Abstract
TP53 mutations are common in colorectal cancer (CRC). Most TP53 sequencing studies have been restricted to coding regions, but recent studies have revealed that splice mutations can generate transcript variants with distinct tumorigenic and prognostic properties. Here, we performed unrestricted sequencing of all coding sequences and splice regions of TP53 in a single-hospital series of 401 primary CRCs. TP53 splice mutations were detected in 4% of the cases (N = 16), considerably more frequent than reported in major databases, and they were mutually exclusive to exon mutations. RNA sequencing revealed high-level expression of aberrant transcript variants in the majority of splice mutated tumors (75%). Most variants were predicted to produce truncated TP53 proteins, including one sample expressing the potentially oncogenic and druggable p53ψ isoform. Despite heterogeneous transcript structures, downstream transcriptional profiling revealed that TP53 splice mutations had similar effects on TP53 target gene expression and pathway activity as exonic mutations. Intriguingly, TP53 splice mutations were associated with worse 5-year relapse-free survival in stage II disease, compared to both TP53 wild-type and exon mutations (P = 0.007). These data highlight the importance of including splice regions when examining the biological and clinical consequences of TP53 mutations in CRC.
Similar content being viewed by others
Introduction
Mutations in the tumor suppressor gene TP53 have long been known to be integral to colorectal carcinogenesis1,2, substantiated by its high mutation rate in manifest colorectal cancers (CRC)3. The TP53 mutation-spectrum and distribution show a large proportion of missense mutations clustering on hotspot codons, while truncating alterations, including nonsense, frameshift and splice site mutations, constitute ~20–25% of the mutations. Specifically, mutations at canonical splice sites account for approximately 2% of all detected TP53 mutations, according to major databases4,5. However, this may be underestimated since mutation analyses have been restricted to coding regions and the DNA-binding domain in many studies.
The consequences of TP53 splice mutations on the expression of the corresponding transcript variants are diverse, with smaller studies providing evidence of alternative splicing by exon skipping, intron retention, generation of novel splice sites and usage of cryptic splice sites6. On the other hand, the effects of TP53 splice mutations on downstream target gene expression and pathway activity have not been elucidated. Intriguingly, some truncating TP53 mutations have been shown to produce transcriptionally inactive isoforms leading to activation of a prometastatic cellular program7,8. These separation-of-function isoforms possess distinct oncogenic properties with potentially prognostic and therapeutic relevance.
In a single-hospital series of 401 primary CRCs, we investigated the downstream consequences of splice mutations in TP53, including expression of aberrant transcript variants, target genes, pathway activity and potential clinical consequences.
Results
The spectrum of coding and splice mutations in TP53
TP53 was mutated in 60% (N = 241) of the 401 cases, including 4% (N = 16) with splice mutations. The total number of mutations was 252, with double mutations in 2.7% (N = 11) of the tumors. Missense mutations were most common (69%, N = 173), while splice mutations accounted for 6% of all TP53 mutations (Fig. 1a) and were more frequent than the corresponding 2% reported in the IARC TP53 database4. Of the 16 TP53 splice mutations, one had not been previously described in cancer, and nine were novel to CRC (according to the IARC TP53 Database in April 2016; Supplementary Table S1)4. Thirteen were point mutations at the consensus splice sites, five in the donor site and eight in the acceptor site, of which 12 were single base substitutions and one was a single base deletion (Table 1). The three remaining splice mutations consisted of a point mutation located five basepairs downstream of exon 4, a duplication of six basepairs spanning the exon 7/intron 7 boundary and a deletion of 14 basepairs spanning the intron 5/exon 6 boundary. The majority of splice mutations (N = 13, 81%) was located adjacent to exons 5 to 8, reflecting the distribution of TP53 exon mutations (Fig. 1b). Notably, all splice mutations were mutually exclusive to TP53 exon mutations.
