Abstract
Objectives
Diabetes-specific nutritional formulas (DSNFs) are frequently used by patients with type 2 diabetes (T2D) as part of nutrition therapy to improve glycemic control and reduce body weight. However, their effects on hunger and satiety hormones when compared to an isocaloric standardized breakfast are not fully understood. This study aims to evaluate the postprandial effects of two DSNFs—Glucerna (GL) and Ultra Glucose Control (UGC)—versus oatmeal on selected satiety and hunger hormones.
Method
After an overnight fast, 22 patients with T2D (mean age 62.3 ± 6.8 years, A1C 6.8 ± 0.7%, body weight 97.4 ± 21.3 kg, and BMI 33.2 ± 5.9 kg/m²) were given 200 kcal of each meal on three separate days. Blood samples for amylin, cholecystokinin (CCK), ghrelin, glucagon, leptin, and peptide-YY (PYY) were collected at baseline and 30, 60, 90, 120, 180, and 240 min after the start of each meal. Incremental area under the curve (iAUC0-240) for each hormone was calculated.
Results
iAUC0-240 for glucagon and PYY were significantly higher after GL and UGC than after oatmeal (p < 0.001 for both). No difference was observed between the three meals on postprandial amylin, CCK, ghrelin, and leptin hormones.
Conclusions
Intake of DSNFs significantly increases secretion of PYY and glucagon, two important satiety hormones. While subjective satiety was not directly evaluated, the increased effect on satiety hormones may partially explain the mechanism of body weight loss associated with DSNF use.
Similar content being viewed by others
Introduction
Nutrition therapy and increased physical activity are first-line therapies for patients with type 2 diabetes (T2D)1. Diabetes-specific nutritional formulas (DSNFs) may be used as a component of medical nutrition therapy (MNT) to help in improving glycemic control and reducing body weight2,3. There is evidence that the integration of meal replacement formulas into an MNT plan leads to better compliance with nutrition therapy and greater weight loss compared to patients on an isocaloric MNT plan4. Recently, the American Diabetes Association included DSNFs in its clinical practice recommendations for lifestyle management5. However, the mechanisms by which DSNFs lead to weight loss and improved blood glucose control are not fully understood.
Regulation of appetite is a complex process that involves intricate pathways of hormonal and neuronal signaling6,7. We previously reported that in comparison to an isocaloric oatmeal breakfast, two commercially available DSNFs significantly increased production of postprandial glucagon-like peptide-1 (GLP-1) hormone8. GLP-1 is an incretin hormone which increases insulin secretion and suppresses glucagon secretion, leading to enhanced glycemic control and increased satiety as a result of DSNF consumption9,10.
This study was conducted to explore the effects of two commercially available DSNFs: Glucerna (GL, Abbott Nutrition Inc., Columbus, OH, USA) and Ultra Glucose Control (UGC, Metagenics, Inc., Aliso Viejo, CA, USA) versus a common breakfast food, namely oatmeal (oatmeal, Quaker Old Fashioned Oats, Quaker Oats Co., Chicago, IL, USA) on several other satiety and hunger hormones in overweight and obese patients with T2D.
Subjects and methods
Study design and subjects
This cross-over, three-way, and open-label, ancillary study was conducted in accordance with the Helsinki Declaration and was approved by the institutional Committee on Human Studies. All participants signed a written informed consent. The study was registered at ClinicalTrials.gov (Identifier: NCT02691481). Eligible subjects were patients with T2D for ≥3 months, ages 18 to 75 years, body mass index (BMI) > 25 kg/m2, and glycated hemoglobin A1C (HbA1c) ≥ 6.5%. Patients using diabetes or cholesterol-lowering medications had to be on stable doses of these medications for ≥3 months. Exclusion criteria included pregnancy or lactation, use of insulin or GLP-1 analogs, history of bariatric surgery, gastroparesis, and malabsorption syndrome. Twenty-five subjects were enrolled in the study, of which 22 subjects completed all study visits. One subject dropped out prior to the first study visit for personal reasons. Two other subjects dropped out after the first visit due to inconvenience of frequent blood sampling. Data from those subjects were excluded from statistical analysis. Mean age of the remaining 22 subjects (±SD) was 62.3 ± 6.8 years, diabetes duration was 9.5 ± 9.8 years, and HbA1c was 6.8 ± 0.7%. Baseline characteristics of the study subjects are summarized in Table 1.
