Abstract
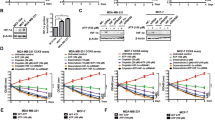
Our previous research demonstrated that extracellular adenosine 5′-triphosphate (ATP) could promote breast cancer cell invasion. However, the impact of extracellular ATP on chemoresistance and the mechanisms behind ATP pro-invasion and pro-chemoresistance remain unclear. Here we aimed to determine the molecules or signaling pathways involved. cDNA microarray was performed to identify the differentially expressed genes before and after ATP treatment. As a result, Sex-determining region Y-box 9 (SOX9) was up-regulated after ATP treatment in breast cancer cells. In vitro invasion and migration assays demonstrated that knocking down SOX9 attenuated ATP-driven invasive capability. Mass spectrometry and co-IP revealed that SOX9 interacted with Janus kinase 1 (JAK1). Afterward, IL-6-JAK1-STAT3 signaling was demonstrated to promote SOX9 expression and invasion following ATP treatment. Notably, ATP-IL-6-SOX9 signaling was shown to stimulate chemoresistance in breast cancer cells. ChIP assays identified some potential SOX9 target genes, among which carcinoembryonic antigen-related cell adhesion molecule 5/6 (CEACAM5/6) was demonstrated to mediate ATP pro-invasive function, while ATP-binding cassette subfamily B member 1 (ABCB1) and ATP-binding cassette subfamily G member 2 (ABCG2) mediated ATP-driven chemoresistance. In addition, SOX9-knockdown and apyrase (an ATP hydrolase)-treated MDA-MB-231 cells illustrated decreased tumor growth and enhanced drug sensitivity in nude mice. In vitro spheroid formation assays also proved the significance of ATP-SOX9 in mediating chemoresistance. Moreover, molecules involved in ATP-SOX9 signaling were up-regulated in human breast carcinoma specimens and were associated with poor prognosis. Altogether, SOX9 signaling is vital in ATP-driven invasion and chemoresistance, which may serve as a potential target for breast cancer therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Patel A, Malinovska L, Saha S, Wang J, Alberti S, Krishnan Y, et al. ATP as a biological hydrotrope. Science. 2017;356:753–6. https://doi.org/10.1126/science.aaf6846
Pearson RA, Dale N, Llaudet E, Mobbs P. ATP released via gap junction hemichannels from the pigment epithelium regulates neural retinal progenitor proliferation. Neuron. 2005;46:731–44. https://doi.org/10.1016/j.neuron.2005.04.024
Atarashi K, Nishimura J, Shima T, Umesaki Y, Yamamoto M, Onoue M, et al. ATP drives lamina propria T(H)17 cell differentiation. Nature. 2008;455:808–12. https://doi.org/10.1038/nature07240
McKenney RJ, Huynh W, Tanenbaum ME, Bhabha G, Vale RD. Activation of cytoplasmic dynein motility by dynactin-cargo adapter complexes. Science. 2014;345:337–41. https://doi.org/10.1126/science.1254198
Eguchi Y, Shimizu S, Tsujimoto Y. Intracellular ATP levels determine cell death fate by apoptosis or necrosis. Cancer Res. 1997;57:1835–40
Galluzzi L, Bravo-San Pedro JM, Kroemer G. Necrosis: linking the inflammasome to inflammation. Cell Rep. 2015;11:1501–2. https://doi.org/10.1016/j.celrep.2015.05.041
Di Virgilio F, Adinolfi E. Extracellular purines, purinergic receptors and tumor growth. Oncogene. 2017;36:293–303. https://doi.org/10.1038/onc.2016.206
Antonioli L, Blandizzi C, Pacher P, Hasko G. Immunity, inflammation and cancer: a leading role for adenosine. Nat Rev Cancer. 2013;13:842–57. https://doi.org/10.1038/nrc3613
