Abstract
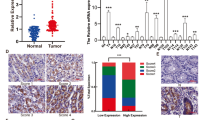
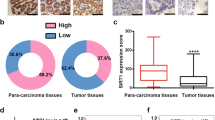
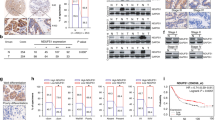
Asporin (ASPN) presents in the tumor microenvironment and exhibits a cancer-promoting effect as a stroma protein. Even though ASPN has already been observed inside cancer cells, the functions of intracellular ASPN and its underlying mechanisms remain unknown. Here we reported that ASPN was upregulated in different stages of gastric cancer (GC), and associated with a poor prognosis. Moreover, we found that ASPN markedly inhibited GC cell apoptosis and promoted cell growth in vitro and in vivo. Further mechanism investigations revealed that ASPN directly binding to lymphoid enhancer-binding factor 1 (LEF1) and promoted LEF1-mediated gene transcription independent of β-catenin, the classic co-factor in the Wnt/LEF1 pathway. We also demonstrated that ASPN selectively facilitated LEF1 binding to and activating the promoters of PTGS2, IL6, and WISP1 to promote their transcription. The suppression of cell apoptosis by ASPN overexpression could be attenuated by LEF1 knockdown or 100 µM aspirin (PTGS2 inhibitor), and siASPN mediated apoptosis could be rescued by LEF1 ectopic expression or adding recombinant IL6. Therefore, we concluded that ASPN repressed GC cell apoptosis via activating LEF1-mediated gene transcription independent of β-catenin, which could serve as a potential prognostic biomarker in GC patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All the data needed to evaluate the conclusions made in the paper are presented in the paper. Additional data related to this paper may be requested from the authors.
References
The Cancer Genome Atlas Research Network. Cancer Genome Atlas Research Network; Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513:202–9.
Song Y, Wang Y, Tong C, Xi H, Zhao X, Wang Y, et al. A unified model of the hierarchical and stochastic theories of gastric cancer. Brit J Cancer. 2017;116:973.
Lordick F, Allum W, Carneiro F, Mitry E, Tabernero J, Tan P, et al. Unmet needs and challenges in gastric cancer: the way forward. Cancer Treat Rev. 2014;40:692–700.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7–30.
Badgwell B, Blum M, Estrella J, Ajani J. Personalised therapy for localised gastric and gastro-oesophageal adenocarcinoma. Lancet Oncol. 2016;17:1628–9.
Van CE, Sagaert X, Topal B, Haustermans K, Prenen H. Gastric cancer. Lancet. 2016;388:2654–64.
Lorenzo P, Aspberg A, Onnerfjord P, Bayliss MT, Neame PJ, Heinegard D. Identification and characterization of asporin. a novel member of the leucine-rich repeat protein family closely related to decorin and biglycan. J Biol Chem. 2001;276:12201–11.
Ikegawa S. Expression, regulation and function of asporin, a susceptibility gene in common bone and joint diseases. Curr Med Chem. 2008;15:724–8.
Clark HF, Gurney AL, Abaya E, Baker K, Baldwin D, Brush J, et al. The secreted protein discovery initiative (SPDI), a large-scale effort to identify novel human secreted and transmembrane proteins: a bioinformatics assessment. Genome Res. 2003;13:2265–70.
Kizawa H, Kou I, Iida A, Sudo A, Miyamoto Y, Fukuda A, et al. An aspartic acid repeat polymorphism in asporin inhibits chondrogenesis and increases susceptibility to osteoarthritis. Nat Genet. 2005;37:138–44.
Rodriguez-Lopez J, Pombo-Suarez M, Liz M, Gomez-Reino JJ, Gonzalez A. Lack of association of a variable number of aspartic acid residues in the asporin gene with osteoarthritis susceptibility: case-control studies in Spanish Caucasians. Arthritis Res Ther. 2006;8:R55.
Simkova D, Kharaishvili G, Slabakova E, Murrayc PG, Bouchala J. Glycoprotein asporin as a novel player in tumour microenvironment and cancer progression. Biomed Pap Med Fac Univ Palacky Olomouc. 2016;160:467–73.
Castellana B, Escuin D, Peiró G, Garcia-Valdecasas B, Vázquez T, Pons C. ASPN and GJB2 are implicated in the mechanisms of invasion of ductal breast carcinomas. J Cancer. 2012;3:1783.
Maris P, Blomme A, Palacios AP, Costanza B, Bellahcène A, Bianchi E, et al. Asporin is a fibroblast-derived TGF-β1 inhibitor and a tumor suppressor associated with good prognosis in breast cancer. PLoS Med. 2015;12:e1001871.
Hurley PJ, Sundi D, Shinder B, Simons BW, Hughes RM, Hughes RM, et al. Germline variants in asporin vary by race, modulate the tumor microenvironment, and are differentially associated with metastatic prostate cancer. Clin Cancer Res. 2016;22:448–58.
