Abstract
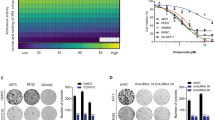
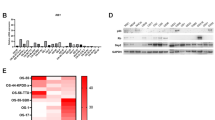
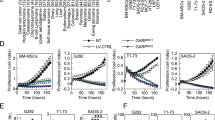
Ewing sarcoma (ES) is the second most common bone tumor in children and young adults. Unfortunately, there have been minimal recent advancements in improving patient outcomes, especially in metastatic and recurrent diseases. In this study, we investigated the biological role of p21-activated kinases (PAKs) in ES, and the ability to therapeutically target them in high-risk disease. Via informatics analysis, we established the inverse association of PAK1 and PAK4 expression with clinical stage and outcome in ES patients. Through expression knockdown and small-molecule inhibition of PAKs, utilizing FRAX-597, KPT-9274, and PF-3758309 in multiple ES cell lines and patient-derived xenograft models, we further explored the role of PAKs in ES tumor growth and metastatic capabilities. In vitro studies in several ES cell lines indicated that diminishing PAK1 and PAK4 expression reduces tumor cell viability, migratory, and invasive properties. In vivo studies using PAK4 inhibitors, KPT-9274 and PF-3758309 demonstrated significant inhibition of primary and metastatic tumor formation, while transcriptomic analysis of PAK4-inhibitor-treated tumors identified concomitant suppression of Notch, β-catenin, and hypoxia-mediated signatures. In addition, the analysis showed enrichment of anti-tumor immune regulatory mechanisms, including interferon (IFN)-ɣ and IFN-α responses. Altogether, our molecular and pre-clinical studies are the first to establish a critical role for PAKs in ES development and progression, and consequently as viable therapeutic targets for the treatment of high-risk ES in the near future.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ries LAG, Smith MA, Gurney JG, Linet M, Tamra T, Young JLBG. Cancer incidence and survival among children and adolescents: United States SEER Program 1975–1995, National Cancer Institute, SEER Program. NIH Pub. 1975. Accessed 2 Apr 2018. https://seer.cancer.gov/archive/publications/childhood/childhood-monograph.pdf
Stiller CA, Bielack SS, Jundt G, Steliarova-Foucher E. Bone tumours in European children and adolescents, 1978–1997. Report from the automated childhood cancer information system project. Eur J Cancer. 2006;42:2124–35.
Turc-Carel C, Philip I, Berger MP, Philip T, Lenoir GM. Chromosome study of Ewing’s sarcoma (ES) cell lines. Consistency of a reciprocal translocation t(11;22)(q24;q12). Cancer Genet Cytogenet. 1984;12:1–19.
Turc-Carel C, Aurias A, Mugneret F, Lizard S, Sidaner I, Volk C, et al. Chromosomes in Ewing’s sarcoma. I. An evaluation of 85 cases of remarkable consistency of t(11;22)(q24;q12). Cancer Genet Cytogenet. 1988;32:229–38.
Zucman J, Delattre O, Desmaze C, Plougastel B, Joubert I, Melot T, et al. Cloning and characterization of the Ewing’s sarcoma and peripheral neuroepithelioma t(11;22) translocation breakpoints. Genes Chromosom Cancer. 1992;5:271–7.
Delattre O, Zucman J, Plougastel B, Desmaze C, Melot T, Peter M, et al. Gene fusion with an ETS DNA-binding domain caused by chromosome translocation in human tumours. Nature. 1992;359:162–5.
Radu M, Semenova G, Kosoff R, Chernoff J. Pak signaling in the development and progression of cancer. Accessed 21 Mar 2018. https://www-ncbi-nlm-nih-gov.ezproxyhost.library.tmc.edu/pmc/articles/PMC4115244/pdf/nihms556174.pdf
Rane CK, Minden A, Lehman S. P21 activated kinase signaling in cancer. Semin Cancer Biol 2018;1–10. Accessed 21 Mar 2018. www.elsevier.com/locate/semcancer
Manser E, Leung T, Salihuddin H, Zhao Z, Lim L. A brain serine/threonine protein kinase activated by Cdc42 and Rac1. Nature. 1994;367:40–6.
