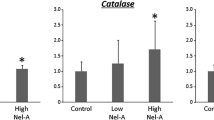
Abstract
Colorectal cancer (CRC) is a major health problem worldwide. Dicarbonyl electrophiles, such as isolevuglandins (isoLGs), are generated from lipid peroxidation and form covalent adducts with amine-containing macromolecules. We have shown high levels of adducts of isoLGs in colonic epithelial cells of patients with CRC. We thus investigated the role of these reactive aldehydes in colorectal cancer development. We found that 2-hydroxybenzylamine (2-HOBA), a natural compound derived from buckwheat seeds that acts as a potent scavenger of electrophiles, is bioavailable in the colon of mice after supplementation in the drinking water and does not affect the colonic microbiome. 2-HOBA reduced the level of isoLG adducts to lysine as well as tumorigenesis in models of colitis-associated carcinogenesis and of sporadic CRC driven by specific deletion of the adenomatous polyposis coli gene in colonic epithelial cells. In parallel, we found that oncogenic NRF2 activation and signaling were decreased in the colon of 2-HOBA-treated mice. Additionally, the growth of xenografted human HCT116 CRC cells in nude mice was significantly attenuated by 2-HOBA supplementation. In conclusion, 2-HOBA represents a promising natural compound for the prevention and treatment of CRC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Bailey CE, Hu CY, You YN, Bednarski BK, Rodriguez-Bigas MA, Skibber JM, et al. Increasing disparities in the age-related incidences of colon and rectal cancers in the United States, 1975-2010. JAMA Surg. 2015;150:17–22.
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49.
Fearon ER, Vogelstein B. A genetic model for colorectal tumorigenesis. Cell. 1990;61:759–67.
He TC, Sparks AB, Rago C, Hermeking H, Zawel L, da Costa LT, et al. Identification of c-MYC as a target of the APC pathway. Science. 1998;281:1509–12.
Vafa O, Wade M, Kern S, Beeche M, Pandita TK, Hampton GM, et al. c-Myc can induce DNA damage, increase reactive oxygen species, and mitigate p53 function: a mechanism for oncogene-induced genetic instability. Mol Cell. 2002;9:1031–44.
Castellone MD, Teramoto H, Williams BO, Druey KM, Gutkind JS. Prostaglandin E2 promotes colon cancer cell growth through a Gs-axin-beta-catenin signaling axis. Science. 2005;310:1504–10.
Eaden JA, Abrams KR, Mayberry JF. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut. 2001;48:526–35.
Crifo B, MacNaughton WK. Cells and mediators of inflammation as effectors of epithelial repair in the inflamed intestine. Am J Physiol Gastrointest Liver Physiol. 2022;322:G169–82.
Davies SS, May-Zhang LS, Boutaud O, Amarnath V, Kirabo A, Harrison DG. Isolevuglandins as mediators of disease and the development of dicarbonyl scavengers as pharmaceutical interventions. Pharmacol Ther. 2020;205:107418.
Carrier EJ, Amarnath V, Oates JA, Boutaud O. Characterization of covalent adducts of nucleosides and DNA formed by reaction with levuglandin. Biochemistry. 2009;48:10775–81.
Boutaud O, Brame CJ, Salomon RG, Roberts LJ 2nd, Oates JA. Characterization of the lysyl adducts formed from prostaglandin H2 via the levuglandin pathway. Biochemistry. 1999;38:9389–96.
Carrier EJ, Zagol-Ikapitte I, Amarnath V, Boutaud O, Oates JA. Levuglandin forms adducts with histone H4 in a cyclooxygenase-2-dependent manner, altering its interaction with DNA. Biochemistry. 2014;53:2436–41.
Esterbauer H. Cytotoxicity and genotoxicity of lipid-oxidation products. Am J Clin Nutr. 1993;57:779S–785S.
Gobert AP, Boutaud O, Asim M, Zagol-Ikapitte IA, Delgado AG, Latour YL, et al. Dicarbonyl electrophiles mediate inflammation-induced gastrointestinal carcinogenesis. Gastroenterology. 2021;160:1256–68.
Skrzydlewska E, Sulkowski S, Koda M, Zalewski B, Kanczuga-Koda L, Sulkowska M. Lipid peroxidation and antioxidant status in colorectal cancer. World J Gastroenterol. 2005;11:403–6.
