Abstract
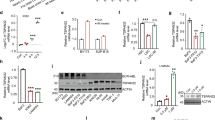
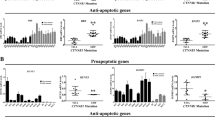
Aberrant activation of Wnt/β-catenin signaling pathway is essential for the development of AML; however, the mechanistic basis for this dysregulation is unclear. PRL-3 is an oncogenic phosphatase implicated in the development of LSCs. Here, we identified Leo1 as a direct and specific substrate of PRL-3. Serine-dephosphorylated form of Leo1 binds directly to β-catenin, promoting the nuclear accumulation of β-catenin and transactivation of TCF/LEF downstream target genes such as cyclin D1 and c-myc. Importantly, overexpression of PRL-3 in AML cells displayed enhanced sensitivity towards β-catenin inhibition in vitro and in vivo, suggesting that these cells are addicted to β-catenin signaling. Altogether, our study revealed a novel regulatory role of PRL-3 in the sustenance of aberrant β-catenin signaling in AML. PRL-3 may serve as a biomarker to select for the subset of AML patients who are likely to benefit from treatment with β-catenin inhibitors. Our study presents a new avenue of cancer inhibition driven by PRL-3 overexpression or β-catenin hyperactivation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bonnet D, Dick JE. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med. 1997;3:730–7.
Reya T, Duncan AW, Ailles L, Domen J, Scherer DC, Willert K, et al. A role for Wnt signalling in self-renewal of haematopoietic stem cells. Nature. 2003;423:409–14.
Mikesch JH, Steffen B, Berdel WE, Serve H, Muller-Tidow C. The emerging role of Wnt signaling in the pathogenesis of acute myeloid leukemia. Leukemia. 2007;21:1638–47.
Chicanne YLG, Demur C, De Toni F, Prade-Houdellier N, Ruidavets JB, et al. Expression of beta-catenin by acute myeloid leukemia cells predicts enhanced clonogenic capacities and poor prognosis. Leukemia. 2006;20:1211–6.
Chung EJ, Hwang SG, Nguyen P, Lee S, Kim JS, Kim JW, et al. Regulation of leukemic cell adhesion, proliferation, and survival by beta-catenin. Blood. 2002;100:982–90.
Zeng Q, Si X, Horstmann H, Xu Y, Hong W, Pallen CJ. Prenylation-dependent association of protein-tyrosine phosphatases PRL-1, -2, and -3 with the plasma membrane and the early endosome. J Biol Chem. 2000;275:21444–52.
Al-Aidaroos AQ, Zeng Q. PRL-3 phosphatase and cancer metastasis. J Cell Biochem. 2010;111:1087–98.
Zeng Q, Dong JM, Guo K, Li J, Tan HX, Koh V, et al. PRL-3 and PRL-1 promote cell migration, invasion, and metastasis. Cancer Res. 2003;1:63. 2716-22
Wang H, Vardy LA, Tan CP, Loo JM, Guo K, Li J, et al. PCBP1 suppresses the translation of metastasis-associated PRL-3 phosphatase. Cancer Cell. 2010;18:52–62.
Zhou JB, CL Bi, WJ Chng, LL Cheong, SC Liu, S Mahara et al. PRL-3, a metastasis associated tyrosine phosphatase, is involved in FLT3-ITD signaling and implicated in Anti-AML therapy. Plos ONE. 2011;6:e19798.
Beekman R, Valkhof M, Erkeland SJ, Taskesen E, Rockova V, Peeters JK, et al. Retroviral integration mutagenesis in mice and comparative analysis in human AML identify reduced PTP4A3 expression as a prognostic indicator. PLoS One. 2011;6:e26537.
Chong PS, Zhou J, Cheong LL, Liu SC, Qian J, Guo T, et al. LEO1 is regulated by PRL-3 and mediates its oncogenic properties in acute myelogenous leukemia. Cancer Res. 2014;74:3043–53.
Saito Y, Kitamura H, Hijikata A, Tomizawa-Murasawa M, Tanaka S, Takagi S, et al. Identification of therapeutic targets for quiescent, chemotherapy-resistant human leukemia stem cells. Sci Transl Med. 2010;2:17ra19.
Patterson KI, Brummer T, O’Brien PM, Daly RJ. Dual-specificity phosphatases: critical regulators with diverse cellular targets. Biochem J. 2009;15:418. 475–89
McParland V, Varsano G, Li X, Thornton J, Baby J, Aravind A, et al. The metastasis-promoting phosphatase PRL-3 shows activity toward phosphoinositides. Biochemistry. 2011;6:50. 7579–90
Mosimann C, Hausmann G, Basler K. Parafibromin/Hyrax activates Wnt/Wg target gene transcription by direct association with beta-catenin/Armadillo. Cell. 2006;125:327–41.
