Abstract
Major histocompatibility complex class II (MHCII) is dynamically expressed on intestinal epithelial cells (IECs) throughout the intestine, but its regulation remains poorly understood. We observed that spontaneous upregulation of IEC MHCII in locally bred Rag1−/− mice correlated with serum Interleukin (IL)-18, was transferrable via co-housing to commercially bred immunodeficient mice and could be inhibited by both IL-12 and IL-18 blockade. Overproduction of intestinal IL-18 due to an activating Nlrc4 mutation upregulated IEC MHCII via classical inflammasome machinery independently of immunodeficiency or dysbiosis. Immunodeficient dysbiosis increased Il-18 transcription, which synergized with NLRC4 inflammasome activity to drive elevations in serum IL-18. This IL-18-MHCII axis was confirmed in several other models of intestinal and systemic inflammation. Elevated IL-18 reliably preceded MHCII upregulation, suggesting an indirect effect on IECs, and mice with IL-18 overproduction showed activation or expansion of type 1 lymphocytes. Interferon gamma (IFNg) was uniquely able to upregulate IEC MHCII in enteroid cultures and was required for MHCII upregulation in several in vivo systems. Thus, we have linked intestinal dysbiosis, systemic inflammation, and inflammasome activity to IEC MHCII upregulation via an intestinal IL-18-IFNg axis. Understanding this process may be crucial for determining the contribution of IEC MHCII to intestinal homeostasis, host defense, and tolerance.
Similar content being viewed by others
Introduction
The mammalian intestinal epithelium is a heterogeneous single-cell barrier that separates host tissues from a complex microbial ecosystem. It must shepherd vital nutrients systemically from the lumen, exclude commensal microbes, repair itself, and mobilize appropriate immune responses to intestinal pathogens. Among the greatest challenges this barrier faces is negotiating the balance between host defense and immune tolerance. Notably, antigen presentation is critical to both outcomes.
Though the expression of major histocompatibility complex class II (MHCII) is canonically reserved for “professional” antigen presenting cells, like B- and dendritic cells (DCs), intestinal epithelial cells (IECs) are also capable of processing peptides and expressing them on surface MHCII1,2. IEC MHCII appears to be expressed on both luminal and basolateral surfaces, and on a wide variety of IECs from the duodenum through the colon2. Both murine and human studies concur that elevated colonic MHCII expression is observed in gut inflammatory disorders including colitis and graft-versus-host disease (GvHD), where local production of interferon gamma (IFNg) by intestinal T cells appears sufficient to upregulate IEC MHCII1,2,3,4,5.
Despite broad appreciation for the phenomenon, little is known about its regulation or contributions to barrier integrity, host defense, and tolerance. The conventional view of intestinal antigen presentation suggests that DCs acquire antigen and migrate to Peyer’s Patches and mesenteric lymph nodes to initiate immune responses to food-, commensal-, and pathogen-derived antigens6,7. Recent studies using conditional deletion of MHCII have largely corroborated this view. For example, intestinal CD11c + DCs, and not IECs, were found to be required for TH17 responses to Segmented Filamentous Bacteria (SFB) colonization, as well as bacterial driven-T cell colitis8,9.
However, other studies suggest exceptions to the “professional” APC paradigm in the intestine. MHCII expression by Rorc expressing cells was required to prevent the development of spontaneous IBD in a microbiota-dependent manner10. The specific role for MHCII expression by IECs remains controversial. Biton et al. showed that IEC MHCII expression was important for epithelial remodeling post infection11. Koyama et al. recently identified that ileal IEC MHCII expression promoted intestinal graft-versus-host responses to cytosolic alloantigens via the microbiota, IL-12, and IFNg1. Consistent with a role for IEC MHCII in gut T-cell activation, mice selectively lacking IEC MHCII developed both more severe infectious (C. rodentium) colitis as well as worsened T-cell transfer or IL-10 receptor blockade colitis (the latter requiring H. hepaticus)4,5.
MHCII expression on traditional APCs can be stimulated by various cytokines and chemokines12. Other than its association with IFNg, little is known about the regulation of IEC MHCII2,12. Specifically, the conditions, chronology, and upstream events leading to IEC MHCII remain largely unknown. Herein, we show that Rag1 deficiency is associated with a transferable dysbiosis that drives MHCII expression on both small and large IECs. IL-18 and IL-12 are necessary drivers for this IEC MHCII expression. Various genetic models of IL-18 overproduction show that constitutive IL-18 can itself (independently of dysbiosis or immunodeficiency) drive MHCII and supports a role for inflammasome machinery in the regulation of both serum IL-18 and SI IEC MHCII. Specifically, our results support a collaborative model in which microbial induction of Il-18 cooperates with inflammasome-mediated proIL-18 maturation to drive changes in serum IL-18, expansion of innate and adaptive type 1 lymphocytes, and IFNg-mediated IEC MHCII. These models, as well as models of systemic inflammation and commensal protist colonization, elucidate a novel mechanism of IEC MHCII regulation via an intestinal IL-18-IL-12/IFNg axis.