a Proportion of TP53 mutation types, plotted as the percentage of the total number of detected mutations (N = 252). b Frequency and distribution of TP53 splice (red) and exon (gray) mutations at each position along the gene (codon number is indicated below). The frequency is calculated relative to all mutations included in this plot (N = 236; exon mutations encompassing multiple codons, i.e. indels, are excluded). Frequency of splice mutations represents all mutations affecting the relevant splice region; see Table 1 for details. TAD transactivation domain, PRD proline-rich domain, DBD DNA-binding domain, NLS nuclear localization domain, OD oligomerization domain, Neg negative-regulation domain
Diverse aberrant transcript variants caused by TP53 splice mutations
RNA sequencing revealed altered splicing patterns corresponding with splice mutations in 12 (75%) of the 16 mutated samples, including exon skipping events, intron retention and usage of cryptic splice acceptor and donor sites not detected in an unmatched normal colonic mucosa sample (Table 1, Fig. 2a–c, Supplementary Fig. 1). Two different aberrant transcript variants per sample, in addition to the canonical splice variant, were identified in six samples (Table 1, Supplementary Fig. 1). Disparate splicing consequences between samples with mutations affecting the same canonical splice site were also observed. Samples 8 and 16 harbored base substitutions one and two nucleotides upstream of exon 8, respectively, but only the former exhibited aberrant splicing in this region (Fig. 2b). Furthermore, the estimated expression levels of the aberrant transcript variant(s) relative to the canonical splicing isoform per sample ranged from 7 to 88%, with a median of 31% (Fig. 2a, Supplementary Table S2). Collectively, this shows diverse effects of splice mutations on expressed transcript structures.
a Estimated relative expression levels of TP53 transcript variants in 16 splice mutated samples. Samples are ordered according to the relative expression level of the canonical splicing variant. Canonical splicing, cryptic splice sites and exon skipping events were quantified in the Sashimi plots, while intron retention values are the median depth of the relevant intronic region as measured by IRFinder. b Sashimi plots from two tumor samples with point mutation in the canonical splice acceptor site of intron 7 (marked with a dashed line) compared with a normal sample. Reads spanning exon junctions are represented by arcs, and each arc is labeled with the number of supporting reads. The arc representing an aberrant splicing event is colored in orange. Heights of bars reflect the read depth at each genomic position (reading frame right to left). Schematic visualization of the canonical TP53 transcript variant is shown in the top panel, and the two aberrant variants caused by the splice site mutation below, with coding sequences in light gray and noncoding sequences in black. In sample 8, 20 junction reads span transcripts using a cryptic acceptor site located 24 basepairs into exon 8, while 106 reads retain intron 7 (median depth of intron 7 as measured by IRFinder). Contrastingly, in both sample 16 and the normal sample all junction reads between exon 7 and exon 8 span the canonical splice sites. The transcript variant retaining intron 7 is predicted to generate a premature stop codon. The usage of an alternative splice acceptor site will lead to loss of nine amino acids in the 5′ part of exon 8 followed by disturbed reading frame but no generation of a premature stop codon. c Sashimi plot visualizing aberrant splicing variants in a sample with a TP53 mutation in the splice acceptor site of intron 6 compared with a nonmatched normal colonic mucosa sample. The transcript variant using a cryptic splice acceptor site located 49 basepairs upstream of the canonical splice site is identical with the p53ψ isoform, which is truncated due to the introduction of a premature stop codon. The aberrant transcript due to intron retention contains a premature stop codon in intron 6. Sashimi plots for the remaining samples with TP53 splice mutations are shown in Supplementary Fig. 1
Nonetheless, most of the aberrant transcript variants (N = 11, 85%) introduced a premature stop codon or altered reading frame, and were predicted to produce truncated TP53, if not degraded by nonsense-mediated decay (Table 1). Among these was the truncated p53ψ isoform which has previously been implicated in cancer8. This was expressed in one sample harboring a point mutation two basepairs upstream of exon 7 (c.673–2A, sample 10, Fig. 2c), as a result of activation of a cryptic splice acceptor site located 49 basepairs upstream of the canonical splice site in intron 6. This separation-of-function isoform has been shown to have poor prognostic associations and to induce epithelial to mesenchymal transition (EMT) in a transcriptionally independent manner8.
