Abstract
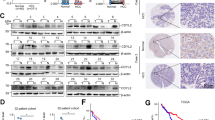
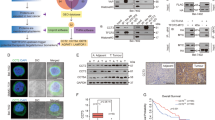
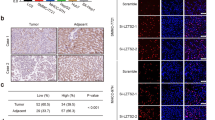
Coiled-coil domain containing 3 (CCDC3) was previously shown to regulate liver lipid metabolism as a secretory protein. Here, we report an unexpected intracellular role of CCDC3 as a tumor suppressor in breast cancer (BrC). Bioinformatics datasets analysis showed that CCDC3 is under-expressed in BrCs, while its higher levels are correlated with higher overall survival and lower relapse of cancer patients, and CCDC3 is positively correlated with p53 and its target genes. Ectopic CCDC3 markedly suppressed proliferation, colony formation, and xenograft tumor growth by augmenting p53 activity in BrC cells. Depletion of endogenous CCDC3 by CRISPR-Cas9 increased proliferation and drug resistance of BrC cells by alleviating 5-Fluorouracil (5-FU)-induced p53 level and activity. Mechanistically, CCDC3 bound to the C-termini of p53 and MDM2, consequently stabilizing p53 in the nucleus and impairing MDM2 recruitment of p53 to the 26S proteosome without inhibiting p53 ubiquitination. p53 induced CCDC3 expression by binding to its promoter in BrC cells. Our results unveil a unique mechanism underlying CCDC3 activation of p53 in a positive feedback fashion to suppress BrC growth.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Vousden KH, Prives C. Blinded by the light: the growing complexity of p53. Cell 2009;137:413–31.
Kenzelmann Broz D, Attardi LD. In vivo analysis of p53 tumor suppressor function using genetically engineered mouse models. Carcinogenesis. 2010;31:1311–8.
Brady CA, Attardi LD. p53 at a glance. J Cell Sci. 2010;123:2527–32.
Vousden KH, Lu X. Live or let die: the cell’s response to p53. Nat Rev Cancer. 2002;2:594–604.
Liao JM, Cao B, Deng J, Zhou X, Strong M, Zeng S, et al. TFIIS.h, a new target of p53, regulates transcription efficiency of pro-apoptotic bax gene. Sci Rep. 2016;6:23542.
Liu YY, Tanikawa C, Ueda K, Matsuda K. INKA2, a novel p53 target that interacts with the serine/threonine kinase PAK4. Int J Oncol. 2019;54:1907–20.
Moll UM, Wolff S, Speidel D, Deppert W. Transcription-independent pro-apoptotic functions of p53. Curr Opin Cell Biol. 2005;17:631–6.
Leu JI, Dumont P, Hafey M, Murphy ME, George DL. Mitochondrial p53 activates Bak and causes disruption of a Bak-Mcl1 complex. Nat Cell Biol. 2004;6:443–50.
Levine AJ, Oren M. The first 30 years of p53: growing ever more complex. Nat Rev Cancer. 2009;9:749–58.
Kruse JP, Gu W. Modes of p53 regulation. Cell 2009;137:609–22.
Haupt Y, Maya R, Kazaz A, Oren M. Mdm2 promotes the rapid degradation of p53. Nature 1997;387:296–9.
Honda R, Tanaka H, Yasuda H. Oncoprotein MDM2 is a ubiquitin ligase E3 for tumor suppressor p53. FEBS Lett. 1997;420:25–27.
Joazeiro CA, Weissman AM. RING finger proteins: mediators of ubiquitin ligase activity. Cell 2000;102:549–52.
Fang S, Jensen JP, Ludwig RL, Vousden KH, Weissman AM. Mdm2 is a RING finger-dependent ubiquitin protein ligase for itself and p53. J Biol Chem. 2000;275:8945–51.
Hu W, Feng Z, Levine AJ. The regulation of multiple p53 stress responses is mediated through MDM2. Genes Cancer. 2012;3:199–208.
Zhang Q, Zeng SX, Lu H. Targeting p53-MDM2-MDMX loop for cancer therapy. Subcell Biochem. 2014;85:281–319.
Pomerantz J, Schreiber-Agus N, Liegeois NJ, Silverman A, Alland L, Chin L, et al. The Ink4a tumor suppressor gene product, p19Arf, interacts with MDM2 and neutralizes MDM2’s inhibition of p53. Cell 1998;92:713–23.
Zhou X, Liao JM, Liao WJ, Lu H. Scission of the p53-MDM2 loop by ribosomal proteins. Genes Cancer. 2012;3:298–310.
Zhou X, Hao Q, Liao P, Luo S, Zhang M, Hu G et al. Nerve growth factor receptor negates the tumor suppressor p53 as a feedback regulator. Elife. 2016;5:e15099.
Chao T, Zhou X, Cao B, Liao P, Liu H, Chen Y, et al. Pleckstrin homology domain-containing protein PHLDB3 supports cancer growth via a negative feedback loop involving p53. Nat Commun. 2016;7:13755.
Goldberg AL. Protein degradation and protection against misfolded or damaged proteins. Nature 2003;426:895–9.
Zwickl P, Ng D, Woo KM, Klenk HP, Goldberg AL. An archaebacterial ATPase, homologous to ATPases in the eukaryotic 26 S proteasome, activates protein breakdown by 20 S proteasomes. J Biol Chem. 1999;274:26008–14.
Sdek P, Ying H, Chang DL, Qiu W, Zheng H, Touitou R, et al. MDM2 promotes proteasome-dependent ubiquitin-independent degradation of retinoblastoma protein. Mol Cell. 2005;20:699–708.
