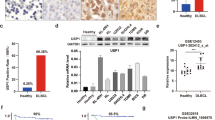
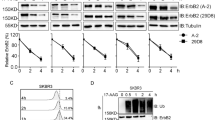
Abstract
Despite the establishment of novel therapeutic interventions, multiple myeloma (MM) remains invariably incurable due to development of drug resistance and subsequent relapse, which are attributed to activation of oncogenic pathways such as autophagy. Deubiquitinating enzymes (DUBs) are promising targets to overcome resistance to proteasome inhibitor-based treatment. Ubiquitin-specific protease-12 (USP12) is a DUB with a known prognostic value in several cancers. We found that USP12 protein levels were significantly higher in myeloma patient samples than in non-cancerous human samples. Depletion of USP12 suppressed cell growth and clonogenicity and inhibited autophagy. Mechanistic studies showed that USP12 interacted with, deubiquitylated and stabilized the critical autophagy mediator HMGB1 (high mobility group box-1) protein. Knockdown of USP12 decreased the level of HMGB1 and suppressed HMGB1-mediated autophagy in MM. Furthermore, basal autophagy activity associated with USP12/HMGB1 was elevated in bortezomib (BTZ)-resistant MM cell lines. USP12 depletion, concomitant with a reduced expression of HMGB1, suppressed autophagy and increased the sensitivity of resistant cells to BTZ. Collectively, our findings have identified an important role of the deubiquitylase USP12 in pro-survival autophagy and resultant BTZ resistance in MM by stabilizing HMGB1, suggesting that the USP12/HMGB1 axis might be pursued as a potential diagnostic and therapeutic target in human MM.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Kumar SK, Anderson KC. Anderson, immune therapies in multiple myeloma. Clin Cancer Res. 2016;22:5453–60.
Kumar SK, Dispenzieri A, Lacy MQ, Gertz MA, Buadi FK, Pandey S, et al. Continued improvement in survival in multiple myeloma: changes in early mortality and outcomes in older patients. Leukemia. 2014;28:1122–8.
Kumar SK, Rajkumar V, Kyle RA, Duin MV, Sonneveld P, Mateos MV, et al. Multiple myeloma. Nat Rev Dis Prim. 2017;3:17046.
Pinto V, Bergantim R, Caires HR, Seca H, Guimarães JE, Vasconcelos MH. Multiple myeloma: available therapies and causes of drug resistance. Cancers (Basel). 2020;12:407.
Milan E, Fabbri M, Cenci S. Autophagy in plasma cell ontogeny and malignancy. J Clin Immunol. 2016;36:18–24.
Pengo N, Scolari M, Oliva L, Milan E, Mainoldi F, Raimondi A, et al. Plasma cells require autophagy for sustainable immunoglobulin production. Nat Immunol. 2013;14:298–305.
Vogl DT, Stadtmauer EA, Tan KS, Heitjan DF, Davis LE, Pontiggia L, et al. Combined autophagy and proteasome inhibition: a phase 1 trial of hydroxychloroquine and bortezomib in patients with relapsed/refractory myeloma. Autophagy. 2014;10:1380–90.
Jarauta V, Jaime P, Gonzalo O, Miguel DD, Ramírez-Labrada A, Martínez-Lostao L, et al. Inhibition of autophagy with chloroquine potentiates carfilzomib-induced apoptosis in myeloma cells in vitro and in vivo. Cancer Lett. 2016;382:1–10.
Lu Y, Wang Y, Xu H, Shi C, Jin FY, Li W. Profilin 1 induces drug resistance through Beclin1 complex-mediated autophagy in multiple myeloma. Cancer Sci. 2018;109:2706–16.
Roy M, Liang L, Xiao XJ, Peng YL, Luo YH, Zhou WH, et al. Lycorine downregulates HMGB1 to inhibit autophagy and enhances bortezomib activity in multiple myeloma. Theranostics. 2016;6:2209–24.
Zhang M, Zhang MJ, He J, Liu ZQ, Lu Y, Zheng YH, et al. Anti-beta(2)-microglobulin monoclonal antibodies overcome bortezomib resistance in multiple myeloma by inhibiting autophagy. Oncotarget. 2015;6:8567–78.
Zhang H, Pang YH, Ma CB, Li JY, Wang HQ, Shao ZH, et al. ClC5 decreases the sensitivity of multiple myeloma cells to bortezomib via promoting prosurvival autophagy. Oncol Res. 2018;26:421–9.
Zhang X, Li WM, Wang CL, Leng XY, Lian SL, Feng JB, et al. Inhibition of autophagy enhances apoptosis induced by proteasome inhibitor bortezomib in human glioblastoma U87 and U251 cells. Mol Cell Biochem. 2014;385:265–75.