Analyzed hormones
Amylin is a satiety hormone that is co-secreted with insulin from pancreatic β-cells11. Its secretion induces satiety by stimulating the brainstem to slow gastric emptying and decrease gastric secretions12,13. Cholecystokinin (CCK) is a satiety hormone secreted by enteroendocrine cells in the duodenum and jejunum14. Its actions include the promotion of gallbladder contraction, inhibition of gastric acid secretion, and slowing of gastric emptying15. Glucagon is secreted by pancreatic alpha cells and induces satiety through the vagus nerve13. Leptin is secreted by adipose tissue and stimulates satiety centers in the hypothalamus16. Peptide-YY3-36 (PYY) is secreted by enteroendocrine L-cells6 and acts as a satiety signal to the hypothalamus while reducing gastric acid secretion and gastrointestinal motility17. Ghrelin is a hunger hormone secreted mainly by the stomach18. Its stimulates gastrointestinal motility and gastric acid secretion19.
Study procedures
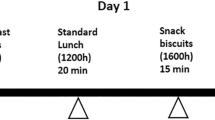
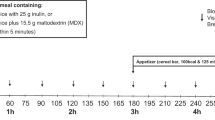
Subjects were asked to come for three visits with a washout period between visits of at least two days. All visits were completed over three weeks. Subjects were instructed to come for each visit after fasting overnight for at least 8 h and were asked to withhold their diabetes and cholesterol-lowering medications on the morning of the visit. In random visit order, each subject was asked to ingest one of the three tested meals (GL, UGC and oatmeal) for breakfast. All meals were 200 kcal each. GL was provided in a 237 mL (8 fl oz) bottle; UGC was prepared by dissolving 200 kcal powder in 296 mL (10 fl oz) of water; and oatmeal was prepared by adding water to 56 g of dry oats and cooking the mixture on a stove for 5–10 min. No milk, sugar, or sweetener was added. Macronutrient composition of the three breakfast meals is shown in Table 2.
For safety, blood glucose was measured at the beginning of each visit. If blood glucose was between 70–300 mg/dL, a venous line was inserted and a baseline blood sample was drawn. This was followed by consumption of the test meal within 3–5 min. Blood samples were collected at 30, 60, 90, 120, 180, and 240 min from the start of each meal. Blood samples were tested for serum active amylin, CCK, ghrelin, glucagon, leptin, and PYY. After collection of the last sample, subjects were given a snack and were instructed to take their regular medications.
Statistical analyses
Values for all measured variables are presented as mean ± SD or standard error of the mean (SEM). Study data were analyzed using SAS statistical software (SAS Institute Inc., Cary, NC, USA). Analysis was performed using linear mixed effects models to model the covariance structure arising from the repeated measures design. Where overall F-tests were significant, pairwise differences of the treatment means were tested with t-tests using Tukey’s p-value adjustments. Outcomes were defined as area under the curve between 0 and 240 min for measured variables over time (AUC0–240) calculated using the trapezoidal formula20. Incremental AUC between 0 and 240 min (iAUC0–240) was calculated using the same formula but representing the area above the fasting level.
Results
Mean fasting serum glucagon for oatmeal, GL, and UGC were similar (35.8 ± 4.4, 41.9 ± 4.7, and 34.5 ± 4.6 pg/mL respectively). Glucagon iAUC0-240 was significantly higher after GL and UGC compared to oatmeal (p < 0.0001 for both); however, there was no difference in glucagon iAUC0–240 between GL and UGC (Fig. 1).