Chaudhary K, Deb S, Moniaux N, Ponnusamy MP, Batra SK. Human RNA polymerase II-associated factor complex: dysregulation in cancer. Oncogene. 2007;29:26. 7499–507
Lepourcelet M, Chen YN, France DS, Wang H, Crews P, Petersen F, et al. Small-molecule antagonists of the oncogenic Tcf/beta-catenin protein complex. Cancer Cell. 2004;5:91–102.
Luu HH, Zhang R, Haydon RC, Rayburn E, Kang Q, Si W, et al. He. Wnt/beta-catenin signaling pathway as a novel cancer drug target. Curr Cancer Drug Targets. 2004;4:653–71.
Gandillet A, Park S, Lassailly F, Griessinger E, Vargaftig J, Filby A, et al. Heterogeneous sensitivity of human acute myeloid leukemia to beta-catenin down-modulation. Leukemia. 2011;25:770–80.
Müller-Tidow C, Steffen B, Cauvet T, Tickenbrock L, Ji P, Diederichs S, et al. Translocation products in acute myeloid leukemia activate the Wnt signaling pathway in hematopoietic cells. Mol Cell Biol. 2004;24:2890–904.
Krieghoff E, Behrens J, Mayr B. Nucleo-cytoplasmic distribution of beta-catenin is regulated by retention. J Cell Sci. 2006;119(Pt 7):1453–63.
Gilliland DG. Molecular genetics of human leukemias: new insights into therapy. Semin Hematol. 2002;39(4 Suppl 3):6–11.
Reindl C, Bagrintseva K, Vempati S, Schnittger S, Ellwart JW, Wenig K, et al. Point mutations in the juxtamembrane domain of FLT3 define a new class of activating mutations in AML. Blood. 2006;107:3700–7.
Simon M, Grandage VL, Linch DC, Khwaja A. Constitutive activation of the Wnt/beta-catenin signalling pathway in acute myeloid leukaemia. Oncogene. 2005;24:2410–20.
Park JE, Yuen HF, Zhou JB, Al-Aidaroos AQ, Guo K, Valk PJ, et al. Oncogenic roles of PRL-3 in FLT3-ITD induced acute myeloid leukaemia. EMBO Mol Med. 2013;5:1351–66.
Acknowledgements
This work was supported by the Singapore National Research Foundation and the Ministry of Education under the Research Center of Excellence Program to WJ Chng and NMRC Clinician-Scientist IRG Grant CNIG11nov38 to Zhou J. WJ Chng is supported by NMRC Singapore Translational Research (STaR) Investigatorship. This research is partly supported by the National Research Foundation Singapore and the Singapore Ministry of Education under the Research Centers of Excellence initiative as well as the RNA Biology Center at the Cancer Science Institute of Singapore, NUS, as part of funding under the Singapore Ministry of Education’s Tier 3 grants, grant number MOE2014-T3-1-006. No potential conflicts of interest were disclosed. We would also like to thank Prof Phil Koeffler, Prof Dan Tenen, Prof Toshio Suda, and Dr Motomi Osato for helpful discussions and critical reading of the manuscript.
Author contributions
PSY Chong, J Zhou, WJ Chng conceptualized and designed the experiments. PSY Chong, Chooi JY, EZL Chan, SHM Toh conducted the experiments and mouse work. S. Wee, J Gunaratne performed the MS analysis and bioinformatics. Zeng Q. provided material support. PSY Chong, J Zhou, WJ Chng wrote and reviewed the paper. None of the material has been published or is under consideration for publication elsewhere.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Chong, P.S.Y., Zhou, J., Chooi, JY. et al. Non-canonical activation of β-catenin by PRL-3 phosphatase in acute myeloid leukemia. Oncogene 38, 1508–1519 (2019). https://doi.org/10.1038/s41388-018-0526-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-018-0526-3
This article is cited by
-
Super-enhancer-driven TOX2 mediates oncogenesis in Natural Killer/T Cell Lymphoma
Molecular Cancer (2023)
-
MYC: a multipurpose oncogene with prognostic and therapeutic implications in blood malignancies
Journal of Hematology & Oncology (2021)
-
Elevated DHODH expression promotes cell proliferation via stabilizing β-catenin in esophageal squamous cell carcinoma
Cell Death & Disease (2020)