Komander D, Clague MJ, Urbe S. Breaking the chains: structure and function of the deubiquitinases. Nat Rev Mol Cell Biol. 2009;10:550–63.
Joo HY, Jones A, Yang CY, Zhai L, Smith AD, Zhang Z, et al. Regulation of histone H2A and H2B deubiquitination and Xenopus development by USP12 and USP46. J Biol Chem. 2011;286:7190–201.
Jahan AS, Lestra M, Swee LK, Fan Y, Lamers MM, Tafesse FG, et al. Usp12 stabilizes the T-cell receptor complex at the cell surface during signaling. Proc Natl Acad Sci USA. 2016;113:E705–14.
McClurg UL, Chit NC, Azizyan M, Edwards J, Nabbi A, Riabowol KT, et al. Molecular mechanism of the TP53-MDM2-AR-AKT signalling network regulation by USP12. Oncogene. 2018;37:4679–91.
Moretti J, Chastagner P, Liang CC, Cohn MA, Israël A, Brou C, et al. The ubiquitin-specific protease 12 (USP12) is a negative regulator of notch signaling acting on notch receptor trafficking toward degradation. J Biol Chem. 2012;287:29429–41.
Nayak TK, Alamuru-Yellapragada NP, Parsa KV. Deubiquitinase USP12 promotes LPS induced macrophage responses through inhibition of IkappaBalpha. Biochem Biophys Res Commun. 2017;483:69–74.
Liu J, Jin LC, Chen XJ, Yuan YK, Zuo YB, Miao Y, et al. USP12 translocation maintains interferon antiviral efficacy by inhibiting CBP acetyltransferase activity. PLoS Pathog. 2020;16:e1008215.
Aron R, Pellegrini P, Green EW, Maddison DC, Opoku-Nsiah K, Oliveira AO, et al. Deubiquitinase Usp12 functions noncatalytically to induce autophagy and confer neuroprotection in models of Huntington’s disease. Nat Commun. 2018;9:3191.
Chauhan D, Tian Z, Nicholson B, Kumar KG, Zhou B, Carrasco R, et al. A small molecule inhibitor of ubiquitin-specific protease-7 induces apoptosis in multiple myeloma cells and overcomes bortezomib resistance. Cancer Cell. 2012;22:345–58.
Tian Z, D’Arcy P, Wang X, Ray A, Tai YT, Hu YG, et al. A novel small molecule inhibitor of deubiquitylating enzyme USP14 and UCHL5 induces apoptosis in multiple myeloma and overcomes bortezomib resistance. Blood. 2014;123:706–16.
Bignell GR, Warren W, Seal S, Takahashi M, Rapley E, Barfoot R, et al. Identification of the familial cylindromatosis tumour-suppressor gene. Nat Genet. 2000;25:160–5.
Hussain S, Bedekovics T, Chesi M, Bergsagel PL, Galardy PJ. UCHL1 is a biomarker of aggressive multiple myeloma required for disease progression. Oncotarget. 2015;6:40704–18.
Sridhar S, Botbol Y, Macian F, Cuervo AM. Autophagy and disease: always two sides to a problem. J Pathol. 2012;226:255–73.
Mah LY, Ryan KM. Autophagy and cancer. Cold Spring Harb Perspect Biol. 2012;4:a008821.
Arnold J, Murera D, Arbogast F, Fauny JD, Muller S, Gros F. Autophagy is dispensable for B-cell development but essential for humoral autoimmune responses. Cell Death Differ. 2016;23:853–64.
Zhuang Y, Jin ZC, Yao H, Ji O, Yang R, Dong W, et al. Targeting autophagy in multiple myeloma. Leuk Res. 2017;59:97–104.
Jacomin AC, Bescond A, Soleilhac E, Gallet B, Schoehn G, Fauvarque MO, et al. The deubiquitinating enzyme UBPY is required for lysosomal biogenesis and productive autophagy in drosophila. PLoS ONE. 2015;10:e0143078.
Nakada S, Tai I, Panier S, Al-Hakim A, Iemura SI, Juang YC, et al. Non-canonical inhibition of DNA damage-dependent ubiquitination by OTUB1. Nature. 2010;466:941–6.
Hanna J, Hathaway NA, Tone Y, Crosas B, Elsasser S, Kirkpatrick DS, et al. Deubiquitinating enzyme Ubp6 functions noncatalytically to delay proteasomal degradation. Cell. 2006;127:99–111.