Mean fasting serum PYY for oatmeal, GL, and UGC were similar (72.2 ± 7.2, 76.5 ± 7.8, and 68.7 ± 8.3 pg/mL respectively). PYY iAUC0–240 was significantly higher after GL and UGC compared to oatmeal (p < 0.0001 for both); however, there was no difference in PYY iAUC0–240 between GL and UGC (Fig. 1).
Mean fasting serum active amylin for oatmeal, GL, and UGC were similar (10.5 ± 1.6, 9.9 ± 1.4, and 8.9 ± 1.3 pg/mL respectively). Active amylin iAUC0-240 showed no significant differences between meals (p = 0.076) (Fig. 2).
Mean fasting serum CCK for oatmeal, GL, and UGC were similar (38.8 ± 18.7, 33.8 ± 18.2, and 37.8 ± 16.0 pg/mL respectively). CCK iAUC0–240 showed no significant differences between meals (p = 0.85) (Fig. 2).
Mean fasting serum ghrelin for oatmeal, GL, and UGC were similar (9.1 ± 1.6, 10.0 ± 1.6, and 10.3 ± 2.1 pg/mL respectively). Ghrelin iAUC0–240 showed no significant differences between meals (p = 0.82) (Fig. 2).
Mean fasting serum leptin for oatmeal, GL, and UGC were similar (25224.9 ± 4273.6, 23649.5 ± 3820.1, and 24790.2 ± 4012.4 pg/mL respectively). Leptin iAUC0–240 showed no significant differences between meals (p = 0.87) (Fig. 2).
Discussion
In the Look AHEAD (Action for Health in Diabetes) study3 and other shorter studies4,21, use of DSNFs as part of a hypocaloric nutrition therapy was associated with weight reduction that was clearly associated with their frequency of use to replace calories or smaller meals. This study provides a mechanistic explanation of that effect, where two of the commercially available DSNFs showed significant increase in two essential weight-modulating hormones that contribute to satiety and increased energy expenditure. Both tested DSNFs increased PYY in comparison to isocaloric oatmeal. This study also showed that both DSNFs significantly stimulate glucagon secretion in comparison to isocaloric oatmeal. Glucagon affects glycemia and satiety. Despite its stimulatory effect on hepatic glucose production, glucagon hormone increases glucose metabolism, and energy expenditure22,23. In addition, glucagon indirectly stimulates satiety through an afferent signal from the hepatic branch of the vagus nerve24. These observations complement our previous observation that both DSNFs stimulate GLP-1 hormone production, another strong satiety hormone, in comparison to isocaloric oatmeal8.
Despite previous claims that all calories are created equal in their effect on body weight25, this study shows that different macronutrients have different effects on key satiety and weight-modulating hormones since all tested meals were of equal caloric content. The two studied DSNFs are higher in their protein and fat content and lower in their carbohydrate content than oatmeal (Table 2). It has been debated which macronutrient(s) elicit the highest postprandial PYY response. An earlier study favored fat in producing the highest PYY response26. However, more recent studies showed that protein induces the highest PYY response27 and carbohydrates induce the smallest effect28. Our results are also in line with previous studies that showed meals higher in both protein and fat content induce higher glucagon response compared to a carbohydrate-rich meal24,29.
Although both tested DSNFs stimulate two opposing weight-modulating hormones, GLP-18 and glucagon, our findings suggest that the stimulatory effect of protein and fat within DSNFs is stronger on glucagon secretion than the inhibitory effect of GLP-1 on glucagon production. Postprandial amylin levels were marginally higher following ingestion of UGC compared to GL and oatmeal, but this difference was not statistically significant (p = 0.076). Furthermore, there were no differences in the postprandial effects of DSNFs on CCK, ghrelin, and leptin hormones. While these changes in satiety hormones provide an attractive potential explanation for the reported success of DSNFs in supporting weight loss, it is also possible that these changes in the satiety hormones, while statistically significant, may not be of sufficient magnitude to explain an effect on satiety that is large enough to interpret their role in improved weight loss.