TP53 splice mutations lead to reduced expression of TP53 target genes
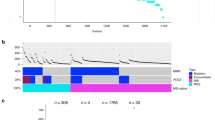
To analyze the downstream transcriptional consequences of TP53 splice mutations relative to exon mutations, TP53 gene expression was compared according to mutation type. Samples with missense mutations had the highest TP53 expression levels, similar to the expression in tumors with TP53 wild type (Fig. 3a). Samples with frameshift and nonsense mutations had a significant reduction in the expression of TP53 (P < 0.001), likely because several of these transcripts are eliminated by nonsense-mediated mRNA decay. Samples with splice mutations had a distinct intra-group dichotomy of high and low TP53 expression, but this was not related to the expression of truncating isoforms. However, differential gene expression analysis showed that both tumors with exon mutations (irrespective of the type) and splice mutations had downregulation of known TP53 transcriptional targets in comparison with wild-type tumors, including MDM2, FAS, HSPA4L and SPATA18 among the top five differentially expressed genes (Supplementary Table 3a)9. No genes were differentially expressed between the two mutation groups. Furthermore, to compare TP53 pathway activity, a single-sample gene set expression enrichment score was calculated for a set of 200 genes involved in the pathway10. All mutation types were associated with significantly lower TP53 signature scores than wild-type samples (P < 0.01), with only minor differences among the mutation types (Fig. 3b, Supplementary Table 3b). Accordingly, the downstream transcriptional consequences of TP53 splice mutations were similar to those of exon mutations.
a TP53 gene expression levels according to mutation type. Due to small sample sizes, in frame indels and silent mutations were not included. b TP53 signature score according to mutation type. Due to small sample sizes, in frame indels and silent mutations were not included. ssGSEA single-sample gene set enrichment analysis. c Kaplan−Meier survival curve showing 5-year relapse-free survival (RFS) for patients with TP53 wt, exon mutations and splice mutations in CRC stage II. d 5-year RFS according to TP53 mutation type in CRC stage II. Due to small sample size, in frame indels and silent mutations (N = 1) were not included
No significant differences between TP53 splice and exon mutations with respect to clinicopathological and molecular associations were found (Supplementary Table 4) and neither splice- nor exon-mutations were associated with survival across all stages (Supplementary Fig. 2a). However, stratification according to TNM stage showed that patients with TP53 splice mutations in stage II had worse 5-year RFS (P = 0.007, Fig. 3c) compared to both TP53 wild-type and exon mutations, although the sample number was low. The same trend was also seen in multivariable analysis comparing splice mutations to exon mutations (RFS: hazard ratio (HR) 4.82 (1.14–20.43); P = 0.033, Supplementary Fig. 2b). Of note, splice mutations were associated with inferior prognosis in stage II compared with other truncating mutation types, i.e. nonsense and frameshift mutations (P = 0.02, Fig. 3d), suggesting that the prognostic impact is not due to the loss of full-length TP53 protein per se.
Discussion
We show that splice mutations account for 6% of all TP53 mutations in CRC, which is threefold higher than estimated in major databases4,5. With respect to expression of corresponding aberrant transcript variants, the splice mutations show great diversity, but on the protein level, the majority are predicted to cause truncated isoforms, in line with the prevailing notion of the effects of such mutations. Nonetheless, all mutation types analyzed in this study, both splice mutations and the different types of exonic mutations, converged on downregulation of both TP53 target genes and signatures of TP53 pathway activity. This, together with the mutual exclusivity to exon mutations, indicate that TP53 splice mutations have similar biological relevance as alterations in protein-coding sequences.
Intriguingly, TP53 splice mutations were associated with a poor prognosis in stage II in our patient series. However, the low number of splice mutated samples within each stage and the lack of a detrimental effect of splice mutations across stages warrants cautious interpretation. Still, TP53 splice mutations have previously been shown to be enriched in metastatic CRC, an association not seen for TP53 mutations in general7. This could conceivably be explained by the generation of novel transcript variants with distinct biological properties beyond their direct effects on the TP53 network, and a propensity for relapse and metastasis, as described for p53ψ and other truncating mutations in exon 67. Our finding of stage-specific inferior prognosis for TP53 splice mutations compared with other truncating mutations supports the notion of these mutations having biological consequences beyond the loss of wild-type TP53 activity. Studies on non-small cell lung cancer have shown that mutations in the splice acceptor site of intron 6 generate the prometastatic isoform p53ψ, conveying poor prognostic properties. The EMT-inducing properties of this isoform can be inhibited through targeting its downstream effector Cyclophilin D by drugs such as cyclosporine A8. To our knowledge, this is the first study to detect the presence and underlying genesis of this potentially prognostic and druggable transcript variant in CRC. We cannot rule out distinct oncogenic properties of the other splice mutations detected in this study.
