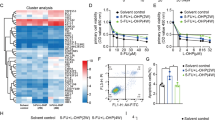
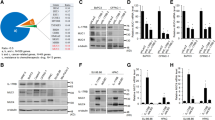
Abstract
Rapid development of drug resistance after chemotherapy is a major cause of treatment failure in individuals with pancreatic ductal adenocarcinoma (PDAC). In this study, we illustrate that tumor-derived interleukin 35 (IL-35) mediates the accelerated resistance of PDAC to gemcitabine (GEM). We observe that GEM resistance can spread from GEM-resistant PDAC cells to GEM-sensitive cells, and that IL-35 is responsible for the propagation of chemoresistance, which is supported by sequencing and experimental data. Additionally, we discover that GEM-resistant cells have significantly higher levels of IL-35 expression. Mechanistically, aberrantly expressed IL-35 triggers transcriptional activation of SOD2 expression via GP130-STAT1 signaling, scavenging reactive oxygen species (ROS) and leading to GEM resistance. Furthermore, GEM treatment stimulates IL-35 expression through activation of the NF-κB pathway, resulting in acquired chemoresistance. In the mouse model, a neutralizing antibody against IL-35 enhances the tumor suppressive effect of GEM. Collectively, our data suggests that IL-35 is critical in mediating GEM resistance in pancreatic cancer, and therefore could be a valuable therapeutic target in overcoming PDAC chemoresistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding authors.
References
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer Statistics, 2021. CA: A Cancer J Clin. 2021;71:7–33.
Nevala-Plagemann C, Hidalgo M, Garrido-Laguna I. From state-of-the-art treatments to novel therapies for advanced-stage pancreatic cancer. Nat Rev Clin Oncol. 2020;17:108–23.
Oettle H, Neuhaus P, Hochhaus A, Hartmann JT, Gellert K, Ridwelski K, et al. Adjuvant chemotherapy with gemcitabine and long-term outcomes among patients with resected pancreatic cancer: the CONKO-001 randomized trial. JAMA. 2013;310:1473–81.
Conroy T, Castan F, Lopez A, Turpin A, Ben Abdelghani M, Wei AC, et al. Five-Year Outcomes of FOLFIRINOX vs gemcitabine as adjuvant therapy for pancreatic cancer: a randomized clinical trial. JAMA Oncol. 2022;8:1571–8.
Dalgleish AG, Stebbing J, Adamson DJ, Arif SS, Bidoli P, Chang D, et al. Randomised, open-label, phase II study of gemcitabine with and without IMM-101 for advanced pancreatic cancer. Br J cancer. 2016;115:789–96.
Zheng H-C. The molecular mechanisms of chemoresistance in cancers. Oncotarget. 2017;8:59950.
Wilson T, Longley D, Johnston P. Chemoresistance in solid tumours. Ann Oncol. 2006;17:x315–x324.
Samanta K, Setua S, Kumari S, Jaggi M, Yallapu MM, Chauhan SC. Gemcitabine combination nano therapies for pancreatic cancer. Pharmaceutics. 2019;11:574.
Eshmuminov D, Aminjonov B, Palm RF, Malleo G, Schmocker RK, Abdallah R, et al. FOLFIRINOX or gemcitabine-based chemotherapy for borderline resectable and locally advanced pancreatic cancer: a multi-institutional, patient-level, meta-analysis and systematic review. Ann Surg Oncol. 2023;30:4417–28.
Ju HQ, Gocho T, Aguilar M, Wu M, Zhuang ZN, Fu J, et al. Mechanisms of overcoming intrinsic resistance to gemcitabine in pancreatic ductal adenocarcinoma through the redox modulation. Mol Cancer Ther. 2015;14:788–98.
Zhou X, An B, Lin Y, Ni Y, Zhao X, Liang X. Molecular mechanisms of ROS-modulated cancer chemoresistance and therapeutic strategies. Biomed Pharmacother. 2023;165:115036.
Srinivas US, Tan BWQ, Vellayappan BA, Jeyasekharan AD. ROS and the DNA damage response in cancer. Redox Biol. 2019;25:101084.