The present study had several limitations which include the difference in texture between oatmeal and DSNFs. A previous study reported difference in satiety between solid and liquid meal replacements30. This study was powered to detect differences in glucose AUC0–240 rather than differences in the analyzed hunger and satiety hormones. Background diets of the study subjects were not controlled and their effect on the study outcomes is unknown. We aimed to minimize that effect by asking subjects to fast for at least 8 h before each visit. In addition, subjects completed all study visits within a three-week window.
In conclusion, this study shows that DSNFs significantly increase secretion of two satiety hormones; PYY and glucagon. This effect may be related to their specific macronutrient composition. While the effect of the three different meals on subjective satiety was not directly evaluated, results from this study may partially explain the mechanism of body weight reduction associated with DSNFs use.
References
Nathan, D. M. et al. Medical management of hyperglycaemia in type 2 diabetes mellitus: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia 52, 17–30 (2009).
Elia, M. et al. Enteral Nutritional Support and Use of Diabetes-Specific Formulas for Patients With Diabetes A systematic review and meta-analysis. Diabetes Care 28, 2267–2279 (2005).
Wadden, T. A. et al. Four-year weight losses in the Look AHEAD study: factors associated with long-term success. Obesity 19, 1987–1998 (2011).
Heymsfield, S., Van Mierlo, C., Van der Knaap, H., Heo, M. & Frier, H. Weight management using a meal replacement strategy: meta and pooling analysis from six studies. Int. J. Obes. 27, 537 (2003).
Association, A. D. 5. Lifestyle Management: Standards of Medical Care in Diabetes—2019. Diabetes Car 42, S46–S60 (2019).
Cummings, D. E. & Overduin, J. Gastrointestinal regulation of food intake. J. Clin. Investig. 117, 13–23 (2007).
Ahima, R. S. & Antwi, D. A. Brain regulation of appetite and satiety. Endocrinol. Metab. Clin. North Am. 37, 811–823 (2008).
Mottalib, A. et al. Impact of diabetes-specific nutritional formulas versus oatmeal on postprandial glucose, insulin, GLP-1 and postprandial lipidemia. Nutrients 8, 443 (2016).
Flint, A., Raben, A., Astrup, A. & Holst, J. J. Glucagon-like peptide 1 promotes satiety and suppresses energy intake in humans. J. Clin. Investig. 101, 515–520 (1998).
Nadkarni, P., Chepurny, O. G. & Holz, G. G. Regulation of glucose homeostasis by GLP-1. Prog. Mol. Biol. Transl. Sci. 121, 23–65 (2014).
Lukinius, A., Wilander, E., Westermark, G. T., Engstrom, U. & Westermark, P. Co-localization of islet amyloid polypeptide and insulin in the B cell secretory granules of the human pancreatic islets. Diabetologia 32, 240–244 (1989).
Young, A. A. Brainstem sensing of meal-related signals in energy homeostasis. Neuropharmacology 63, 31–45 (2012).
Woods, S. C., Lutz, T. A., Geary, N. & Langhans, W. Pancreatic signals controlling food intake; insulin, glucagon and amylin. Philos. Trans. R. Soc. Lond. Ser. B, Biol. Sci. 361, 1219–1235 (2006).
Kissileff, H. R., Pi-Sunyer, F. X., Thornton, J. & Smith, G. P. C-terminal octapeptide of cholecystokinin decreases food intake in man. Am. J. Clin. Nutr. 34, 154–160 (1981).
Chaudhri, O., Small, C. & Bloom, S. Gastrointestinal hormones regulating appetite. Philos. Trans. R. Soc. Lond. Ser. B, Biol. Sci. 361, 1187–1209 (2006).
Myers, M. G., Cowley, M. A. & Munzberg, H. Mechanisms of leptin action and leptin resistance. Annu. Rev. Physiol. 70, 537–556 (2008).
le Roux, C. W. & Bloom, S. R. Peptide YY, appetite and food intake. Proc. Nutr. Soc. 64, 213–216 (2005).
Cummings, D. E. et al. A preprandial rise in plasma ghrelin levels suggests a role in meal initiation in humans. Diabetes 50, 1714–1719 (2001).
Cummings, D. E. Ghrelin and the short- and long-term regulation of appetite and body weight. Physiol. Behav. 89, 71–84 (2006).