The lack of RNA sequencing data and TP53 transcript variant analysis from tumors with nonsplice mutations is a potential limitation of our study. Pan-cancer analysis has shown that synonymous and missense exonic mutations can cause aberrant splicing of tumor suppressor genes in general and most frequently in TP53. This occurs most commonly through mutations at residues adjacent to splice junctions11,12. Our study cannot rule out such splice-altering exonic mutations, but only two mutations in such residues were detected. This suggests only a very limited subset of splice-altering synonymous and missense mutations leading to truncated TP53 protein with distinct impact on TP53 pathway activity and clinical outcome.
In conclusion, splice mutations account for a 6% subgroup of TP53-mutated CRCs, which are mutually exclusive to exon mutations, cause aberrant transcript variant expression, downregulation of downstream target gene expression and pathway activity, and may identify high-risk stage II patients.
Material and methods
Patient material
A total of 401 fresh-frozen primary CRCs from a population-representative series of patients operated for CRC stage I−IV at Oslo University Hospital, Norway between 2005 and 2014 were included. DNA extraction was performed as previously described13,14. Comprehensive clinicopathological data were prospectively registered for all patients. The research conformed to the Helsinki Declaration and was approved by the Regional Committee for Medical and Health Research Ethics (REC number 1.2005.1629). Written consent was obtained from all patients. The research biobanks have been registered according to national legislation.
Mutation analyses, microsatellite instability status and CMS classification
TP53 mutation status was assessed in all 401 samples using Sanger sequencing of the entire coding region (exons 2–11), as well as the first ten and last ten nucleotides of each intron. Splice mutations were defined as any mutation affecting any of these intronic regions, based on the finding that intron mutations outside the invariant AG and GT dinucleotides in the consensus splice acceptor (SA) and splice donor (SD) site, respectively, may lead to missplicing15. Tumors harboring only synonymous mutations were classified as TP53 wild-type (wt) in survival analysis. Microsatellite instability (MSI) analysis, consensus molecular subtype (CMS) classification and mutation analyses for KRAS (exon 2: codons 12 and 13) and BRAF (codon 600) were performed in all samples as previously described16,17,18,19. KRAS exon 3 codon 61 was analyzed in a subset of samples (N = 127).
Differential gene expression and gene set enrichment analysis
All samples have previously been analyzed for gene expression at the exon-level using Affymetrix GeneChip® Human Exon 1.0 ST Array (HuEx, N = 199)13,20 and are available from GEO (GSE24550, GSE29638, GSE69182, GSE79959), or Human Transcriptome Array 2.0 (HTA 2.0, N = 202) (GSE79959)21 (GSE9652)18. Differential gene expression analysis and single-sample gene set enrichment analysis were performed using the R packages limma22 and GSVA23, respectively. The “HALLMARK_P53_PATHWAY” gene set from the Molecular Signatures Database (v5.2)10 was used to assess differential TP53 pathway activity among mutation groups. Dunn’s test implemented in the R package dunn.test was used for statistical significance testing. This is a nonparametric post hoc test after Kruskal−Wallis rank sum tests to identify medians that are significantly different between the possible pairs.