Satterfield L, Shuck R, Kurenbekova L, Allen-Rhoades W, Edwards D, Huang S, et al. miR-130b directly targets ARHGAP1 to drive activation of a metastatic CDC42-PAK1-AP1 positive feedback loop in Ewing sarcoma. Int J Cancer. 2017;141:2062–75.
He H, Baldwin GS. p21-activated kinases and gastrointestinal cancer. 2013. Accessed 21 Mar 2018. https://ac-els-cdn-com.ezproxyhost.library.tmc.edu/S0167488912002972/1-s2.0-S0167488912002972-main.pdf?_tid=f385ce32-a98c-4bc5-807b-081eb8aa95e1&acdnat=1521651015_73053adf197a81b146eacc7a615a4f2f
Yeo D, He H, Baldwin GS, Nikfarjam M. The role of p21-activated kinases in pancreatic cancer. 2018. https://insights-ovid-com.ezproxyhost.library.tmc.edu/pubmed?pmid=25760284
Lu W, Qu J-J, Li B-L, Lu C, Yan Q, Wu X-M, et al. Overexpression of p21-activated kinase 1 promotes endometrial cancer progression. Oncol Rep. 2013;29:1547–55.
Aboukameel A, Muqbil I, Senapedis W, Baloglu E, Landesman Y, Shacham S, et al. Novel p21-activated kinase 4 (PAK4) allosteric modulators overcome drug resistance and stemness in pancreatic ductal adenocarcinoma. Mol Cancer Ther. 2017;16:76–87.
Jin-Xing Y, Yu-Jing H, Hang Z, Rong-Cheng L. Expression of PAK4 in breast cancer and benign breast pathological changes. 2018. http://www.j-smu.com/pdf2/201005/201005981.pdf
Rane C, Senapedis W, Baloglu E, Landesman Y, Crochiere M, Das-Gupta S, et al. A novel orally bioavailable compound KPT-9274 inhibits PAK4, and blocks triple negative breast cancer tumor growth. Sci Rep. 2017;7. Accessed 21 Mar 2018. https://www-ncbi-nlm-nih-gov.ezproxyhost.library.tmc.edu/pmc/articles/PMC5309789/pdf/srep42555.pdf
Arias-Romero LE, Villamar-Cruz O, Pacheco A, Kosoff R, Huang M, Muthuswamy SK, et al. A Rac-Pak signaling pathway is essential for ErbB2-mediated transformation of human breast epithelial cancer cells Luis. Oncogene 2010;29:5839–49.
Park M-H, Lee H-S, Lee C-S, You S, Kim D-J, Park B-H, et al. p21-Activated kinase 4 promotes prostate cancer progression through CREB. Oncogene. 2012;32. Accessed 21 Mar 2018. http://www.nature.com.ezproxyhost.library.tmc.edu/articles/onc2012255.pdf
Siu MK, Chan HY, Kong DS, Wong ES, Wong OG, Ngan HY, et al. p21-activated kinase 4 regulates ovarian cancer cell proliferation, migration, and invasion and contributes to poor prognosis in patients. 2018. https://www-ncbi-nlm-nih-gov.ezproxyhost.library.tmc.edu/pmc/articles/PMC2972956/pdf/pnas.200907481.pdf
Salamon J, Hoffmann T, Elies E, Peldschus K, Johansen JS, Lü Ers G, et al. Antibody directed against human YKL-40 increases tumor volume in a human melanoma xenograft model in scid mice. Hirsch E, editor. PLoS ONE. 2014;9:e95822
Abo A. PAK4, a novel effector for Cdc42Hs, is implicated in the reorganization of the actin cytoskeleton and in the formation of filopodia. EMBO J. 1998;17:6527–40.
Callow MG, Clairvoyant F, Zhu S, Schryver B, Whyte DB, Bischoff JR, et al. Requirement for PAK4 in the anchorage-independent growth of human cancer cell lines. J Biol Chem. 2002;277:550–8.
