Abstract
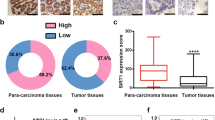
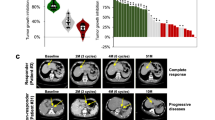
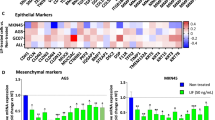
5-FU is one of the key drugs in the treatment of gastric cancer (GC). Much evidence has shown that cancer stem cells (CSCs) play a key role in the acquisition of drug resistance. The organoid is a novel 3D cell culture system technology that sustains stem-cell-driven formation of near-physiological, self-renewing tissues using specific niche factors in a dish. In this study, we established GC organoids (GCOs) and gradually treated them with higher concentrations of 5-FU. We successfully harvested four 5-FU-resistant GCOs, which were supported by significant changes in the expression of molecules related to 5-FU metabolism. We then performed microarray analysis using three normal gastric organoids and three pairs of 5-FU-resistant and parental GCOs. Through the comparison of expression profiles and further validation, we chose KHDRBS3 as a target gene. We found KHDRBS3 to be an independent prognostic factor in GC patients, especially in GC patients treated with 5-FU chemotherapy. We also determined that KHDRBS3 might play an important role in the acquisition of stem cell-like features, such as multi-drug resistance and organoid formation, by regulating CD44 variant expression. We found KHDRBS3, which is thought to play an important role in the acquisition of characteristics of CSCs in GC, to be a promising candidate marker for predicting therapeutic effect and prognosis in GC patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bray Freddie, Ferlay Jacques, Soerjomataram Isabelle, Siegel RebeccaL, Torre LindseyA, Jemal Ahmedin, et al. Global Cancer Statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Al-Batran S-E, Hofheinz RD, Pauligk C, Kopp H-G, Haag GM, Luley KB, et al. Histopathological regression after neoadjuvant docetaxel, oxaliplatin, fluorouracil, and leucovorin versus epirubicin, cisplatin, and fluorouracil or capecitabine in patients with resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4-AIO): results from the phase 2 part of a multicentre, open-label, randomised phase 2/3 trial. Lancet Oncol. 2016;17:1697–708.
Nakamura A, Nakajima G, Okuyama R, Kuramochi H, Kondoh Y, Kanemura T, et al. Enhancement of 5-fluorouracil-induced cytotoxicity by leucovorin in 5-fluorouracil-resistant gastric cancer cells with upregulated expression of thymidylate synthase. Gastric Cancer. 2014;17:188–95.
Holohan C, Van Schaeybroeck S, Longley DB, Johnston PG. Cancer drug resistance: an evolving paradigm. Nat Rev Cancer. 2013;13:714–26.
Shibue T, Weinberg RA. EMT, CSCs, and drug resistance: the mechanistic link and clinical implications. Nat Rev Clin Oncol. 2017;14:611–29.
Sato T, Vries RG, Snippert HJ, van de Wetering M, Barker N, Stange DE, et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 2009;459:262–5.
Boj SF, Hwang CI, Baker LA, Chio II, Engle DD, Corbo V, et al. Organoid models of human and mouse ductal pancreatic cancer. Cell. 2015;160:324–38.
Lancaster MA, Knoblich JA. Organogenesis in a dish: modeling development and disease using organoid technologies. Science. 2014;345:1247125.
Seidlitz T, Merker SR, Rothe A, Zakrzewski F, von Neubeck C, Grutzmann K, et al. Human gastric cancer modelling using organoids. Gut. 2019;68:207–17.
Yan HHN, Siu HC, Law S, Ho SL, Yue SSK, Tsui WY, et al. A comprehensive human gastric cancer organoid biobank captures tumor subtype heterogeneity and enables therapeutic screening. Cell Stem Cell. 2018;23:882–97. e811
Steele NG, Chakrabarti J, Wang J, Biesiada J, Holokai L, Chang J, et al. An organoid-based preclinical model of human gastric cancer. Cell Mol Gastroenterol Hepatol. 2019;7:161–84.
van de Wetering M, Francies HE, Francis JM, Bounova G, Iorio F, Pronk A, et al. Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell. 2015;161:933–45.
Sato T, Clevers H. Growing self-organizing mini-gutsfrom a single intestinal stem cell: mechanism and applications. Science. 2013;340:1190–4.
Venables CV JP, Chew SL, Elliott DJ, Cowmeadow RB, Wu J, Cooke HJ, et al. T-STAR/ÉTOILE: a novel relative of SAM68 that interacts with an RNA-binding protein implicated in spermatogenesis. Hum Mol Genet. 1999;8:959–69.