RNA sequencing, alignment and transcript variant analysis
RNA sequencing was performed for all samples harboring TP53 splice mutations (N = 16) detected by Sanger sequencing, and one normal sample for comparison. Strand-specific libraries for TruSeq total RNA sequencing (Illumina Inc.) were prepared according to protocol. Sequencing was performed on the Illumina HiSeq 2500 platform (2 × 101 bp, paired end) to an average of 77.4 million read pairs per sample. FastQC was run for quality control, and reads were aligned to the hg38 (HGMF genome_snp_tran) reference using HiSat2 v. 2.0.4 24. Aligned reads were inspected for transcript variants of TP53 using the Integrative Genome Viewer (IGV). The discovered transcript variants were visualized with the IGV built-in Sashimi plot function25. Exon skipping, usage of cryptic splice sites and canonical splicing in the relevant TP53 splice regions were detected and quantified by the Sashimi plot estimates, further described in legend to Fig. 2b. To avoid false positives due to sequencing or alignment artifacts, only aberrant transcript variants detected in ≥5 reads and accounting for ≥5% of all detected relevant reads in the sample are considered. As intron retention is not quantified by the Sashimi plots, we used the IRFinder algorithm to detect and quantify this class of splice variants26. Only events detected by the IRFinder algorithm with default settings were considered26. The threshold for calling intron retention was set at a minimum of 10% of transcripts retaining the intron (IR ratio > 0.1) and with a minimum coverage of three reads for each base pair of the intron, after excluding nonmeasurable intronic regions, as recommended by the authors. The expression levels for intron retention events are reported as the median depth of the relevant intronic region, given as output by IRFinder. Intron 4 of TP53 could not be evaluated by IRFinder, due to exclusion of low complexity regions or overlapping feature annotations preventing unique mapping of reads. For samples with splice mutations affecting this region, evidence of intron retention was visually analyzed in the Sashimi plots and quantified by the Bedtools coverageBed function27.
Statistical analyses
Statistical analyses were performed using the SPSS 21.0 software (SPSS Inc.). Fisher’s exact or Spearman correlation tests were applied when appropriate to evaluate associations between categorical variables. Five-year overall survival (OS) and 5-year relapse-free survival (RFS) plots were generated by the Kaplan–Meier method and the log-rank test was used to compare plots. The Cox proportional hazards model was used for univariable and multivariable analyses to assess the independence of prognostic factors. RFS and OS were calculated with date of surgery as starting point. All tests are two-tailed and P values < 0.05 were considered significant.
References
Fearon, E. R. & Vogelstein, B. A genetic model for colorectal tumorigenesis. Cell 61, 759–767 (1990).
Lothe, R. A. et al. Molecular genetic studies of tumor suppressor gene regions on chromosomes 13 and 17 in colorectal tumors. J. Natl. Cancer Inst. 84, 1100–1108 (1992).
TCGA. Comprehensive molecular characterization of human colon and rectal cancer. Nature 487, 330–337 (2012).
Bouaoun, L. et al. TP53 variations in human cancers: new lessons from the IARC TP53 database and genomics data. Hum. Mutat. 37, 865–876 (2016).
Leroy, B., Anderson, M. & Soussi, T. TP53 mutations in human cancer: database reassessment and prospects for the next decade. Hum. Mutat. 35, 672–688 (2014).
Holmila, R., Fouquet, C., Cadranel, J., Zalcman, G. & Soussi, T. Splice mutations in the p53 gene: case report and review of the literature. Hum. Mutat. 21, 101–102 (2003).
Shirole, N. H. et al. TP53 exon-6 truncating mutations produce separation of function isoforms with pro-tumorigenic functions. Elife 5, e17929 (2016).
Senturk, S. et al. p53Psi is a transcriptionally inactive p53 isoform able to reprogram cells toward a metastatic-like state. Proc. Natl Acad. Sci. USA 111, E3287–E3296 (2014).
Fischer, M. Census and evaluation of p53 target genes. Oncogene 36, 3943–3956 (2017).
Liberzon, A. et al. Molecular signatures database (MSigDB) 3.0. Bioinformatics 27, 1739–1740 (2011).
Jung, H. et al. Intron retention is a widespread mechanism of tumor-suppressor inactivation. Nat. Genet. 47, 1242–1248 (2015).
Giacomelli, A. O. et al. Mutational processes shape the landscape of TP53 mutations in human cancer. Nat. Genet. 50, 1381–1387 (2018).
