Abstract
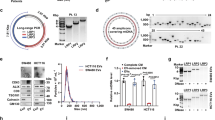
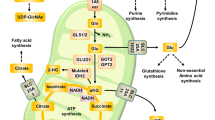
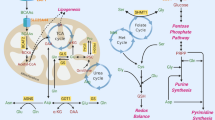
Metastasis is the leading cause of colorectal cancer (CRC)-induced death. However, the underlying molecular mechanisms of CRC metastasis are poorly understood. Metabolic reprogramming is an intrinsic feature of cancer, which have complicated effects on cancer metastasis. Here, we find that a novel metastasis-related protein, cell migration-inducing and hyaluronan-binding protein (CEMIP), can act as a novel adaptor protein of O-GlcNAc transferase (OGT) to promote CRC metastasis through glutamine metabolic reprogramming. Mechanistically, CEMIP interacts with OGT and β-catenin, which leads to elevated O-GlcNAcylation of β-catenin and enhanced β-catenin nuclear translocation from cytomembrane. Furthermore, accumulated β-catenin in nucleus enhances the transcription of CEMIP to reciprocally regulate β-catenin and contributes to over-expression of glutaminase 1 and glutamine transporters (SLC1A5 and SLC38A2). Combinational inhibition of CEMIP and glutamine metabolism could dramatically attenuate the metastasis of CRC in vivo. Collectively, this study reveals the importance of glutamine metabolic reprogramming in CEMIP-induced CRC metastasis, indicating the great potential of CEMIP and glutamine metabolism for CRC metastasis prevention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7–30.
Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB. Colorectal cancer. Lancet. 2019;394:1467–80.
Yoshida H, Nagaoka A, Kusaka-Kikushima A, Tobiishi M, Kawabata K, Sayo T, et al. KIAA1199, a deafness gene of unknown function, is a new hyaluronan binding protein involved in hyaluronan depolymerization. Proc Natl Acad Sci USA. 2013;110:5612–7.
Tiwari A, Schneider M, Fiorino A, Haider R, Okoniewski MJ, Roschitzki B, et al. Early insights into the function of KIAA1199, a markedly overexpressed protein in human colorectal tumors. PLoS One. 2013;8:e69473.
Zhao L, Zhang DJ, Shen Q, Jin M, Lin ZY, Ma H, et al. KIAA1199 promotes metastasis of colorectal cancer cells via microtubule destabilization regulated by a PP2A/stathmin pathway. Oncogene. 2019;38:935–49.
Zhang D, Zhao L, Shen Q, Lv Q, Jin M, Ma H, et al. Down-regulation of KIAA1199/CEMIP by miR-216a suppresses tumor invasion and metastasis in colorectal cancer. Int J Cancer. 2017;140:2298–309.
Evensen NA, Kuscu C, Nguyen HL, Zarrabi K, Dufour A, Kadam P, et al. Unraveling the role of KIAA1199, a Novel Endoplasmic Reticulum Protein, In Cancer Cell Migration. J Natl Cancer Inst. 2013;105:1402–16.
Shostak K, Zhang X, Hubert P, Goktuna SI, Jiang ZS, Klevernic I, et al. NF-kappa B-induced KIAA1199 promotes survival through EGFR signalling. Nat Commun. 2014;5:5232.
Tang ZF, Li CW, Kang BX, Gao G, Li C, Zhang ZM. GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017;45:W98–W102.
Schild T, Low V, Blenis J, Gomes AP. Unique metabolic adaptations dictate distal organ-specific metastatic colonization. Cancer Cell. 2018;33:347–54.
Bergers G, Fendt SM. The metabolism of cancer cells during metastasis. Nat Rev Cancer. 2021;21:162–80.
Wei Q, Qian Y, Yu J, Wong CC. Metabolic rewiring in the promotion of cancer metastasis: mechanisms and therapeutic implications. Oncogene. 2020;39:6139–56.
Hensley CT, Wasti AT, DeBerardinis RJ. Glutamine and cancer: cell biology, physiology, and clinical opportunities. J Clin Investig. 2013;123:3678–84.
Altman BJ, Stine ZE, Dang CV. From Krebs to clinic: glutamine metabolism to cancer therapy. Nat Rev Cancer. 2016;16:619–34.
Sharma NS, Gupta VK, Garrido VT, Hadad R, Durden BC, Kesh K, et al. Targeting tumor-intrinsic hexosamine biosynthesis sensitizes pancreatic cancer to anti-PD1 therapy. J Clin Invest. 2020;130:451–65.