Matsumoto Y, Itou J, Sato F, Toi M. SALL4 - KHDRBS3 network enhances stemness by modulating CD44 splicing in basal-like breast cancer. Cancer Med. 2018;7:454–62.
Lei KF, Liu BY, Wang YF, Chen XH, Yu BQ, Guo Y, et al. SerpinB5 interacts with KHDRBS3 and FBXO32 in gastric cancer cells. Oncol Rep. 2011;26:1115–20.
Shen Xiao-Ming, Zhou Chong, Lian Lian, Li Li-Qun, Li Wei, Tao Min. Relationship between the DPD and TS mRNA expression and the response to S-1-based chemotherapy and prognosis in patients with advanced gastric cancer. Cell Biochem Biophys. 2015;71:1653–61.
Gao Y, Zhao Q, Mu X, Zhu H, Liu B, Yao B, et al. SREBP1 promotes 5-FU resistance in colorectal cancer cells by inhibiting the expression of caspase7. Int J Clin Exp Pathol. 2019;12:1095–1100.
Wang W, Cassidy J, O’Brien V, Ryan KM, Collie-Duguid E. Mechanistic and predictive profiling of 5-Fluorouracil resistance in human cancer cells. Cancer Res. 2004;64:8167–76.
Mazzoldi EL, Pasto A, Pilotto G, Minuzzo S, Piga I, Palumbo P, et al. Comparison of the genomic profile of cancer stem cells and their non-stem counterpart: the case of ovarian cancer. J. Clin. Med. 2020;9:368.
Okuyama K, Fukushima H, Naruse T, Yanamoto S, Tsuchihashi H, Umeda M. CD44 variant 6 expression and tumor budding in the medullary invasion front of mandibular gingival squamous cell carcinoma are predictive factors for cervical lymph node metastasis. Pathol Oncol Res. 2019;25:603–9.
Pacifico MD, Grover R, Richman PI, Daley FM, Buffa F, Wilson GD. CD44v3 levels in primary cutaneous melanoma are predictive of prognosis: assessment by the use of tissue microarray. Int J Cancer. 2006;118:1460–4.
Ishikawa A, Sakamoto N, Honma R, Taniyama D, Fukada K, Hattori T. et al. Annexin A10 is involved in the induction of pancreatic duodenal homeobox1 in gastric cancer tissue, cells and organoids. Oncol Rep. 2020;43:581–90.
Honma R, Goto K, Sakamoto N, Sekino Y, Sentani K, Oue N, et al. Expression and function of Uc.160+, a transcribed ultraconserved region, in gastric cancer. Gastric Cancer. 2017;20:960–9.
Sakamoto N, Naito Y, Oue N, Sentani K, Uraoka N, Oo HZ, et al. MicroRNA-148a is downregulated in gastric cancer, targets MMP7, and indicates tumor invasiveness and poor prognosis. Cancer Sci. 2014;105:236–43.
Acknowledgements
We thank Mr. Shinichi Norimura (Technical Center, Hiroshima University) for his excellent technical assistance. This research was carried out with the kind cooperation of the Research Center for Molecular Medicine of the Faculty of Medicine of Hiroshima University. We also thank the Analysis Center of Life Science of Hiroshima University for the use of their facilities. The R-spondin-producing cell line was a kind gift from Professor Jeffery Whitsett (Cincinnati Children’s Hospital Medical Center, Cincinnati, OH, USA). We would like to thank Professor Eric Fearon (University of Michigan, Ann Arbor, MI, USA) for providing collaborative research resources and comments.
Funding
The present study was supported by Grants in Aid for Scientific Research (JP15H04713, JP16K08691, JP16H06999) and for Challenging Exploratory Research (26670175, JP16K15247) from the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ukai, S., Honma, R., Sakamoto, N. et al. Molecular biological analysis of 5-FU-resistant gastric cancer organoids; KHDRBS3 contributes to the attainment of features of cancer stem cell. Oncogene 39, 7265–7278 (2020). https://doi.org/10.1038/s41388-020-01492-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-020-01492-9
This article is cited by
-
Patient-derived organoids in human cancer: a platform for fundamental research and precision medicine
Molecular Biomedicine (2024)
-
3D Models of Sarcomas: The Next-generation Tool for Personalized Medicine
Phenomics (2023)
-
Knockout KHDRBS3 inhibits cell proliferation and glucocorticoid resistance in acute lymphoblastic leukemia
Molecular & Cellular Toxicology (2023)
-
Tumor organoids: applications in cancer modeling and potentials in precision medicine
Journal of Hematology & Oncology (2022)
-
Translational models of 3-D organoids and cancer stem cells in gastric cancer research
Stem Cell Research & Therapy (2021)