Agesen, T. H. et al. ColoGuideEx: a robust gene classifier specific for stage II colorectal cancer prognosis. Gut 61, 1560–1567 (2012).
Berg, M. et al. Distinct high resolution genome profiles of early onset and late onset colorectal cancer integrated with gene expression data identify candidate susceptibility loci. Mol. Cancer 9, 100 (2010).
Anderson, S. L. et al. Familial dysautonomia is caused by mutations of the IKAP gene. Am. J. Hum. Genet. 68, 753–758 (2001).
Dienstmann, R. et al. Prediction of overall survival in stage II and III colon cancer beyond TNM system: a retrospective, pooled biomarker study. Ann. Oncol. 28, 1023–1031 (2017).
Vedeld, H. M. et al. CpG island methylator phenotype identifies high risk patients among microsatellite stable BRAF mutated colorectal cancers. Int J. Cancer 141, 967–976 (2017).
Sveen, A. et al. Colorectal cancer consensus molecular subtypes translated to preclinical models uncover potentially targetable cancer cell dependencies. Clin. Cancer Res. 24, 794–806 (2018).
Smeby, J. et al. CMS-dependent prognostic impact of KRAS and BRAFV600E mutations in primary colorectal cancer. Ann. Oncol. 29, 1227–1234 (2018).
Sveen, A. et al. ColoGuidePro: a prognostic 7-gene expression signature for stage III colorectal cancer patients. Clin. Cancer Res. 18, 6001–6010 (2012).
Sveen, A. et al. Multilevel genomics of colorectal cancers with microsatellite instability-clinical impact of JAK1 mutations and consensus molecular subtype 1. Genome Med. 9, 46 (2017).
Ritchie, M. E. et al. Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Hanzelmann, S., Castelo, R. & Guinney, J. GSVA: gene set variation analysis for microarray and RNA-Seq data. Bmc Bioinforma. 14, 7 (2013).
Kim, D., Langmead, B. & Salzberg, S. L. HISAT: a fast spliced aligner with low memory requirements. Nat. Methods 12, 357–360 (2015).
Katz, Y. et al. Quantitative visualization of alternative exon expression from RNA-seq data. Bioinformatics 31, 2400–2402 (2015).
Middleton, R. et al. IRFinder: assessing the impact of intron retention on mammalian gene expression. Genome Biol. 18, 51 (2017).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Acknowledgements
This work was supported by the Norwegian Cancer Society (project numbers 6824048-2016 to A.S. and 182759-2016 to R.A.L.); the foundation Stiftelsen Kristian Gerhard Jebsen; and the Research Council of Norway (FRIPRO Toppforsk, project number 250993 to R.A.L.).
Author information
Authors and Affiliations
Contributions
Conception and design: J.S., A.S. and R.A.L. Development of methodology: A.S, A.M.H., R.I.S. and R.A.L. Acquisition of data: J.S., I.A.E., S.A.D., M.H., M.G.G. and A.N. Analysis and interpretation of data: J.S., A.S., I.A.E., S.A.D., A.M.H., P.W.E., B.J., A.N. and R.A.L. Writing of the manuscript: J.S., A.S., M.G.G. and R.A.L. Review and/or revision of the manuscript: all authors. Study supervision: A.S. and R.A.L.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Smeby, J., Sveen, A., Eilertsen, I.A. et al. Transcriptional and functional consequences of TP53 splice mutations in colorectal cancer. Oncogenesis 8, 35 (2019). https://doi.org/10.1038/s41389-019-0141-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41389-019-0141-3
This article is cited by
-
Mapping alternative splicing events in colorectal cancer
Discover Oncology (2024)
-
Colorectal adenocarcinoma with clear cell changes: immunohistological and molecular findings in three cases
Virchows Archiv (2024)
-
Selective CK1α degraders exert antiproliferative activity against a broad range of human cancer cell lines
Nature Communications (2024)
-
The role of alternative pre-mRNA splicing in cancer progression
Cancer Cell International (2023)
-
Genomic signatures of past and present chromosomal instability in Barrett’s esophagus and early esophageal adenocarcinoma
Nature Communications (2023)