Abstract
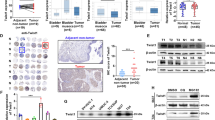
Bladder cancer (BLCA) is the most common malignant tumor of the urinary system and is characterized by high metastatic rates and poor prognosis. The expression of tight junction protein 1 (TJP1) is associated with bladder cancer invasion; however, the mechanism by which TJP1 affects vasculature remodeling remains unknown. In this study, we found that TJP1 expression correlated with tumor angiogenesis and poor overall survival in clinical samples. Furthermore, TJP1 overexpression promoted tumor angiogenesis in BLCA cells and stimulated recruitment of macrophages to tumors by upregulating CCL2 expression. Mechanistically, TJP1 interacted with TWIST1 and enhanced the transcriptional activity of CCL2. The impairment of tumor angiogenesis caused by knockdown of TJP1 was dramatically rescued by overexpression of TWIST1. Furthermore, TJP1 recruited USP2, which deubiquitinated TWIST1, thereby protecting TWIST1 from proteasome-mediated protein degradation. In conclusion, our results suggest that TJP1 controls angiogenesis in BLCA via TWIST1-dependent regulation of CCL2. We demonstrate that TJP1 functions as a scaffold for the interaction between USP2 and TWIST1 and this may provide potential therapeutic targets in bladder cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Tran L, Xiao JF, Agarwal N, Duex JE, Theodorescu D. Advances in bladder cancer biology and therapy. Nat Rev Cancer. 2021;21:104–21.
Witjes JA, Compérat E, Cowan NC, De Santis M, Gakis G, Lebret T, et al. EAU guidelines on muscle-invasive and metastatic bladder cancer: summary of the 2013 guidelines. Eur Urol. 2014;65:778–92.
Naito S, Algaba F, Babjuk M, Bryan RT, Sun YH, Valiquette L, et al. The Clinical Research Office of the Endourological Society (CROES) multicentre randomised trial of narrow band imaging-assisted transurethral resection of bladder tumour (TURBT) versus conventional white light imaging-assisted turbt in primary non-muscle-invasive bladder cancer patients: trial protocol and 1-year results. Eur Urol. 2016;70:506–15.
Zhou L, Chang Y, Xu L, Hoang ST, Liu Z, Fu Q, et al. Prognostic value of vascular mimicry in patients with urothelial carcinoma of the bladder after radical cystectomy. Oncotarget. 2016;7:76214–23.
Li T, Kang G, Wang T, Huang H. Tumor angiogenesis and anti-angiogenic gene therapy for cancer. Oncol Lett. 2018;16:687–702.
Flamme I, Frölich T, Risau W. Molecular mechanisms of vasculogenesis and embryonic angiogenesis. J Cell Physiol. 1997;173:206–10.
Risau W. Mechanisms of angiogenesis. Nature. 1997;386:671–4.
Shibuya M. Vascular endothelial growth factor (VEGF) and its receptor (VEGFR) signaling in angiogenesis: a crucial target for anti- and pro-angiogenic therapies. Genes Cancer. 2011;2:1097–105.
Zhao Y, Adjei AA. Targeting angiogenesis in cancer therapy: moving beyond vascular endothelial growth factor. Oncologist. 2015;20:660–73.
Black PC, Dinney CP. Bladder cancer angiogenesis and metastasis-translation from murine model to clinical trial. Cancer metastasis Rev. 2007;26:623–34.
Beutel O, Maraspini R, Pombo-García K, Martin-Lemaitre C, Honigmann A. Phase separation of zonula occludens proteins drives formation of tight junctions. Cell. 2019;179:923–936. e911
Bauer H, Zweimueller-Mayer J, Steinbacher P, Lametschwandtner A, Bauer HC. The dual role of zonula occludens (ZO) proteins. J biomedicine Biotechnol. 2010;2010:402593.
Laakkonen JP, Lappalainen JP, Theelen TL, Toivanen PI, Nieminen T, Jauhiainen S, et al. Differential regulation of angiogenic cellular processes and claudin-5 by histamine and VEGF via PI3K-signaling, transcription factor SNAI2 and interleukin-8. Angiogenesis. 2017;20:109–24.
Liu X, Dreffs A, Díaz-Coránguez M, Runkle EA, Gardner TW, Chiodo VA, et al. Occludin S490 phosphorylation regulates vascular endothelial growth factor-induced retinal neovascularization. Am J Pathol. 2016;186:2486–99.
Martin TA, Mansel RE, Jiang WG. Antagonistic effect of NK4 on HGF/SF induced changes in the transendothelial resistance (TER) and paracellular permeability of human vascular endothelial cells. J Cell Physiol. 2002;192:268–75.