Wang L, Wu H, Wang L, Zhang H, Lu J, Liang Z, et al. Asporin promotes pancreatic cancer cell invasion and migration by regulating the epithelial-to-mesenchymal transition (EMT) through both autocrine and paracrine mechanisms. Cancer Lett. 2017;398:24–36.
Huo W, Jing XQ, Cheng X, He YG, Hu L, Wu HX, et al. Asporin enhances colorectal cancer metastasis through activating the EGFR/src/cortactin signaling pathway. Oncotarget. 2016;7:73402–13.
Hrckulak D, Kolar M, Strnad H, Korinek V. TCF/LEF transcription factors: an update from the internet resources. Cancers. 2016;8:70.
Longo KA, Kennell JA, Ochocinska MJ, Ross SE, Wright WS, MacDougald OA. Wnt signaling protects 3T3-L1 preadipocytes from apoptosis through induction of insulin-like growth factors. J Biol Chem. 2002;277:38239–44.
Moon RT, Kohn AD, De Ferrari GV, Kaykas A. WNT and β-catenin signalling: diseases and therapies. Nat Rev Genet. 2004;5:691–704.
Janna W, Katja R, Elham BH, Constanze L, Gilles R, Vivien F, et al. Loss of the nuclear Wnt pathway effector TCF7L2 promotes migration and invasion of human colorectal cancer cells. Oncogene. 2020;39:3893–909.
Rexhep U, Christian B, Anja K, Claudia M, Daniela M, Michal JO, et al. Temporal activation of WNT/β-catenin signaling is sufficient to inhibit SOX10 expression and block melanoma growth. Oncogene. 2020;39:4132–54.
Wang L, Dehm SM, Hillman DW, Sicotte H, Tan W, Gormley M, et al. A prospective genome-wide study of prostate cancer metastases reveals association of wnt pathway activation and increased cell cycle proliferation with primary resistance to abiraterone acetate–prednisone. Ann Oncol. 2017;29:352–60.
Stewart DJ. Wnt signaling pathway in non–small cell lung cancer. J Natl Cancer I. 2014;106:djt356.
Spranger S, Bao R, Gajewski TF. Melanoma-intrinsic β-catenin signalling prevents anti-tumour immunity. Nature. 2015;523:231–5.
Nusse R. Wnt signaling in disease and in development. Cell Res. 2005;15:28–32.
Kahn M. Can we safely target the WNT pathway? Nat Rev Drug Disco. 2014;13:513–32.
Park JH, Lee JM, Lee EJ, Hwang WB, Kim DJ. Indole-3-Carbinol Promotes Goblet-Cell Differentiation Regulating Wnt and Notch Signaling Pathways AhR-Dependently. Mol Cells. 2018;41:290–300.
Kessler M, Hoffmann K, Brinkmann V, Thieck O, Jackisch S, Toelle B, et al. The Notch and Wnt pathways regulate stemness and differentiation in human fallopian tube organoids. Nat Commun. 2015;6:8989.
Acebron SP, Niehrs C. β-Catenin-Independent Roles of Wnt/LRP6 Signaling. Trends Cell Biol. 2016;26:956–67.
D’Errico M, de Rinaldis E, Blasi MF, Viti V, Falchett M, Calcagnile A, et al. Genome-wide expression profile of sporadic gastric cancers with microsatellite instability. Eur J Cancer. 2009;45:461–9.
Cui J, Chen Y, Chou WC, Sun L, Chen L, Suo J, et al. An integrated transcriptomic and computational analysis for biomarker identification in gastric cancer. Nucleic Acids Res. 2010;39:1197–207.
Uhlen M, Bandrowski A, Carr S, Edwards A, Ellenberg J, Lundberg E, et al. A proposal for validation of antibodies. Nat Methods. 2016;13:823.
Ooi CH, Ivanova T, Wu J, Lee M, Tan IB, Tao J, et al. Oncogenic pathway combinations predict clinical prognosis in gastric cancer. PLoS Genet. 2009;5:e1000676.
Cristescu R, Lee J, Nebozhyn M, Kim KM, Ting JC, Wong SS, et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat Med. 2015;21:449–56.
Kim HK, Choi IJ, Kim CG, Kim HS, Oshima A, Michalowski A, et al. A gene expression signature of acquired chemoresistance to cisplatin and fluorouracil combination chemotherapy in gastric cancer patients. PloS ONE. 2011;6:e16694.
Pérez-Garijo A, Steller H. Spreading the word: non-autonomous effects of apoptosis during development, regeneration and disease. Development. 2015;142:3253–62.
Cottini F, Hideshima T, Xu C, Sattler M, Dori M, Agnelli L, et al. Rescue of Hippo coactivator YAP1 triggers DNA damage-induced apoptosis in hematological cancers. Nat Med. 2014;20:599–606.
