 17.0 for Windows; SPSS Inc., USA).
Results
The anthropometric and biochemical data of the subjects are demonstrated in Table 1. Eighty-seven children were classified as healthy, 35 as M-OSAS and 39 as M-S-OSAS. We did not find any difference in age, SBP, DBP and HRrest among the three groups. BMI was significantly higher in the M-S-OSAS group than that of the M-OSAS group or the control group (P < 0.05), while VO2peak and Powermax were lower in the M-S-OSAS group than the M-OSAS group or control group (P < 0.05). VO2AT and HRR values in either OSAS subgroup were lower than those of the control group, and the values in the M-S-OSAS group were even lower than those of the M-OSAS group (P < 0.05). As for the PSG indexes, LogODI, LogArousal index and Time/percentage below 90% SpO2 in the M-S-OSAS group were the highest among those three groups. Meanwhile, AverSpO2 and MinSpO2 in the M-S-OSAS group were the lowest among those three groups.
We tested for correlations of HRR and AHI values with other clinical parameters (Table 2). VO2peak, VO2AT, and AverSpO2 were positively correlated with HRR (r = 0.341, r = 0.263 and r = 0.168, respectively), and BMI, AHI and Time/percentage below 90% SpO2 were inversely correlated with HRR (r = −0.309, r = −0.190 and r = −0.162, respectively). There was a positive correlation between BMI and AHI (r = 0.248), whereas VO2peak, AverSpO2, and MinSpO2 were negatively correlated with AHI (r = −0.165, r = −0.081 and r = −0.156, respectively).
Multivariable linear regression analysis showed that SBP, DBP and HRrest were not significantly associated with HRR in model 1, after adjusting age, sex, and BMI. In model 2, we assessed whether the individual PSG index was independently associated with HRR. Among those indexes, AverSpO2 exhibited a significant association with HRR (β = 0.193, P < 0.05) and AHI with HRR (β = 0.183, P < 0.01). When the final regression analysis including all PSG indexes was carried out in model 3, the association between HRR and AHI was significant (β = 0.174, P < 0.01).
Discussion
The present study demonstrated that HRR values were significantly lower in M-S-OSAS children than M-OSAS children or normal children. HRR value was negatively correlated with BMI, AHI, and Time/percentage below 90%SpO2, while it was positively correlated with VO2peak, VO2AT, and AverSpO2. Moreover, HRR was independently negatively associated with AHI after adjusting age, sex, and BMI. These findings confirmed that HRR was closely associated with OSAS severity, which indicated that HRR might be a promising index for risk stratification and clinical management in children and adolescents with OSAS.
In adults, CPET is typically used to assess physical conditioning, and VO2peak is regarded as one of the best indicators of cardiovascular fitness. The peak exercise performance of OSAS adults has been revealed to be attenuated to a certain extent, and comparable clinical studies have been carried out in children in recent years. Evans et al.21 reported that children aged 7–12 years old with OSAS had a lower VO2peak than weight-matched children without OSAS. Meanwhile, they found OSAS children’s cardiac output at maximal exercise load was lower. Those two factors were responsible for impaired exercise performance. Similarly, it was observed that OSAS children had lower VO2peak, maximum HR at peak exercise, and higher SBP at peak exercise than healthy controls.22 In our study, we found that VO2peak in the M-S-OSAS group was smaller than that of the M-OSAS group or control group, although we did not find any difference between the M-OSAS group and the control group. Moreover, we would like to emphasize the significance of AT. It stands for the point above which the muscles derive their energy from nonoxygenic rather than oxygenic sources during exercise. We observed that VO2AT was lower in either OSAS subgroup than the control group, and it was even lower in the M-S-OSAS group than the M-OSAS group. These results indicated that both peak exercise performance and aerobic capacity were attenuated in M-S-OSAS children. VO2AT might be a more sensitive parameter than VO2peak to assess the relationship between exercise performance impairment and OSAS severity in children. Besides, regular exercise training, one of the fundamental obesity interventions, is encouraged in children with OSAS23; therefore, the difference observed in VO2AT gave us an indication that exercise prescription based on individual AT levels tested in CPET would be a more proper way to improve OSAS children’s exercise performance.