Di Virgilio F. Purines, purinergic receptors, and cancer. Cancer Res. 2012;72:5441–7. https://doi.org/10.1158/0008-5472.CAN-12-1600
Chen L, He HY, Li HM, Zheng J, Heng WJ, You JF, et al. ERK1/2 and p38 pathways are required for P2Y receptor-mediated prostate cancer invasion. Cancer Lett. 2004;215:239–47. https://doi.org/10.1016/j.canlet.2004.05.023
Li WH, Qiu Y, Zhang HQ, Liu Y, You JF, Tian XX, et al. P2Y2 receptor promotes cell invasion and metastasis in prostate cancer cells. Br J Cancer. 2013;109:1666–75. https://doi.org/10.1038/bjc.2013.484
Li WH, Qiu Y, Zhang HQ, Tian XX, Fang WG. P2Y2 receptor and EGFR cooperate to promote prostate cancer cell invasion via ERK1/2 pathway. PLoS ONE. 2015;10:e0133165. https://doi.org/10.1371/journal.pone.0133165
Qiu Y, Li WH, Zhang HQ, Liu Y, Tian XX, Fang WG. P2X7 mediates ATP-driven invasiveness in prostate cancer cells. PLoS ONE. 2014;9:e114371. https://doi.org/10.1371/journal.pone.0114371
Zhang JL, Liu Y, Yang H, Zhang HQ, Tian XX, Fang WG. ATP-P2Y2-beta-catenin axis promotes cell invasion in breast cancer cells. Cancer Sci. 2017;108:1318–27. https://doi.org/10.1111/cas.13273
Qiu Y, Liu Y, Li WH, Zhang HQ, Tian XX, Fang WG. P2Y2 receptor promotes the migration and invasion of breast cancer cells via EMT-related genes Snail and E-cadherin. Oncol Rep. 2018;39:138–50. https://doi.org/10.3892/or.2017.6081
Liu Y, Geng YH, Yang H, Yang H, Zhou YT, Zhang HQ, et al. Extracellular ATP drives breast cancer cell migration and metastasis via S100A4 production by cancer cells and fibroblasts. Cancer Lett. 2018;430:1–10. https://doi.org/10.1016/j.canlet.2018.04.043
Yang H, Geng YH, Wang P, Zhou YT, Yang H, Huo YF, et al. Extracellular ATP promotes breast cancer invasion and epithelial-mesenchymal transition via hypoxia-inducible factor 2alpha signaling. Cancer Sci. 2019;110:2456–70. https://doi.org/10.1111/cas.14086
Kumar S, Liu J, Pang P, Krautzberger AM, Reginensi A, Akiyama H, et al. Sox9 activation highlights a cellular pathway of renal repair in the acutely injured mammalian kidney. Cell Rep. 2015;12:1325–38. https://doi.org/10.1016/j.celrep.2015.07.034
Wang H, Yu C, Gao X, Welte T, Muscarella AM, Tian L, et al. The osteogenic niche promotes early-stage bone colonization of disseminated breast cancer cells. Cancer Cell. 2015;27:193–210. https://doi.org/10.1016/j.ccell.2014.11.017
Larsimont JC, Youssef KK, Sanchez-Danes A, Sukumaran V, Defrance M, Delatte B, et al. Sox9 controls self-renewal of oncogene targeted cells and links tumor initiation and invasion. Cell Stem Cell. 2015;17:60–73. https://doi.org/10.1016/j.stem.2015.05.008
Cai C, Wang H, He HH, Chen S, He L, Ma F, et al. ERG induces androgen receptor-mediated regulation of SOX9 in prostate cancer. J Clin Investig. 2013;123:1109–22. https://doi.org/10.1172/JCI66666
Chen H, Garbutt CC, Spentzos D, Choy E, Hornicek FJ, Duan Z. Expression and therapeutic potential of SOX9 in Chordoma. Clin Cancer Res. 2017;23:5176–86. https://doi.org/10.1158/1078-0432.CCR-17-0177
Wang H, Leav I, Ibaragi S, Wegner M, Hu GF, Lu ML, et al. SOX9 is expressed in human fetal prostate epithelium and enhances prostate cancer invasion. Cancer Res. 2008;68:1625–30. https://doi.org/10.1158/0008-5472.CAN-07-5915
Crystal AS, Shaw AT, Sequist LV, Friboulet L, Niederst MJ, Lockerman EL, et al. Patient-derived models of acquired resistance can identify effective drug combinations for cancer. Science. 2014;346:1480–6. https://doi.org/10.1126/science.1254721