Mao X, Xu J, Wang W, Liang C, Hua J, Liu J, et al. Crosstalk between cancer-associated fibroblasts and immune cells in the tumor microenvironment: new findings and future perspectives. Mol Cancer. 2021;20:131.
Farran B, Nagaraju GP. Exosomes as therapeutic solutions for pancreatic cancer. Drug Discov today. 2020;25:2245–56.
Collison LW, Workman CJ, Kuo TT, Boyd K, Wang Y, Vignali KM, et al. The inhibitory cytokine IL-35 contributes to regulatory T-cell function. Nature. 2007;450:566–9.
Shen P, Roch T, Lampropoulou V, O’Connor RA, Stervbo U, Hilgenberg E, et al. IL-35-producing B cells are critical regulators of immunity during autoimmune and infectious diseases. Nature. 2014;507:366–70.
Collison LW, Delgoffe GM, Guy CS, Vignali KM, Chaturvedi V, Fairweather D, et al. The composition and signaling of the IL-35 receptor are unconventional. Nat Immunol. 2012;13:290–9.
Huang C, Li N, Li Z, Chang A, Chen Y, Zhao T, et al. Tumour-derived Interleukin 35 promotes pancreatic ductal adenocarcinoma cell extravasation and metastasis by inducing ICAM1 expression. Nat Commun. 2017;8:14035.
Huang C, Li Z, Li N, Li Y, Chang A, Zhao T, et al. Interleukin 35 expression correlates with microvessel density in pancreatic ductal adenocarcinoma, recruits monocytes, and promotes growth and angiogenesis of xenograft tumors in mice. Gastroenterology. 2018;154:675–88.
Collison LW, Chaturvedi V, Henderson AL, Giacomin PR, Guy C, Bankoti J, et al. IL-35-mediated induction of a potent regulatory T cell population. Nat Immunol. 2010;11:1093–101.
Mirzaei S, Mohammadi AT, Gholami MH, Hashemi F, Zarrabi A, Zabolian A, et al. Nrf2 signaling pathway in cisplatin chemotherapy: potential involvement in organ protection and chemoresistance. Pharmacol Res. 2021;167:105575.
Landriscina M, Maddalena F, Laudiero G, Esposito F. Adaptation to oxidative stress, chemoresistance, and cell survival. Antioxid redox Signal. 2009;11:2701–16.
Chang A, Liu L, Ashby JM, Wu D, Chen Y, O’Neill SS, et al. Recruitment of KMT2C/MLL3 to DNA damage sites mediates DNA damage responses and regulates PARP inhibitor sensitivity in cancer. Cancer Res. 2021;81:3358–73.
Spratlin JL, Mackey JR. Human equilibrative nucleoside transporter 1 (hENT1) in pancreatic adenocarcinoma: towards individualized treatment decisions. Cancers. 2010;2:2044–54.
Kohan HG, Boroujerdi M. Time and concentration dependency of P-gp, MRP1 and MRP5 induction in response to gemcitabine uptake in Capan-2 pancreatic cancer cells. Xenobiotica. 2015;45:642–52.
Azarabadi S, Abdollahi H, Torabi M, Salehi Z, Nasiri J. ROS generation, oxidative burst and dynamic expression profiles of ROS-scavenging enzymes of superoxide dismutase (SOD), catalase (CAT) and ascorbate peroxidase (APX) in response to Erwinia amylovora in pear (Pyrus communis L). Eur J Plant Pathol. 2017;147:279–94.
Furlan-Magaril M, Rincón-Arano H, Recillas-Targa F. Sequential chromatin immunoprecipitation protocol: ChIP-reChIP. DNA-Protein Interactions: Princ Protoc, Third Ed. 2009;543:253–66.
Arlt A, Gehrz A, Müerköster S, Vorndamm J, Kruse M-L, Fölsch UR, et al. Role of NF-κB and Akt/PI3K in the resistance of pancreatic carcinoma cell lines against gemcitabine-induced cell death. Oncogene. 2003;22:3243–51.
Zhang Z, Duan Q, Zhao H, Liu T, Wu H, Shen Q, et al. Gemcitabine treatment promotes pancreatic cancer stemness through the Nox/ROS/NF-κB/STAT3 signaling cascade. Cancer Lett. 2016;382:53–63.