Hsu YL, Hung JY, Chang WA, Lin YS, Pan YC, Tsai PH, et al. Hypoxic lung cancer-secreted exosomal miR-23a increased angiogenesis and vascular permeability by targeting prolyl hydroxylase and tight junction protein ZO-1. Oncogene. 2017;36:4929–42.
Lesage J, Suarez-Carmona M, Neyrinck-Leglantier D, Grelet S, Blacher S, Hunziker W, et al. Zonula occludens-1/NF-kappaB/CXCL8: a new regulatory axis for tumor angiogenesis. FASEB J. 2017;31:1678–88.
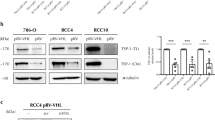
Tsai KW, Kuo WT, Jeng SY. Tight junction protein 1 dysfunction contributes to cell motility in bladder cancer. Anticancer Res. 2018;38:4607–15.
Zhao Z, Rahman MA, Chen ZG, Shin DM. Multiple biological functions of Twist1 in various cancers. Oncotarget. 2017;8:20380–93.
Yang MH, Wu MZ, Chiou SH, Chen PM, Chang SY, Liu CJ, et al. Direct regulation of TWIST by HIF-1alpha promotes metastasis. Nat Cell Biol. 2008;10:295–305.
Sun T, Sun BC, Zhao XL, Zhao N, Dong XY, Che N, et al. Promotion of tumor cell metastasis and vasculogenic mimicry by way of transcription coactivation by Bcl-2 and Twist1: a study of hepatocellular carcinoma. Hepatology. 2011;54:1690–706.
Hu L, Roth JM, Brooks P, Ibrahim S, Karpatkin S. Twist is required for thrombin-induced tumor angiogenesis and growth. Cancer Res. 2008;68:4296–302.
Wagenblast E, Soto M, Gutiérrez-Ángel S, Hartl CA, Gable AL, Maceli AR, et al. A model of breast cancer heterogeneity reveals vascular mimicry as a driver of metastasis. Nature. 2015;520:358–62.
Weis SM, Cheresh DA. Tumor angiogenesis: molecular pathways and therapeutic targets. Nat Med. 2011;17:1359–70.
Rao Q, Chen Y, Yeh CR, Ding J, Li L, Chang C, et al. Recruited mast cells in the tumor microenvironment enhance bladder cancer metastasis via modulation of ERbeta/CCL2/CCR2 EMT/MMP9 signals. Oncotarget. 2016;7:7842–55.
Low-Marchelli JM, Ardi VC, Vizcarra EA, van Rooijen N, Quigley JP, Yang J. Twist1 induces CCL2 and recruits macrophages to promote angiogenesis. Cancer Res. 2013;73:662–71.
Yang J, Mani SA, Donaher JL, Ramaswamy S, Itzykson RA, Come C, et al. Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell. 2004;117:927–39.
Singh S, Sadanandam A, Singh RK. Chemokines in tumor angiogenesis and metastasis. Cancer metastasis Rev. 2007;26:453–67.
Neufeld G, Kessler O. Pro-angiogenic cytokines and their role in tumor angiogenesis. Cancer metastasis Rev. 2006;25:373–85.
Wang S, Xu M, Li F, Wang X, Bower KA, Frank JA, et al. Ethanol promotes mammary tumor growth and angiogenesis: the involvement of chemoattractant factor MCP-1. Breast cancer Res Treat. 2012;133:1037–48.
Koga M, Kai H, Egami K, Murohara T, Ikeda A, Yasuoka S, et al. Mutant MCP-1 therapy inhibits tumor angiogenesis and growth of malignant melanoma in mice. Biochemical biophysical Res Commun. 2008;365:279–84.
Anghelina M, Krishnan P, Moldovan L, Moldovan NI. Monocytes and macrophages form branched cell columns in matrigel: implications for a role in neovascularization. Stem cells Dev. 2004;13:665–76.
Lewis CE, Pollard JW. Distinct role of macrophages in different tumor microenvironments. Cancer Res. 2006;66:605–12.
Henry N, Eeckhout Y, van Lamsweerde AL, Vaes G. Co-operation between metastatic tumor cells and macrophages in the degradation of basement membrane (type IV) collagen. FEBS Lett. 1983;161:243–6.
de Iongh RU, Wederell E, Lovicu FJ, McAvoy JW. Transforming growth factor-beta-induced epithelial-mesenchymal transition in the lens: a model for cataract formation. Cells, tissues, organs. 2005;179:43–55.