Qu J, Cammarano MS, Shi Q, Ha KC, de Lanerolle P, Minden A. Activated PAK4 regulates cell adhesion and anchorage-independent growth. Mol Cell Biol. 2001;21:3523–33.
Liu Y, Xiao H, Tian Y, Nekrasova T, Hao X, Lee HJ, et al. The pak4 protein kinase plays a key role in cell survival and tumorigenesis in athymic mice. Mol Cancer Res. 2008;6:1215–24.
Yeo D, He H, Patel O, Lowy AM, Baldwin GS, Nikfarjam M. FRAX597, a PAK1 inhibitor, synergistically reduces pancreatic cancer growth when combined with gemcitabine. 2018. https://www-ncbi-nlm-nih-gov.ezproxyhost.library.tmc.edu/pmc/articles/PMC4715347/pdf/12885_2016_Article_2057.pdf
Mileshkin LR, Rosen LS, Blumenkopf T, Breazna A, Darang S, Davison J, et al. Phase I, dose-escalation, safety, pharmacokinetic, and pharmacodynamic study of single-agent PF-03758309, an oral PAK inhibitor, in patients with advanced solid tumors. J Clin Oncol. 2011;29:e13607–e13607.
Naing A, Leong S, Pishvaian MJ, Razak ARA, Mahipal A, Berlin J, et al. 374PDA first in human phase 1 study of KPT-9274, a first in class dual inhibitor of PAK4 and NAMPT, in patients with advanced solid malignancies or NHL. Ann Oncol 2017;28. Accessed 6 Jan 2020. https://oncologypro.esmo.org/Meeting-Resources/ESMO-2017-Congress/A-First-in-Human-Phase-1-Study-of-KPT-9274-a-First-in-Class-Dual-Inhibitor-of-PAK4-and-NAMPT-in-Patients-with-Advanced-Solid-Malignancies-or-NHL
Li Y, Shao Y, Tong Y, Shen T, Zhang J, Li Y, et al. Nucleo-cytoplasmic shuttling of PAK4 modulates β-catenin intracellular translocation and signaling. Biochim Biophys Acta. 2012;1823:465–75.
Aboud OA, Chen C-H, Senapedis W, Baloglu E, Argueta C, Weiss RH. Dual and specific inhibition of NAMPT and PAK4 by KPT-9274 decreases kidney cancer growth. Mol Cancer Ther. 2016;15:2119–29.
Hawkins AG, Basrur V, Da Veiga Leprevost F, Pedersen E, Sperring C, Nesvizhskii AI, et al. The Ewing sarcoma secretome and its response to activation of Wnt/beta-catenin signaling. Mol Cell Proteom. 2018;17:901–12.
Ryu BJ, Lee H, Kim S-H, Heo J-N, Choi S-W, Yeon J-T, et al. PF-3758309, p21-activated kinase 4 inhibitor, suppresses migration and invasion of A549 human lung cancer cells via regulation of CREB, NF-κB, and β-catenin signalings. Mol Cell Biochem. 2014;389:69–77.
Schmalhofer O, Brabletz S, Brabletz T. E-cadherin, β-catenin, and ZEB1 in malignant progression of cancer. Cancer Metastasis Rev. 2009;28:151–66.
Pedersen EA, Menon R, Bailey KM, Thomas DG, Van Noord RA, Tran J, et al. Activation of Wnt/ -Catenin in Ewing Sarcoma Cells Antagonizes EWS/ETS function and promotes phenotypic transition to more metastatic cell states. Cancer Res. 2016;76:5040–53.
Volchenboum SL, Andrade J, Huang L, Barkauskas DA, Krailo M, Womer RB, et al. Gene expression profiling of Ewing sarcoma tumours reveals the prognostic importance of tumour-stromal interactions: a report from the Children’s oncology group. J Pathol Clin Res. 2015;1:83–94.