Intermittent hypoxia, hypercapnia and disruption of sleep through recurrent arousals is the fundamental feature of OSAS. A large number of previous investigations in OSAS adults demonstrated intermittent hypoxemia could lead to increased sympathetic nervous tone and the imbalance of sympathetic−parasympathetic activities.23 Nevertheless, investigations on autonomic nervous changes in children with OSAS are fewer. One of important reasons is any kind of invasive measurement is difficult to conduct in children.5 However, noninvasive tests of autonomic nervous function were identified for their association with severity of OSAS recently, such as overnight urinary noradrenaline and adrenaline levels24 and heart rate variability analysis using power spectral analysis25 and pulse arterial tonometry signals.26 HRR is an easy and reliable indicator of autonomic nervous tone, and slower HRR is predictive of cardiovascular adverse events independently both in adults and children.10,11 Previous studies in OSAS adults revealed HRR was significantly attenuated; Hargens et al.27 found this result through comparing overweight subjects with OSAS, overweight subjects without OSAS and normal weight controls without OSAS. Similarly, Maeder et al.28 found HRR was smaller in severe OSAS patients than that of mild-moderate OSAS patients, and AHI was inversely associated with HRR. To our knowledge, our study was the first study in China focusing on evaluating the role of HRR in children with OSAS.
Furthermore, the correlation analysis showed that VO2peak, VO2AT, and AverSpO2 were positively correlated with HRR, and BMI, AHI and Time/percentage below 90% SpO2 were inversely correlated with HRR. As for the negative relationship between BMI and HRR, it has been widely investigated in adults, and it was also demonstrated in children and adolescents.29 VO2peak and VO2AT belong to oxygen kinetics parameters in CPET. It was consistent with an adults’ report, which revealed maximal exercise capacity, aerobic metabolic dynamics and HRR were reduced in M-S-OSAS patients.30 What is more important was that HRR was also significantly associated with AHI, which was OSAS severity determination criteria, after multiple regression analysis. Combination of local structural abnormalities and neuromuscular factors is very important in the development of OSAS in children and adolescents.31 Adenotonsillar hypertrophy and obesity are commonly recognized as two of the most vital contributors.31 Autonomic dysfunction, oxidative stress, abnormal endothelial function and those mutual interactions are gradually deemed as important pathophysiological links in the past two decades’ clinical studies.32,33 Our observation would be helpful to explore the underlying mechanisms of autonomic dysfunction in children and adolescents with OSAS.
Despite our meaningful findings, some limitations should be mentioned. First, the number of OSAS children enrolled in the present study was small relatively; second, the subjects’ single night of laboratory PSG data were used for our analysis. Some reports proposed a single PSG test would likely introduce additional variability in measures of OSAS severity, while others supported the stability of AHI between repeated sleep studies; third, our work was a cross-sectional study. It would be more informative to have serial values of HRR and PSG parameters to explore causality. Further work enrolling more subjects investigating comprehensive and extensive markers representing autonomic nervous system are needed to verify as well as expand the findings of the current study.
References
Joosten, K. F. et al. How do we recognize the child with OSAS? Pediatr. Pulmonol. 52, 260–271 (2017).
Li, A. M. et al. Epidemiology of obstructive sleep apnoea syndrome in Chinese children: a two-phase community study. Thorax 65, 991–997 (2010).
Blechner, M. & Williamson, A. A. Consequences of obstructive sleep apnea in children. Curr. Probl. Pediatr. Adolesc. Health Care 46, 19–26 (2016).
Walter, L. M. et al. Autonomic dysfunction in children with sleep disordered breathing. Sleep Breath. 17, 605–613 (2013).
Kheirandish-Gozal, L., Bhattacharjee, R. & Gozal, D. Autonomic alterations and endothelial dysfunction in pediatric obstructive sleep apnea. Sleep Med. 11, 714–720 (2010).
Torre-Bouscoulet, L. et al. Mechanisms of cardiovascular damage in obstructive sleep apnea. Rev. Invest. Clin. 60, 502–516 (2008).
Muzumdar, H. V. et al. Changes in heart rate variability after adenotonsillectomy in children with obstructive sleep apnea. Chest 139, 1050–1059 (2011).
El-Hamad, F. et al. Altered nocturnal cardiovascular control in children with sleep-disordered breathing. Sleep 1, 40 (2017).
Liu, X. et al. Effect of adenotonsillectomy for childhood obstructive sleep apnea on nocturnal heart rate patterns. Sleep 41, zsy171 (2018).
Qiu, S. et al. Heart rate recovery and risk of cardiovascular events and all-cause mortality: a meta-analysis of prospective cohort studies. J. Am. Heart Assoc. 6, e005505 (2017).
Bjelakovic, L. et al. Heart rate recovery time in metabolically healthy and metabolically unhealthy obese children. Phys. Sportsmed. 45, 438–442 (2017).
Alvarado, A. M. et al. Heart rate recovery is impaired after maximal exercise testing in children with sickle cell anemia. J. Pediatr. 166, 389–393 (2015).