Results
Rag1 deficiency is associated with a microbiota-dependent increase in small and large intestinal epithelial MHCII
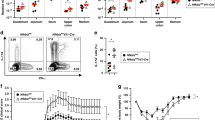
Inducible expression of MHCII has been reported on murine small and large IECs in a variety of circumstances4,5,9. We observed consistently high MHCII expression on Epcam+ colonic and small intestinal (SI) epithelial cells in Rag1−/− mice relative to wild-type (WT) controls (Fig. 1a and Supplementary Fig. 1). SI IEC MHCII was sometimes observed to be elevated in unstimulated WT mice, suggesting greater sensitivity of SI IECs vs. colonic IECs. Treatment of Rag1−/− mice with broad spectrum antibiotics normalized colonic MHCII expression (Fig. 1b). Given this microbial association, we compared IEC MHCII in Rag1−/− mice bred in our colony (Pitt) vs. mice imported directly from a commercial source (Jackson Laboratory). Imported Rag1−/− mice showed neither colonic nor SI IEC MHCII expression, suggesting a commensal inducer of IEC MHCII present uniquely in our colony (Fig. 1c). Though stool from Rag1−/− mice in our colony contained DNA from SFB (a vancomycin-sensitive commensal that can induce IEC MHCII expression), treatment with vancomycin alone did not diminish IEC MHCII (data not shown)13.
gMFI and representative histograms of colonic and/or SI epithelial cell MHCII in Rag1−/− vs. C57Bl/6J mice at baseline (a), following 3 weeks of broad spectrum antibiotics (b), and in commercially bred (Jackson) vs. locally bred (Pitt) Rag1−/− mice (c). Combined results of 2-(b) or 3-(a) independent experiments with 3–5 mice per group. c Representative of three independent experiments with 2–3 mice per experimental group. Statistical significance is shown on each graph and was determined by unpaired t tests. Abx antibiotics, gMFI geometric mean fluorescent intensity, MANV antibiotics (metronidazole, ampicillin, neomycin, vancomycin), Unt untreated (no antibiotics).
Elevated IEC MHCII is transferable to immunodeficient, but not immunocompetent hosts
To determine the nature of microbiota-driven IEC MHCII expression in Pitt Rag1−/− mice, we co-housed them for 5 weeks with imported Jackson Rag1−/− mice. Co-housing resulted in a significant increase in IEC MHCII (Fig. 2a). To identify unique microbial factors driving changes in MHCII, we performed 16S sequencing on cecal stool. Principal component analysis demonstrated an obvious shift in the Jackson Rag1−/− microbiome towards that of Pitt Rag1−/− cage mates (Fig. 2b). Analyzing the specific genera acquired or lost by Rag1−/− mice imported and co-housed (versus imported and housed separately), we found that co-housing was associated with increased abundance of Paraprevotellacea and Helicobacteraceae, and to a lesser extent Deferribacteraceae (Fig. 2c). These bacteria are frequently bound by IgA, which is absent in Rag1−/− mice14,15.
Locally (Pitt) and commercially bred (Jackson) Rag1−/− mice were kept isolated or co-housed for 5 weeks. At sacrifice, mice were analyzed for epithelial MHCII expression by flow cytometry (a). Each dot represents a single mouse and is representative of two independent experiments with 2–3 mice per condition. Cecal stool was collected and its DNA analyzed by 16S analysis. Jaccard principal coordinate analysis plots of cecal fecal diversity (b) and family diversity (c), for each mouse and is representative of a single experiment. gMFI geometric mean fluorescent intensity, PC principal coordinate.
To investigate whether the transfer of the MHCII phenotype, and its microbial correlates, were unique to Rag1−/− mice, we housed locally bred WT mice separately or with Pitt Rag1−/− mice. After 5 weeks, WT mice had normal colonic MHCII regardless of their housing partners, suggesting induction of IEC MHCII by transferred commensal bacteria occurred only in immunodeficient hosts (Supplementary Fig. 2a). This effect was obscured in the SI by spontaneous MHCII upregulation in some WT mice. Though co-housed WT mice did not upregulate colonic MHCII, 16S analysis of their cecal contents demonstrated a shift toward the Rag1−/− colonization pattern, with increases in the relative abundance of Helicobacteraceae, Paraprevotellacea, and Deferribacteracea upon co-housing (Supplementary Fig. 2b, c). Overall, these data suggest that colonization of Rag1-deficient (but not WT) mice with certain commensal bacteria stably upregulates colonic MHCII.