Mutz CN, Schwentner R, Aryee DNT, Bouchard EDJ, Mejia EM, Hatch GM, et al. EWS-FLI1 confers exquisite sensitivity to NAMPT inhibition in Ewing sarcoma cells. Oncotarget. 2017;8:24679–93.
Heske CM, Davis MI, Baumgart JT, Wilson K, Gormally MV, Chen L, et al. Matrix screen identifies synergistic combination of PARP inhibitors and nicotinamide phosphoribosyltransferase (NAMPT) inhibitors in Ewing sarcoma. Clin Cancer Res. 2017;23:7301–11.
Giard ld J, Aaronson SA, Todaro GJ, Arnstein P, Kersey JH. In vitro cultivation of human tumors: Establishment of cell lines derived from a series of solid tumors. J Natl Cancer Inst. 1973;51:1417–23.
Batra S, Reynolds CP, Maurer BJ. Fenretinide cytotoxicity for Ewing’s sarcoma and primitive neuroectodermal tumor cell lines is decreased by hypoxia and synergistically enhanced by ceramide modulators. Cancer Res. 2004;64:5415–24.
Reynolds CP, Tomayko MM, Donner L, Helson L, Seeger RC, Triche TJ, et al. Biological classification of cell lines derived from human extracranial neural tumors. Prog Clin Biol Res. 1988;271:291–306.
Whang-Peng J, Triche TJ, Knutsen T, Miser J, Kao-Shan S, Tsai S, et al. Cytogenetic characterization of selected small round cell tumors of childhood. Cancer Genet Cytogenet. 1986;21:185–208.
Pietsch T, Gottert E, Feickart H-J, Riehm H, Blin N, Kovacs G. MHH-ES-1, a new Ewing sarcoma cell line. 2018. https://ac-els-cdn-com.ezproxyhost.library.tmc.edu/0165460889905682/1-s2.0-0165460889905682-main.pdf?_tid=50613fc5-a1eb-4fce-98c1-b8d188254436&acdnat=1522263530_fbc11162f6668ea36654d2a203d50f16
Bloom ET. Further definition by cytotoxicity tests of cell surface antigens of human sarcomas in culture1. Cancer Res. 1972;32:960–7.
Technologies L. Lipofectamine® RNAiMAX reagent protocol outline lipofectamine® RNAiMAX transfection protocol. 2018. https://tools.thermofisher.com/content/sfs/manuals/Lipofectamine_RNAiMAX_Reag_protocol.pdf
CellTiter-Glo® Luminescent cell viability assay instructions for use of products G7570, G7571, G7572 and G7573. 2018;608–277. https://www.promega.com/-/media/files/resources/protocols/technical-bulletins/0/celltiter-glo-luminescent-cell-viability-assay-protocol.pdf
Co LLC S-A. 96992 Cell Counting Kit—8. Accessed 29 Mar 2018. https://www.sigmaaldrich.com/content/dam/sigma-aldrich/docs/Sigma/Datasheet/6/96992dat.pdf
Chou TC, Motzer RJ, Tong Y, Bosl GJ. Computerized quantitation of synergism and antagonism of taxol, topotecan, and cisplatin against human teratocarcinoma cell growth: a rational approach to clinical protocol design. J Natl Cancer Inst. 1994;86:1517–24.
Subramanian A, Tamayo P, Mootha VK, Mukherjee BL, Ebert MA, Gillette A, et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci. 2005;102:15545–50.
Acknowledgements
This work was supported by Cancer Prevention and Research Institute of Texas (CPRIT) post-doctoral training grant RP160283 (S.Q.) and The Faris D. Virani Ewing sarcoma Center. This project was supported by the Cytometry and Cell Sorting Core at Baylor College of Medicine with funding from the CPRIT Core Facility Support Award (CPRIT-RP180672) and the NIH (CA125123 and RR024574).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Qasim, S.L., Sierra, L., Shuck, R. et al. p21-activated kinases as viable therapeutic targets for the treatment of high-risk Ewing sarcoma. Oncogene 40, 1176–1190 (2021). https://doi.org/10.1038/s41388-020-01600-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-020-01600-9