Giardini, A. et al. Impairment of heart rate recovery after peak exercise predicts poor outcome after pediatric heart transplantation. Impairment of heart rate recovery after peak exercise predicts poor outcome after pediatric heart transplantation. Circulation 128, S199–S204 (2013).
Arias Téllez, M. J., Soto-Sánchez, J. P. & Weisstaub, S. G. Physical fitness, cardiometabolic risk and heart rate recovery in Chilean children. Nutr. Hosp. 35, 44–49 (2018).
Fernando, R. J., Ravichandran, K. & Vaz, M. Aerobic fitness, heart rate recovery and heart rate recovery time in Indian school children. Indian J. Physiol. Pharmacol. 59, 407–413 (2015).
Lin, L. Y. et al. Inverse correlation between heart rate recovery and metabolic risks in healthy children and adolescents: insight from the National Health and Nutrition Examination Survey 1999-2002. Diabetes Care 31, 1015–1020 (2008).
American Academy of Sleep Medicine. International Classification of Sleep Disorders, 3rd edn (American Academy of Sleep Medicine, Darien, 2014).
Berry, R. B. et al. Rules for scoring respiratory events in sleep: update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J. Clin. Sleep Med. 8, 597–619 (2012).
Georgoulias, P. et al. Abnormal heart rate recovery immediately after treadmill testing: correlation with clinical, exercise testing, and myocardial perfusion parameters. J. Nucl. Cardiol. 10, 498–505 (2003).
Watanabe, J. et al. Heart rate recovery immediately after treadmill exercise and left ventricular systolic dysfunction as predictors of mortality: the case of stress echocardiography. Circulation 104, 1911–1916 (2001).
Evans, C. A. et al. Effects of obstructive sleep apnea and obesity on exercise function in children. Sleep 37, 1103–1110 (2014).
Damianidou, L. et al. Reduced exercise capacity in Greek children with mild to moderate obstructive sleep apneasyndrome. Pediatr. Pulmonol. 48, 1237–1245 (2013).
Lévy, P. et al. Obstructive sleep apnoea syndrome. Nat. Rev. Dis. Prim. 1, 15015 (2015).
O’Driscoll, D. M. et al. Increased sympathetic activity in children with obstructive sleep apnea: cardiovascular implications. Sleep Med. 12, 483–488 (2011).
Deng, Z. D. et al. Heart rate variability in pediatric obstructive sleep apnea. Conf. Proc. Ieee. Eng. Med. Biol. Soc. 1, 3565–3568 (2006).
O’Brien, L. M. & Gozal, D. Autonomic dysfunction in children with sleepdisordered breathing. Sleep 28, 747–752 (2005).
Hargens, T. A. et al. Attenuated heart rate recovery following exercise testing in overweight young men with untreated obstructive sleep apnea. Sleep 31, 104–110 (2008).
Maeder, M. T. et al. Association between heart rate recovery and severity of obstructive sleep apnea syndrome. Sleep Med. 9, 753–761 (2008).
Laguna, M. et al. Heart rate recovery is associated with obesity traits and related cardiometabolic riskfactors in children and adolescents. Nutr. Metab. Cardiovasc. Dis. 23, 995–1001 (2013).
Nanas, S. et al. Heart rate recovery and oxygen kinetics after exercise in obstructive sleep apneasyndrome. Clin. Cardiol. 33, 46–51 (2010).
Perez, C. Obstructive sleep apnea syndrome in children. Gen. Dent. 66, 46–50 (2018).
Villa, M. P. et al. Urinary concentration of 8-isoprostane as marker of severity of pediatric OSAS. Sleep Breath. 18, 723–729 (2014).
Zhang, F. et al. Polysomnographic correlates of endothelial function in children with obstructive sleep apnea. Sleep Med. 52, 45–50 (2018).
Acknowledgements
This work was supported by the Natural Science Research Program in Minhang District of Shanghai (2019MHZ129).
Author information
Authors and Affiliations
Contributions
H.W. and L.F. were responsible for data collection, analysis and interpretation; H.Z. was responsible for article revising; Y.Z. was responsible for data collection; B.L. was responsible for study design and article revising.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Patient consent
Written informed consents were provided by parents or children (whose age was more than 8 years old) before information was collected.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, H., Feng, L., Zheng, H. et al. Attenuated heart rate recovery in children and adolescents with obstructive sleep apnea syndrome. Pediatr Res 89, 900–904 (2021). https://doi.org/10.1038/s41390-020-0953-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-020-0953-z