Yoo HC, Park SJ, Nam M, Kang J, Kim K, Yeo JH, et al. A variant of SLC1A5 is a mitochondrial glutamine transporter for metabolic reprogramming in cancer cells. Cell Metab. 2020;31:267–83.e212.
Broer A, Rahimi F, Broer S. Deletion of amino acid transporter ASCT2 (SLC1A5) reveals an essential role for transporters SNAT1 (SLC38A1) and SNAT2 (SLC38A2) to sustain glutaminolysis in cancer cells. J Biol Chem. 2016;291:13194–205.
Morotti M, Zois CE, El-Ansari R, Craze ML, Rakha EA, Fan SJ, et al. Increased expression of glutamine transporter SNAT2/SLC38A2 promotes glutamine dependence and oxidative stress resistance, and is associated with worse prognosis in triple-negative breast cancer. Br J Cancer. 2021;124:494–505.
Schulte ML, Fu A, Zhao P, Li J, Geng L, Smith ST, et al. Pharmacological blockade of ASCT2-dependent glutamine transport leads to antitumor efficacy in preclinical models. Nat Med. 2018;24:194–202.
Ding MC, Bu X, Li ZH, Xu HK, Feng L, Hu JB, et al. NDRG2 ablation reprograms metastatic cancer cells towards glutamine dependence via the induction of ASCT2. Int J Biol Sci. 2020;16:3100–15.
Gao P, Tchernyshyov I, Chang TC, Lee YS, Kita K, Ochi T, et al. c-Myc suppression of miR-23a/b enhances mitochondrial glutaminase expression and glutamine metabolism. Nature. 2009;458:762–U100.
Mafra ACP, Dias SMG. Several faces of glutaminase regulation in cells. Cancer Res. 2019;79:1302–4.
Shen YA, Hong JX, Asaka R, Asaka S, Hsu FC, Rahmanto YS, et al. Inhibition of the MYC-regulated glutaminase metabolic axis is an effective synthetic lethal approach for treating chemoresistant ovarian cancers. Cancer Res. 2020;80:4514–26.
Chen C, Lu YC, Liu J, Li LS, Zhao N, Lin BY. Genome-wide ChIP-seq analysis of TCF4 binding regions in colorectal cancer cells. International Journal Of. Clin Exp Med. 2014;7:4253–U1510.
Duong HQ, Nemazanyy I, Rambow F, Tang SC, Delaunay S, Tharun L, et al. The endosomal protein CEMIP links WNT signaling to MEK1-ERK1/2 activation in selumetinib-resistant intestinal organoids. Cancer Res. 2018;78:4533–48.
Sabates-Bellver J, Van der Flier LG, de Palo M, Cattaneo E, Maake C, Rehrauer H, et al. Transcriptome profile of human colorectal adenomas. Mol Cancer Res. 2007;5:1263–75.
Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B, et al. Activation of β-catenin–Tcf signaling in colon cancer by mutations in β-catenin or APC. Science. 1997;275:1787–90.
Behrens J. Cadherins and catenins: role in signal transduction and tumor progression. Cancer Metastasis Rev. 1999;18:15–30.
Ha JR, Hao L, Venkateswaran G, Huang YH, Garcia E, Persad S. beta-catenin is O-GlcNAc glycosylated at Serine 23: implications for beta-catenin’s subcellular localization and transactivator function. Exp Cell Res. 2014;321:153–66.
Sayat R, Leber B, Grubac V, Wiltshire L, Persad S. O-GlcNAc-glycosylation of beta-catenin regulates its nuclear localization and transcriptional activity. Exp Cell Res. 2008;314:2774–87.
Kasprowicz A, Spriet C, Terryn C, Rigolot V, Hardiville S, Alteen MG, et al. Exploring the potential of beta-catenin O-GlcNAcylation by using fluorescence-based engineering and imaging. Molecules. 2020;25:4501.
Gao S, Miao Y, Liu Y, Liu X, Fan X, Lin Y, et al. Reciprocal regulation between O-GlcNAcylation and beta-catenin facilitates cell viability and inhibits apoptosis in liver cancer. DNA Cell Biol. 2019;38:286–96.
Olivier-Van Stichelen S, Guinez C, Mir AM, Perez-Cervera Y, Liu C, Michalski JC, et al. The hexosamine biosynthetic pathway and O-GlcNAcylation drive the expression of beta-catenin and cell proliferation. Am J Physiol Endocrinol Metab. 2012;302:E417–24.