Leung EY, Crozier JE, Talwar D, O’Reilly DS, McKee RF, Horgan PG, et al. Vitamin antioxidants, lipid peroxidation, tumour stage, the systemic inflammatory response and survival in patients with colorectal cancer. Int J Cancer. 2008;123:2460–4.
Koyama M, Obata Y, Sakamura S. Identification of hydroxybenzylamines in buckwheat seeds (Fagopyrum esculentum Moench). Agric Biol Chem. 1971;35:1870–9.
Pitchford LM, Smith JD, Abumrad NN, Rathmacher JA, Fuller JC Jr. Acute and 28-day repeated dose toxicity evaluations of 2-hydroxybenzylamine acetate in mice and rats. Regul Toxicol Pharmacol. 2018;98:190–8.
Fuller JC Jr., Pitchford LM, Abumrad NN, Rathmacher JA. Subchronic (90-day) repeated dose toxicity study of 2-hydroxybenzylamine acetate in rats. Regul Toxicol Pharmacol. 2018;99:225–32.
Fuller JC Jr., Pitchford LM, Abumrad NN, Rathmacher JA. Subchronic (90-day) repeated dose oral toxicity study of 2-hydroxybenzylamine acetate in rabbit. Regul Toxicol Pharmacol. 2018;100:52–58.
Wu J, Saleh MA, Kirabo A, Itani HA, Montaniel KR, Xiao L, et al. Immune activation caused by vascular oxidation promotes fibrosis and hypertension. J Clin Invest. 2016;126:50–67.
Egnatchik RA, Brittain EL, Shah AT, Fares WH, Ford HJ, Monahan K, et al. Dysfunctional BMPR2 signaling drives an abnormal endothelial requirement for glutamine in pulmonary arterial hypertension. Pulm Circ. 2017;7:186–99.
Tao H, Huang J, Yancey PG, Yermalitsky V, Blakemore JL, Zhang Y, et al. Scavenging of reactive dicarbonyls with 2-hydroxybenzylamine reduces atherosclerosis in hypercholesterolemic Ldlr(−/−) mice. Nat Commun. 2020;11:4084.
Davies SS, Bodine C, Matafonova E, Pantazides BG, Bernoud-Hubac N, Harrison FE, et al. Treatment with a gamma-ketoaldehyde scavenger prevents working memory deficits in hApoE4 mice. J Alzheimers Dis. 2011;27:49–59.
Pitchford LM, Rathmacher JA, Fuller JC Jr., Daniels JS, Morrison RD, Akers WS, et al. First-in-human study assessing safety, tolerability, and pharmacokinetics of 2-hydroxybenzylamine acetate, a selective dicarbonyl electrophile scavenger, in healthy volunteers. BMC Pharmacol Toxicol. 2019;20:1.
Pitchford LM, Driver PM, Fuller JC Jr., Akers WS, Abumrad NN, Amarnath V, et al. Safety, tolerability, and pharmacokinetics of repeated oral doses of 2-hydroxybenzylamine acetate in healthy volunteers: a double-blind, randomized, placebo-controlled clinical trial. BMC Pharmacol Toxicol. 2020;21:3.
Zagol-Ikapitte IA, Matafonova E, Amarnath V, Bodine CL, Boutaud O, Tirona RG, et al. Determination of the pharmacokinetics and oral bioavailability of salicylamine, a potent gamma-ketoaldehyde scavenger, by LC/MS/MS. Pharmaceutics. 2010;2:18–29.
Gobert AP, Asim M, Smith TM, Williams KJ, Barry DP, Allaman MM, et al. The nutraceutical electrophile scavenger 2-hydroxybenzylamine (2-HOBA) attenuates gastric cancer development caused by Helicobacter pylori. Biomed Pharmacother. 2023;158:114092.
Kim J, Lee HK. Potential role of the gut microbiome in colorectal cancer progression. Front Immunol. 2021;12:807648.
Lee IA, Bae EA, Hyun YJ, Kim DH. Dextran sulfate sodium and 2,4,6-trinitrobenzene sulfonic acid induce lipid peroxidation by the proliferation of intestinal gram-negative bacteria in mice. J Inflamm (Lond). 2010;7:7.
Lei L, Yang J, Zhang J, Zhang G. The lipid peroxidation product EKODE exacerbates colonic inflammation and colon tumorigenesis. Redox Biol. 2021;42:101880.