Yuan S, Liu ZP, Xu ZR, Liu J, Zhang J. High mobility group box 1 (HMGB1): a pivotal regulator of hematopoietic malignancies. J Hematol Oncol. 2020;13:91.
D’Eliseo D, Renzo LD, Santoni A, Velotti F. Docosahexaenoic acid (DHA) promotes immunogenic apoptosis in human multiple myeloma cells, induces autophagy and inhibits STAT3 in both tumor and dendritic cells. Genes Cancer. 2017;8:426–37.
Nomura S, Ito T, Yoshimura H, Hotta M, Nakanishi T, Fujita S, et al. Evaluation of thrombosis-related biomarkers before and after therapy in patients with multiple myeloma. J Blood Med. 2018;9:1–7.
Inoue Y, Saito T, Tsuruoka Y, Sato K, Nishio Y, Suzuki Y, et al. Recombinant thrombomodulin improved Stevens-Johnson syndrome with high serum high-mobility group-B1 DNA-binding protein induced by lenalidomide administered to treat multiple myeloma. Thromb Res. 2013;132:493–4.
Gao D, Lv AE, Li HP, Han DH, Zhang YP. LncRNA MALAT-1 elevates HMGB1 to promote autophagy resulting in inhibition of tumor cell apoptosis in multiple myeloma. J Cell Biochem. 2017;118:3341–8.
Guo X, He DH, Zhang EF, Chen J, Chen QX, Li Y, et al. HMGB1 knockdown increases MM cell vulnerability by regulating autophagy and DNA damage repair. J Exp Clin Cancer Res. 2018;37:205.
Kane RC, Bross PF, Farrell AT, Pazdur R. Velcade: U.S. FDA approval for the treatment of multiple myeloma progressing on prior therapy. Oncologist. 2003;8:508–13.
Tang D, Kang R, Livesey KM, Cheh CW, Farkas A, Loughran P, et al. Endogenous HMGB1 regulates autophagy. J Cell Biol. 2010;190:881–92.
Boutouja F, Brinkmeier R, Mastalski T, Magraoui FE, Platta HW. Regulation of the tumor-suppressor BECLIN 1 by distinct ubiquitination cascades. Int J Mol Sci. 2017;18:2541.
Xia J, He YJ, Meng B, Chen SL, Zhang JY, Wu X, et al. NEK2 induces autophagy-mediated bortezomib resistance by stabilizing Beclin-1 in multiple myeloma. Mol Oncol. 2020;14:763–78.
Salminen A, Kaarniranta K, Kauppinen A, Ojala J, Haapasalo A, Soininen H, et al. Impaired autophagy and APP processing in Alzheimer’s disease: the potential role of Beclin 1 interactome. Prog Neurobiol. 2013;106-107:33–54.
Funding
This study was supported by grants from the National Key Research and Development Program of China (2018YFA0107800), the National Natural Science Foundation of China (Nos. 21890744, 81672760, 82003286, 81970195, 81920108004, 81772496 and 82073099), the Natural Science Foundation of Hunan Province (No. 2021JJ40054, 2019JJ40391), the China Postdoctoral Science Foundation (2020M672474, 2021T140195 and 2018M630896), the Hunan Provincial Key Research and Development Plan (2018SK2128), the Changsha Municipal Natural Science Foundation (kq2014041, kq2014042 and kq2001012).
Author information
Authors and Affiliations
Contributions
HL and MR contributed equally to this work. JL, BZ, MY and SS designed the research, HL, MR, LL, WC, BH, YL, XX and HW performed the experiments, analyzed and interpreted the data; BZ, SS, MY and JL analyzed and interpreted the data; MR, HL, JL and MY drafted and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Li, H., Roy, M., Liang, L. et al. Deubiquitylase USP12 induces pro-survival autophagy and bortezomib resistance in multiple myeloma by stabilizing HMGB1. Oncogene 41, 1298–1308 (2022). https://doi.org/10.1038/s41388-021-02167-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-021-02167-9
This article is cited by
-
Spotlights on ubiquitin-specific protease 12 (USP12) in diseases: from multifaceted roles to pathophysiological mechanisms
Journal of Translational Medicine (2023)
-
N6-methyladenosine-modified USP13 induces pro-survival autophagy and imatinib resistance via regulating the stabilization of autophagy-related protein 5 in gastrointestinal stromal tumors
Cell Death & Differentiation (2023)
-
USP32 deubiquitinase: cellular functions, regulatory mechanisms, and potential as a cancer therapy target
Cell Death Discovery (2023)
-
USP13 regulates HMGB1 stability and secretion through its deubiquitinase activity
Molecular Medicine (2022)