Pruessner, J. C., Kirschbaum, C., Meinlschmid, G. & Hellhammer, D. H. Two formulas for computation of the area under the curve represent measures of total hormone concentration versus time-dependent change. Psychoneuroendocrinology 28, 916–931 (2003).
Cheskin, L. J. et al. Efficacy of meal replacements versus a standard food-based diet for weight loss in type 2 diabetes: a controlled clinical trial. Diabetes Educ. 34, 118–127 (2008).
Jiang, G. & Zhang, B. B. Glucagon and regulation of glucose metabolism. Am. J. Physiol. Endocrinol. Metab. 284, E671–E678 (2003).
Salem, V. et al. Glucagon increases energy expenditure independently of brown adipose tissue activation in humans. Diabetes, Obes. Metab. 18, 72–81 (2016).
Geary, N. Pancreatic glucagon signals postprandial satiety. Neurosci. Biobehav. Rev. 14, 323–338 (1990).
Sacks, F. M. et al. Comparison of weight-loss diets with different compositions of fat, protein, and carbohydrates. N. Engl. J. Med. 360, 859–873 (2009).
Essah, P. A., Levy, J. R., Sistrun, S. N., Kelly, S. M. & Nestler, J. E. Effect of macronutrient composition on postprandial peptide YY levels. J. Clin. Endocrinol. Metab. 92, 4052–4055 (2007).
van der Klaauw, A. A. et al. High protein intake stimulates postprandial GLP1 and PYY release. Obesity 21, 1602–1607 (2013).
Cooper, J. A. Factors affecting circulating levels of peptide YY in humans: a comprehensive review. Nutr. Res. Rev. 27, 186–197 (2014).
Raben, A., Agerholm-Larsen, L., Flint, A., Holst, J. J. & Astrup, A. Meals with similar energy densities but rich in protein, fat, carbohydrate, or alcohol have different effects on energy expenditure and substrate metabolism but not on appetite and energy intake. Am. J. Clin. Nutr. 77, 91–100 (2003).
Tieken, S. M. et al. Effects of solid versus liquid meal-replacement products of similar energy content on hunger, satiety, and appetite-regulating hormones in older adults. Horm. Metab. Res. 39, 389–394 (2007).
Acknowledgements
This is an investigator-initiated study funded by Metagenics, Inc. Metagenics, Inc. had no role in the study design, conduct, preparation of the study manuscript, or presentation of the study results. All study data and study intellectual properties are owned by the study investigators and Joslin Diabetes Center, Boston, MA. Data from this study were presented at the 76th Annual Conference of the American Diabetes Association, June 2016, New Orleans, LA, USA.
Author information
Authors and Affiliations
Contributions
Authors’ responsibilities: A.M. and O.H. designed the study, wrote the manuscript, were the guarantors of this work and, as such, had full access to all of the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis; M.J.A. reviewed and edited the manuscript; D.M.P. analyzed data and wrote the statistical analyses section; R.P. assisted in conducting study visits, data entry, and reviewed and edited the manuscript; A.H.E., S.A. and S.T. reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
O.H. is on the advisory board of AstraZeneca and Sanofi-Aventis U.S. LLC and is a consultant to Merck & Co., Inc. and Abbott Nutrition. He is a shareholder of Healthimation, Inc. and receives research grants from the National Dairy Council and Novo Nordisk. M.J.A. is on the advisory boards of Novo Nordisk and WebMD Health Services and owns stock in Health IQ. A.M., D.M.P., R.P., A.H.E., S.T. and S.A. declare no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mottalib, A., Abrahamson, M.J., Pober, D.M. et al. Effect of diabetes-specific nutrition formulas on satiety and hunger hormones in patients with type 2 diabetes. Nutr. Diabetes 9, 26 (2019). https://doi.org/10.1038/s41387-019-0093-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41387-019-0093-x
This article is cited by
-
Impact of Oats on Appetite Hormones and Body Weight Management: A Review
Current Nutrition Reports (2023)