Verhalen B, Dastvan R, Thangapandian S, Peskova Y, Koteiche HA, Nakamoto RK, et al. Energy transduction and alternating access of the mammalian ABC transporter P-glycoprotein. Nature. 2017;543:738–41. https://doi.org/10.1038/nature21414
Wang X, Li Y, Qian Y, Cao Y, Shriwas P, Zhang H, et al. Extracellular ATP, as an energy and phosphorylating molecule, induces different types of drug resistances in cancer cells through ATP internalization and intracellular ATP level increase. Oncotarget. 2017;8:87860–77. https://doi.org/10.18632/oncotarget.21231
Zheng X, Carstens JL, Kim J, Scheible M, Kaye J, Sugimoto H, et al. Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature. 2015;527:525–30. https://doi.org/10.1038/nature16064
Fischer KR, Durrans A, Lee S, Sheng J, Li F, Wong ST, et al. Epithelial-to-mesenchymal transition is not required for lung metastasis but contributes to chemoresistance. Nature. 2015;527:472–6. https://doi.org/10.1038/nature15748
Yu H, Pardoll D, Jove R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer. 2009;9:798–809. https://doi.org/10.1038/nrc2734
Putoczki T, Ernst M. More than a sidekick: the IL-6 family cytokine IL-11 links inflammation to cancer. J Leukoc Biol. 2010;88:1109–17. https://doi.org/10.1189/jlb.0410226
Johnson DE, O’Keefe RA, Grandis JR. Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat Rev Clin Oncol. 2018;15:234–48. https://doi.org/10.1038/nrclinonc.2018.8
Zalzali H, Naudin C, Bastide P, Quittau-Prevostel C, Yaghi C, Poulat F, et al. CEACAM1, a SOX9 direct transcriptional target identified in the colon epithelium. Oncogene. 2008;27:7131–8. https://doi.org/10.1038/onc.2008.331
Prevostel C, Blache P. The dose-dependent effect of SOX9 and its incidence in colorectal cancer. Eur J Cancer. 2017;86:150–7. https://doi.org/10.1016/j.ejca.2017.08.037
Leung CO, Mak WN, Kai AK, Chan KS, Lee TK, Ng IO, et al. Sox9 confers stemness properties in hepatocellular carcinoma through Frizzled-7 mediated Wnt/beta-catenin signaling. Oncotarget. 2016;7:29371–86. https://doi.org/10.18632/oncotarget.8835
Solini A, Chiozzi P, Morelli A, Fellin R, Di Virgilio F. Human primary fibroblasts in vitro express a purinergic P2X7 receptor coupled to ion fluxes, microvesicle formation and IL-6 release. J Cell Sci. 1999;112(Pt 3):297–305
Gyorffy B, Lanczky A, Eklund AC, Denkert C, Budczies J, Li Q, et al. An online survival analysis tool to rapidly assess the effect of 22,277 genes on breast cancer prognosis using microarray data of 1,809 patients. Breast Cancer Res Treat. 2010;123:725–31. https://doi.org/10.1007/s10549-009-0674-9
Nagy A, Lanczky A, Menyhart O, Gyorffy B. Validation of miRNA prognostic power in hepatocellular carcinoma using expression data of independent datasets. Sci Rep. 2018;8:9227 https://doi.org/10.1038/s41598-018-27521-y
Auger R, Motta I, Benihoud K, Ojcius DM, Kanellopoulos JM. A role for mitogen-activated protein kinase(Erk1/2) activation and non-selective pore formation in P2X7 receptor-mediated thymocyte death. J Biol Chem. 2005;280:28142–51. https://doi.org/10.1074/jbc.M501290200
Gilbert SM, Oliphant CJ, Hassan S, Peille AL, Bronsert P, Falzoni S, et al. ATP in the tumour microenvironment drives expression of nfP2X7, a key mediator of cancer cell survival. Oncogene 2018. https://doi.org/10.1038/s41388-018-0426-6