Luehders K, Sasai N, Davaapil H, Kurosawa-Yoshida M, Hiura H, Brah T, et al. The small leucine-rich repeat secreted protein Asporin induces eyes in Xenopus embryos through the IGF signalling pathway. Development. 2015;142:3351–61.
Packham S, Warsito D, Lin Y, Sadi S, Karlsson R, Sehat B. Nuclear translocation of IGF-1R via p150 Glued and an importin-β/RanBP2-dependent pathway in cancer cells. Oncogene. 2015;34:2227–38.
Warsito D, Sjöström S, Andersson S, Larsson O, Sehat B. Nuclear IGF1R is a transcriptional co-activator of LEF1/TCF. EMBO Rep. 2012;13:244–50.
Hughes RM, Simons BW, Khan H, Miller R, Kugler V, Torquato S, et al. Asporin restricts mesenchymal stromal cell differentiation, alters the tumor microenvironment, and drives metastatic progression. Cancer Res. 2019;79:3636–50.
Pinto M, Oliveira C, Cirnes L, Machado JC, Ramires M, Nogueira A, et al. Promoter methylation of TGFβ receptor I and mutation of TGFβ receptor II are frequent events in MSI sporadic gastric carcinomas. J Pathol. 2003;200:32–8.
Jingwen L, Xuqiao C, Ping L. The role of TGF-β and its receptors in gastrointestinal cancers. Transl Oncol. 2019;12:475–84.
Satoyoshi R, Kuriyama S, Aiba N, Yashiro M, Tanaka M. Asporin activates coordinated invasion of scirrhous gastric cancer and cancer-associated fibroblasts. Oncogene. 2015;34:650–60.
Lee HH. ASO Author Reflections: borrmann type as a characteristic phenotype of advanced gastric cancer. Ann Surg Oncol. 2018;25:778–9.
Li C, Oh SJ, Kim S, Hyung WJ, Yan M, Zhu ZG, et al. Macroscopic Borrmann type as a simple prognostic indicator in patients with advanced gastric cancer. Oncol. 2009;77:197–204.
An JY, Kang TH, Choi MG, Noh JH, Sohn TS, Kim S. Borrmann type IV: an independent prognostic factor for survival in gastric cancer. J Gastrointest Surg. 2008;12:1364.
Ikeguchi M, Yamamoto O, Kaibara N. Management protocol for scirrhous gastric cancer. In Vivo. 2004;18:577–80.
Santiago L, Daniels G, Wang D, Deng FM, Lee P. Wnt signaling pathway protein LEF1 in cancer, as a biomarker for prognosis and a target for treatment. Am J Cancer Res. 2017;7:1389–406.
Chiurillo MA. Role of the Wnt/β-catenin pathway in gastric cancer: An indepth literature review. World J Exp Med. 2015;5:84–102.
Hengcun L, Zheng Z, Lei C, Xiujing S, Yu Z, Qingdong G, et al. Cytoplasmic Asporin promotes cell migration by regulating TGF-β/Smad2/3 pathway and indicates a poor prognosis in colorectal cancer. Cell Death Dis. 2019;10:109.
Fukata M, Chen A, Klepper A, Krishnareddy S, Vamadevan AS, Thomas LS, et al. Cox-2 is regulated by Toll-like receptor-4 (TLR4) signaling: Role in proliferation and apoptosis in the intestine. Gastroenterology. 2006;131:862–77.
Kovalovich K, Li W, DeAngelis R, Greenbaum LE, Ciliberto G, Taub R. IL-6 protects against Fas-mediated death by establishing a critical level of anti-apoptotic hepatic proteins FLIP, Bcl-2 and Bcl-xL. J Biol Chem. 2001;276:26605–13.
Su F, Overholtzer M, Besser D, Levine AJ. WISP-1 attenuates p53-mediated apoptosis in response to DNA damage through activation of the Akt kinase. Gene Dev. 2002;16:46–57.
Acknowledgements
This study was supported by National Natural Science Foundation of China (81570507, 81702314) and National Key Research and Development Program of China (2017YFC0113600).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Zhang, Z., Min, L., Li, H. et al. Asporin represses gastric cancer apoptosis via activating LEF1-mediated gene transcription independent of β-catenin. Oncogene 40, 4552–4566 (2021). https://doi.org/10.1038/s41388-021-01858-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-021-01858-7
This article is cited by
-
ZMIZ2 facilitates hepatocellular carcinoma progression via LEF1 mediated activation of Wnt/β-catenin pathway
Experimental Hematology & Oncology (2024)
-
Carcinogenesis promotion in oral squamous cell carcinoma: KDM4A complex-mediated gene transcriptional suppression by LEF1
Cell Death & Disease (2023)