Zhang T, Ahn K, Emerick B, Modarai SR, Opdenaker LM, Palazzo J, et al. APC mutations in human colon lead to decreased neuroendocrine maturation of ALDH+ stem cells that alters GLP-2 and SST feedback signaling: clue to a link between WNT and retinoic acid signalling in colon cancer development. PLoS One. 2020;15:e0239601.
Huang EH, Hynes MJ, Zhang T, Ginestier C, Dontu G, Appelman H, et al. Aldehyde dehydrogenase 1 is a marker for normal and malignant human colonic stem cells (SC) and tracks SC overpopulation during colon tumorigenesis. Cancer Res. 2009;69:3382–9.
Ma Q. Role of NRF2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol. 2013;53:401–26.
Torrente L, Maan G, Oumkaltoum Rezig A, Quinn J, Jackson A, Grilli A, et al. High NRF2 levels correlate with poor prognosis in colorectal cancer patients and with sensitivity to the kinase inhibitor AT9283 in vitro. Biomolecules. 2020;10:1365.
Hammad A, Zheng ZH, Gao Y, Namani A, Shi HF, Tang X. Identification of novel Nrf2 target genes as prognostic biomarkers in colitis-associated colorectal cancer in Nrf2-deficient mice. Life Sci. 2019;238:116968.
Song CH, Kim N, Nam RH, Choi SI, Kang C, Jang JY, et al. Nuclear factor erythroid 2-related factor 2 knockout suppresses the development of aggressive colorectal cancer formation induced by azoxymethane/dextran sulfate sodium-treatment in female mice. J Cancer Prev. 2021;26:41–53.
Papaioannou D, Cooper KL, Carroll C, Hind D, Squires H, Tappenden P, et al. Antioxidants in the chemoprevention of colorectal cancer and colorectal adenomas in the general population: a systematic review and meta-analysis. Colorectal Dis. 2011;13:1085–99.
Roberts LJ 2nd, Oates JA, Linton MF, Fazio S, Meador BP, Gross MD, et al. The relationship between dose of vitamin E and suppression of oxidative stress in humans. Free Radic Biol Med. 2007;43:1388–93.
Funding
This work was funded by NIH grants R41CA257262 (KTW and JAR), R01DK128200 (KTW), P01CA116087 (KTW); Department of Defense grant W81XWH-18-1-0301 (KTW); Veterans Affairs Merit Review grants I01CX002171 (KTW) and I01BX004366 (LAC); Senior Research Award 703003 from the Crohn’s and Colitis Foundation (KTW and APG); the Thomas F. Frist Sr. Endowment (K.T.W.); and the Vanderbilt Center for Mucosal Inflammation and Cancer (KTW). Histopathology studies were supported in part by the Tissue Morphology Subcore of the Translational Analysis Core of NIH grant P30DK058404.
Author information
Authors and Affiliations
Contributions
APG was responsible for conceptualization, investigation, formal analysis, creation of the figures, writing the original draft, review and editing the manuscript, and funding acquisition. MA, TMS, KJW, DPB, MMA, KMM, CVH, AGD performed the experiments. SZ was responsible for data curation and formal analysis of the colonic microbiome. MBP analyzed the immunostaining. MKW performed the formal analysis of the histology. LAC performed the experiments and reviewed and edited the manuscript. JAR was responsible for conceptualization, formal analysis, review and editing the manuscript, and funding acquisition. KTW was responsible for conceptualization, review and editing the manuscript, and funding acquisition.
Corresponding author
Ethics declarations
Competing interests
APG and KTW are named inventors on a Vanderbilt University patent application for the use of electrophile scavengers. In addition, APG and KTW are named on a licensing agreement between Vanderbilt University and MTI Biotech for the future use of electrophile scavengers. All other authors have declared that no conflict of interest exists. JAR is an employee of MTI BioTech and is listed as an inventor on 2-HOBA patent applications. MTI BioTech intends to market/license 2-HOBA for commercial purposes.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Gobert, A.P., Asim, M., Smith, T.M. et al. Electrophilic reactive aldehydes as a therapeutic target in colorectal cancer prevention and treatment. Oncogene 42, 1685–1691 (2023). https://doi.org/10.1038/s41388-023-02691-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-023-02691-w