Naka Y, Okada T, Nakagawa T, Kobayashi E, Kawasaki Y, Tanaka Y, et al. Enhancement of O-linked N-acetylglucosamine modification promotes metastasis in patients with colorectal cancer and concurrent type 2 diabetes mellitus. Oncol Let.t 2020;20:1171–8.
Zhou L, Luo M, Cheng LJ, Li RN, Liu B, Linghu H. Glutamine-fructose-6-phosphate transaminase 2 (GFPT2) promotes the EMT of serous ovarian cancer by activating the hexosamine biosynthetic pathway to increase the nuclear location of beta-catenin. Pathol Res Pract. 2019;215:152681.
Hart GW, Housley MP, Slawson C. Cycling of O-linked beta-N-acetylglucosamine on nucleocytoplasmic proteins. Nature. 2007;446:1017–22.
Long GV, Dummer R, Hamid O, Gajewski TF, Caglevic C, Dalle S, et al. Epacadostat plus pembrolizumab versus placebo plus pembrolizumab in patients with unresectable or metastatic melanoma (ECHO-301/KEYNOTE-252): a phase 3, randomised, double-blind study. Lancet Oncol. 2019;20:1083–97.
Bienz M, Clevers H. Linking colorectal cancer to Wnt signaling. Cell. 2000;103:311–20.
Zhang Y, Wang X. Targeting the Wnt/beta-catenin signaling pathway in cancer. J Hematol Oncol. 2020;13:165.
Birkenkamp-Demtroder K, Maghnouj A, Mansilla F, Thorsen K, Andersen CL, Oster B, et al. Repression of KIAA1199 attenuates Wnt-signalling and decreases the proliferation of colon cancer cells. Br J Cancer. 2011;105:552–61.
Liang G, Fang X, Yang Y, Song Y. Silencing of CEMIP suppresses Wnt/beta-catenin/Snail signaling transduction and inhibits EMT program of colorectal cancer cells. Acta Histochem. 2018;120:56–63.
Cheung WD, Hart GW. AMP-activated protein kinase and p38 MAPK activate O-GlcNAcylation of neuronal proteins during glucose deprivation. J Biol Chem. 2008;283:13009–20.
Maclean KH, Keller UB, Rodriguez-Galindo C, Nilsson JA, Cleveland JL. c-Myc augments gamma irradiation-induced apoptosis by suppressing Bcl-XL. Mol Cell Biol. 2003;23:7256–70.
Zhang P, Song Y, Sun Y, Li X, Chen L, Yang L, et al. AMPK/GSK3beta/beta-catenin cascade-triggered overexpression of CEMIP promotes migration and invasion in anoikis-resistant prostate cancer cells by enhancing metabolic reprogramming. FASEB J. 2018;32:3924–35.
Coller HA, Grandori C, Tamayo P, Colbert T, Lander ES, Eisenman RN, et al. Expression analysis with oligonucleotide microarrays reveals that MYC regulates genes involved in growth, cell cycle, signaling, and adhesion. Proc Natl Acad Sci USA. 2000;97:3260–5.
Acknowledgements
This work is supported by the National Natural Science Foundation of China (81874061 to TZ, 81702392 to LZ, and 81903103 to DZ).
Author information
Authors and Affiliations
Contributions
Conception and design: QLH, LZ, and TZ. Acquisition of data: QLH, BYZ, and GJX. Analysis and interpretation of data: QLH, BYZ, GJX, LQW, and HHW. Writing, review, and/or revision of the manuscript: QLH, BYZ, GJX, LQW, HHW, ZYL, DDY, JHR, DJZ, LZ, and TZ.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hua, Q., Zhang, B., Xu, G. et al. CEMIP, a novel adaptor protein of OGT, promotes colorectal cancer metastasis through glutamine metabolic reprogramming via reciprocal regulation of β-catenin. Oncogene 40, 6443–6455 (2021). https://doi.org/10.1038/s41388-021-02023-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-021-02023-w
This article is cited by
-
Elucidating immunological characteristics of the adenoma-carcinoma sequence in colorectal cancer patients in South Korea using a bioinformatics approach
Scientific Reports (2024)
-
A Prognostic Methylation-Driven Two-Gene Signature in Medulloblastoma
Journal of Molecular Neuroscience (2024)
-
O‐GlcNAcylation of CSNK2A1 by OGT is Involved in the Progression of Colorectal Cancer
Molecular Biotechnology (2024)
-
Metabolic reprogramming in colorectal cancer: regulatory networks and therapy
Cell & Bioscience (2023)
-
Construction and validation of a prognostic model for colon adenocarcinoma based on bile acid metabolism-related genes
Scientific Reports (2023)