Patel GK, Khan MA, Bhardwaj A, Srivastava SK, Zubair H, Patton MC, et al. Exosomes confer chemoresistance to pancreatic cancer cells by promoting ROS detoxification and miR-155-mediated suppression of key gemcitabine-metabolising enzyme, DCK. Br J Cancer. 2017;116:609–19.
Fan J, Wei Q, Koay EJ, Liu Y, Ning B, Bernard PW, et al. Chemoresistance transmission via exosome-mediated EphA2 transfer in pancreatic cancer. Theranostics. 2018;8:5986–94.
Liu K, Huang A, Nie J, Tan J, Xing S, Qu Y, et al. IL-35 regulates the function of immune cells in tumor microenvironment. Front Immunol. 2021;12:683332.
Tamama K, Funnell J, Cesarz Z. Bone morphogenic protein 2 is a key regulator of spheroidal aggregates of mesenchymal stem cells. FASEB J. 2016;30:695.692–695.692.
Zou Z, Chang H, Li H, Wang S. Induction of reactive oxygen species: an emerging approach for cancer therapy. Apoptosis. 2017;22:1321–35.
Raza MH, Siraj S, Arshad A, Waheed U, Aldakheel F, Alduraywish S, et al. ROS-modulated therapeutic approaches in cancer treatment. J Cancer Res Clin Oncol. 2017;143:1789–809.
Palma FR, He C, Danes JM, Paviani V, Coelho DR, Gantner BN, et al. Mitochondrial superoxide dismutase: what the established, the intriguing, and the novel reveal about a key cellular redox switch. Antioxid Redox Signal. 2020;32:701–14.
Fan C, Chen J, Wang Y, Wong YS, Zhang Y, Zheng W, et al. Selenocystine potentiates cancer cell apoptosis induced by 5-fluorouracil by triggering reactive oxygen species-mediated DNA damage and inactivation of the ERK pathway. Free Radic Biol Med. 2013;65:305–16.
Kim W, Lee S, Seo D, Kim D, Kim K, Kim E, et al. Cellular stress responses in radiotherapy. Cells. 2019;8:1105.
Mirlekar B. Tumor promoting roles of IL-10, TGF-β, IL-4, and IL-35: its implications in cancer immunotherapy. SAGE Open Med. 2022;10:20503121211069012.
Olson BM, Jankowska-Gan E, Becker JT, Vignali DA, Burlingham WJ, McNeel DG. Human prostate tumor antigen-specific CD8+ regulatory T cells are inhibited by CTLA-4 or IL-35 blockade. J Immunol. 2012;189:5590–601.
Xue W, Yan D, Kan Q. Interleukin-35 as an emerging player in tumor microenvironment. J Cancer. 2019;10:2074–82.
Funding
This work was supported by the National Natural Science Foundation of China (grants 82072659, 82272680, 82272799, 82030092, 82271895, 82072752, 82072657, 82072716, 82173295, 82072691, 82272767, 82273362, 82103006, 82103222, 82273259, 82273284, 82203402, 82203019 and 82203169), Tianjin Science Foundation for Distinguished Young Scholars (grants 19JCJQJC63100), the National Key Research and Development Program of China(2021YFA1201100); by the NIH grant R01CA233844 (to Shengyu Yang), Tianjin Research Innovation Project for Postgraduate Students(2022BKY162, 2021YJSB262), Tianjin Key Medical Discipline(Specialty) Construction Project(TJYXZDXK-009A), Key Programs of Science Foundation of Heilongjiang Province (ZD2019H009).
Author information
Authors and Affiliations
Contributions
JH and CH was responsible for designing and supervising the whole research. YG and HS was responsible for performing the experiments, analysing data and writing the manuscript. ZL, HL and JL conducted the data analyses. AC, HW and SG analysed and interpretated the experimental data. SY and PS contributed to revising the work. TZ, XW, YF, and SL provided supports on methodology and resources. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, H., Ge, Y., Liu, J. et al. Tumor-derived interleukin 35 mediates the dissemination of gemcitabine resistance in pancreatic adenocarcinoma. Oncogene 43, 776–788 (2024). https://doi.org/10.1038/s41388-024-02938-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-024-02938-0