Orlowski M, Wilk S. Ubiquitin-independent proteolytic functions of the proteasome. Arch Biochem Biophys. 2003;415:1–5.
Kulikov R, Letienne J, Kaur M, Grossman SR, Arts J, Blattner C. Mdm2 facilitates the association of p53 with the proteasome. Proc Natl Acad Sci USA. 2010;107:10038–43.
Kobayashi S, Fukuhara A, Taguchi T, Matsuda M, Tochino Y, Otsuki M, et al. Identification of a new secretory factor, CCDC3/Favine, in adipocytes and endothelial cells. Biochem Biophys Res Commun. 2010;392:29–35.
Kobayashi S, Fukuhara A, Otsuki M, Suganami T, Ogawa Y, Morii E, et al. Fat/vessel-derived secretory protein (Favine)/CCDC3 is involved in lipid accumulation. J Biol Chem. 2015;290:7443–51.
Liao W, Liu H, Zhang Y, Jung JH, Chen J, Su X, et al. Ccdc3: a new P63 target involved in regulation of liver lipid metabolism. Sci Rep. 2017;7:9020.
Azad AK, Chakrabarti S, Xu Z, Davidge ST, Fu Y. Coiled-coil domain containing 3 (CCDC3) represses tumor necrosis factor-alpha/nuclear factor kappaB-induced endothelial inflammation. Cell Signal. 2014;26:2793–2800.
Ju J, Schmitz JC, Song B, Kudo K, Chu E. Regulation of p53 expression in response to 5-fluorouracil in human cancer RKO cells. Clin Cancer Res. 2007;13:4245–51.
Mukhopadhyay D, Riezman H. Proteasome-independent functions of ubiquitin in endocytosis and signaling. Science 2007;315:201–5.
Murata T, Shimotohno K. Ubiquitination and proteasome-dependent degradation of human eukaryotic translation initiation factor 4E. J Biol Chem. 2006;281:20788–20800.
Thrower JS, Hoffman L, Rechsteiner M, Pickart CM. Recognition of the polyubiquitin proteolytic signal. EMBO J. 2000;19:94–102.
Zhang Y, Zhang Q, Zeng SX, Hao Q, Lu H. Inauhzin sensitizes p53-dependent cytotoxicity and tumor suppression of chemotherapeutic agents. Neoplasia. 2013;15:523–34.
Choong ML, Yang H, Lee MA, Lane DP. Specific activation of the p53 pathway by low dose actinomycin D: a new route to p53 based cyclotherapy. Cell Cycle. 2009;8:2810–8.
Zhang Q, Zeng SX, Zhang Y, Zhang Y, Ding D, Ye Q, et al. A small molecule Inauhzin inhibits SIRT1 activity and suppresses tumour growth through activation of p53. EMBO Mol Med. 2012;4:298–312.
Vassilev LT, Vu BT, Graves B, Carvajal D, Podlaski F, Filipovic Z, et al. In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science. 2004;303:844–8.
Zhang Y, Lu H. Signaling to p53: ribosomal proteins find their way. Cancer Cell. 2009;16:369–77.
Sparks A, Dayal S, Das J, Robertson P, Menendez S, Saville MK. The degradation of p53 and its major E3 ligase Mdm2 is differentially dependent on the proteasomal ubiquitin receptor S5a. Oncogene. 2014;33:4685–96.
Yuan J, Luo K, Zhang L, Cheville JC, Lou Z. USP10 regulates p53 localization and stability by deubiquitinating p53. Cell 2010;140:384–96.
Ahn J, Prives C. The C-terminus of p53: the more you learn the less you know. Nat Struct Biol. 2001;8:730–2.
Li M, Brooks CL, Wu-Baer F, Chen D, Baer R, Gu W. Mono- versus polyubiquitination: differential control of p53 fate by Mdm2. Science. 2003;302:1972–5.
Liao JM, Lu H. ChIP for identification of p53 responsive DNA promoters. Methods Mol Biol. 2013;962:201–10.
Acknowledgements
We thank James Jackson, Wesley D. Frey, Joe Gray, and Jean-Paul Borg for offering some BrC cell lines (Cal 51, MDA-MB-175, MPE600, SKBR7) and the CRISPR-p53KO-mCherry construct (James Jackson) and Heather Machado for sharing her animal protocol (Mammary fat pad injection). HL and SXZ were supported in part by NIH-NCI grants R01CA095441, R01CA234605, and R01CA127724 as well as R21CA272890. HL was also in part supported by the Reynolds and Ryan Families Endowed Chair fund and the NIH-NCI grant U01CA252965.
Author information
Authors and Affiliations
Contributions
CYL and HML equally conducted the majority of the experiments, analyzed the results and organized them into figures under supervision of SXZ and HL; JHJ initiated the study and conducted some experiments on regulation of p53 by CCDC3; JQW and AS assisted in the animal experiment; CL assisted in some in vitro assay; YWZ assisted in the animal protocol; RLS assisted in cellular fractionation; SXZ conducted some in vitro and animal experiments with HML and AS; SXZ and HL instructed the whole project; CYL and HL composed the manuscript; CYL, HML, SXZ, and HL revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, C., Lee, H., Jung, J.H. et al. Coiled-coil domain containing 3 suppresses breast cancer growth by protecting p53 from proteasome-mediated degradation. Oncogene 42, 154–164 (2023). https://doi.org/10.1038/s41388-022-02541-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-022-02541-1
This article is cited by
-
Visualization of breast cancer-related protein synthesis from the perspective of bibliometric analysis
European Journal of Medical Research (2023)
-
A novel coiled-coil domain containing-related gene signature for predicting prognosis and treatment effect of breast cancer
Journal of Cancer Research and Clinical Oncology (2023)