Antonetti DA, Barber AJ, Hollinger LA, Wolpert EB, Gardner TW. Vascular endothelial growth factor induces rapid phosphorylation of tight junction proteins occludin and zonula occluden 1. A potential mechanism for vascular permeability in diabetic retinopathy and tumors. J Biol Chem. 1999;274:23463–7.
Tornavaca O, Chia M, Dufton N, Almagro LO, Conway DE, Randi AM, et al. ZO-1 controls endothelial adherens junctions, cell-cell tension, angiogenesis, and barrier formation. J cell Biol. 2015;208:821–38.
Ribatti D, Nico B, Crivellato E, Vacca A. The structure of the vascular network of tumors. Cancer Lett. 2007;248:18–23.
Dhanasekaran R, Baylot V, Kim M, Kuruvilla S, Bellovin DI, Adeniji N, et al. MYC and Twist1 cooperate to drive metastasis by eliciting crosstalk between cancer and innate immunity. eLife. 2020;9:e50731
Lee KW, Yeo SY, Sung CO, Kim SH. Twist1 is a key regulator of cancer-associated fibroblasts. Cancer Res. 2015;75:73–85.
Roberts CM, Shahin SA, Loeza J, Dellinger TH, Williams JC, Glackin CA. Disruption of TWIST1-RELA binding by mutation and competitive inhibition to validate the TWIST1 WR domain as a therapeutic target. BMC Cancer. 2017;17:184.
Yang WH, Su YH, Hsu WH, Wang CC, Arbiser JL, Yang MH. Imipramine blue halts head and neck cancer invasion through promoting F-box and leucine-rich repeat protein 14-mediated Twist1 degradation. Oncogene. 2016;35:2287–98.
Li CW, Xia W, Lim SO, Hsu JL, Huo L, Wu Y, et al. AKT1 inhibits epithelial-to-mesenchymal transition in breast cancer through phosphorylation-dependent twist1 degradation. Cancer Res. 2016;76:1451–62.
Li F, Hu Q, He T, Xu J, Yi Y, Xie S, et al. The deubiquitinase USP4 stabilizes twist1 protein to promote lung cancer cell stemness. Cancers (Basel). 2020;12:1582
He J, Lee HJ, Saha S, Ruan D, Guo H, Chan CH. Inhibition of USP2 eliminates cancer stem cells and enhances TNBC responsiveness to chemotherapy. Cell Death Dis. 2019;10:285.
Meng J, Chen S, Lei YY, Han JX, Zhong WL, Wang XR, et al. Hsp90beta promotes aggressive vasculogenic mimicry via epithelial-mesenchymal transition in hepatocellular carcinoma. Oncogene. 2019;38:228–43.
Wang X, Wu Q, Xu B, Wang P, Fan W, Cai Y, et al. MiR-124 exerts tumor suppressive functions on the cell proliferation, motility and angiogenesis of bladder cancer by fine-tuning UHRF1. FEBS J. 2015;282:4376–88.
Yang Z, Chen J, Xie H, Liu T, Chen Y, Ma Z, et al. Androgen receptor suppresses prostate cancer metastasis but promotes bladder cancer metastasis via differentially altering miRNA525-5p/SLPI-mediated vasculogenic mimicry formation. Cancer Lett. 2020;473:118–29.
Ponce ML. Tube formation: an in vitro matrigel angiogenesis assay. Methods Mol Biol (Clifton, NJ). 2009;467:183–8.
Acknowledgements
We thank Prof. Kang (Sun Yat-sen University Cancer Center) for kindly providing T24 and 5637 bladder cancer cells and the faculty (Fifth Affiliated Hospital of Sun Yat-sen University) for collecting the clinical samples. This work was supported in part by the National Natural Science Foundation of China (grant no. 81970191; 32170789; 3210050130); supported by Guangdong Natural Science Foundation of China (grant no. 2018A0303130090); supported by Shenzhen Science and Technology Innovation Commission, China (JCYJ20190807160209294, JCYJ20190807160813467).
Author information
Authors and Affiliations
Contributions
XL, XS, and YL performed most of the cell experiments and manuscript writing. ZD and SC performed animal experiments. YS, SC, and XW performed database analysis. LZ, LQ, XZ, and LS reviewed and suggested the manuscript. TL and PY collected the clinical samples, completed the IHC assay, and prepared the case report. YY, LL, XL, and WZ gave instructions of the RNA sequence analysis. XZ and ML conceived and designed the study. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, XQ., Shao, XR., Liu, Y. et al. Tight junction protein 1 promotes vasculature remodeling via regulating USP2/TWIST1 in bladder cancer. Oncogene 41, 502–514 (2022). https://doi.org/10.1038/s41388-021-02112-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-021-02112-w