Roger S, Pelegrin P. P2X7 receptor antagonism in the treatment of cancers. Expert Opin Investig Drugs. 2011;20:875–80. https://doi.org/10.1517/13543784.2011.583918
Ghiringhelli F, Apetoh L, Tesniere A, Aymeric L, Ma Y, Ortiz C, et al. Activation of the NLRP3 inflammasome in dendritic cells induces IL-1beta-dependent adaptive immunity against tumors. Nat Med. 2009;15:1170–8. https://doi.org/10.1038/nm.2028
Di Virgilio F, Falzoni S, Giuliani AL, Adinolfi E. P2 receptors in cancer progression and metastatic spreading. Curr Opin Pharm. 2016;29:17–25. https://doi.org/10.1016/j.coph.2016.05.001
Fang WG, Pirnia F, Bang YJ, Myers CE, Trepel JB. P2-purinergic receptor agonists inhibit the growth of androgen-independent prostate carcinoma cells. J Clin Investig. 1992;89:191–6. https://doi.org/10.1172/JCI115562
Xia M, Zhu Y. The regulation of Sox2 and Sox9 stimulated by ATP in spinal cord astrocytes. J Mol Neurosci. 2015;55:131–40. https://doi.org/10.1007/s12031-014-0393-5
Shan L, Zhou X, Liu X, Wang Y, Su D, Hou Y, et al. FOXK2 elicits massive transcription repression and suppresses the hypoxic response and breast cancer carcinogenesis. Cancer Cell. 2016;30:708–22. https://doi.org/10.1016/j.ccell.2016.09.010
Wilson M, Koopman P. Matching SOX: partner proteins and co-factors of the SOX family of transcriptional regulators. Curr Opin Genet Dev. 2002;12:441–6
Qiu M, Li G, Wang P, Li X, Lai F, Luo R, et al. aarF domain containing kinase 5 gene promotes invasion and migration of lung cancer cells through ADCK5-SOX9-PTTG1 pathway. Exp Cell Res. 2020;392:112002. https://doi.org/10.1016/j.yexcr.2020.112002
Santos JC, Carrasco-Garcia E, Garcia-Puga M, Aldaz P, Montes M, Fernandez-Reyes M, et al. SOX9 elevation acts with canonical WNT signaling to drive gastric cancer progression. Cancer Res. 2016;76:6735–46. https://doi.org/10.1158/0008-5472.CAN-16-1120
Abbosh C, Birkbak NJ, Wilson GA, Jamal-Hanjani M, Constantin T, Salari R, et al. Phylogenetic ctDNA analysis depicts early-stage lung cancer evolution. Nature. 2017;545:446–51. https://doi.org/10.1038/nature22364
Zeng H, Qu J, Jin N, Xu J, Lin C, Chen Y, et al. Feedback activation of leukemia inhibitory factor receptor limits response to histone deacetylase inhibitors in breast cancer. Cancer Cell. 2016;30:459–73. https://doi.org/10.1016/j.ccell.2016.08.001
Acknowledgements
This work was supported by grants to XXT and WGF from the National Natural Science Foundation of China (No 81872382 and 81621063).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Yang, H., Geng, YH., Wang, P. et al. Extracellular ATP promotes breast cancer invasion and chemoresistance via SOX9 signaling. Oncogene 39, 5795–5810 (2020). https://doi.org/10.1038/s41388-020-01402-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-020-01402-z
This article is cited by
-
An emerging master inducer and regulator for epithelial-mesenchymal transition and tumor metastasis: extracellular and intracellular ATP and its molecular functions and therapeutic potential
Cancer Cell International (2023)
-
P2X1 enhances leukemogenesis through PBX3-BCAT1 pathways
Leukemia (2023)
-
Direct and indirect effects of IFN-α2b in malignancy treatment: not only an archer but also an arrow
Biomarker Research (2022)
-
Extracellular ATP promotes breast cancer chemoresistance via HIF-1α signaling
Cell Death & Disease (2022)
-
Lighting up ATP in cells and tissues using a simple aptamer-based fluorescent probe
Microchimica Acta (2021)