MHCII upregulation correlates with and requires IL-18
We have previously shown a connection between IEC IL-18 production and SI IEC MHCII transcription16. Therefore, we assessed serum IL-18 in Rag1−/− mice and noted elevated serum IL-18 relative to WT controls (Fig. 3a). Consistent with our MHCII findings, treatment of Rag1−/− mice with broad spectrum antibiotics returned serum IL-18 to baseline, suggesting that microbial dysbiosis drove both IEC MHCII and elevated IL-18 (Fig. 3b). Congruent with our MHCII findings, we also observed that locally bred Rag1−/− mice had elevated levels of serum IL-18 relative to imported Jackson mice (Fig. 3c). Upon co-housing, serum IL-18 in imported Rag1−/−, but not WT mice, rose to that of locally bred mice, suggesting that IL-18 was induced by a combination of dysbiosis and immunodeficiency and was necessary for MHCII expression (Fig. 3d and Supplementary Fig. 2d). To confirm a direct effect of IL-18 on MHCII, we treated Rag1−/− mice with an IL-18 receptor blocking antibody and found reduced colonic MHCII expression (Fig. 3e). IL-18 canonically acts in synergy with other cytokines, particularly IL-12, to promote type I cytokine production and cytotoxicity17. Consistent with this interaction, a similar downregulation of colonic MHCII was seen following IL-12 blockade (Fig. 3f). SI MHCII was altered by neither IL-18R nor IL-12 blockade (Supplementary Fig. 3a, b). Thus, IL-18 and IL-12 are necessary contributors for microbially driven colonic IEC MHCII expression in Rag1−/− mice.
Serum IL-18 in Rag1−/− mice at baseline (a), following antibiotic treatment (b) and in commercially (Jackson) vs. locally bred (Pitt) mice without (c) or with (d) co-housing (paired analysis). Rag−/− mice were injected every 3 days with IL-18R (e) or IL-12 (f) blocking antibodies or isotype control and colons analyzed for epithelial MHCII expression. Figures are combined results from three of five experiments (a), n = 2 experiments (b, e, f) or representative of n = 2 experiments (c) with 3–5 mice per condition. To correct for inter-experiment variation in e, gMFI was normalized for each sample to the mean of that experiment’s isotype control group. Statistical significance is shown on each graph and was determined by unpaired t test (n = 2 groups) or one-way ANOVA (n = 3 groups). Abx antibiotics, gMFI geometric mean fluorescent intensity, MANV antibiotics (Metronidazole, ampicillin, neomycin, vancomycin), Unt untreated (no antibiotics).
Genetic models of IL-18 overproduction cause SI MHCII upregulation independently of the microbiome or immunodeficiency
We have previously shown that mice bearing an activating mutation in the NLRC4 inflammasome (Nlrc4TS/TS) exhibited increased serum IL-18 derived from IECs16. SI IECs from these mice also showed increased proliferation and transcription of genes related to MHCII antigen presentation. We readily detected spontaneous IEC MHCII upregulation on Epcam+ SI IECs (and to a lesser extent colonic IECs) from Nlrc4TS/TS mice (Fig. 4a). Contrasting with Rag1−/− mice, Nlrc4TS/TS mice treated with broad spectrum antibiotics (MANV) showed neither diminution of their serum IL-1816 nor a decrease in SI MHCII expression (Fig. 4b). As these mice have neither immunodeficiency nor increased IEC Il-18 transcription16, this suggests that IEC inflammasome activation, and resultant IL-18 maturation, is sufficient for SI IEC MHCII upregulation.
Epithelial MHCII including representative histograms in Nlrc4TS/TS mice at baseline (a), following treatment with broad spectrum antibiotics (b), and with or without deletion of intestinal epithelial Il-18 (c). SI MHCII in Il18Tg/wt mice (d), Il18bp−/− mice (e), and MEFVV726A/V726A mice (including serum IL-18) (f). MEFVV726A/V726A mice were assessed at 4 weeks of age due to spontaneous systemic inflammation arising in adulthood. Samples are SI unless otherwise indicated. Each symbol represents a single mouse. Representative of n ≥ 3 experiments (a, c), combined results of n = 2 experiments (b, d–f) and representative of a single experiment (h). Statistical significance is shown on each graph and is determined by unpaired t test (n = 2 groups). MANV antibiotics (Metronidazole, ampicillin, neomycin, vancomycin), gMFI geometric mean fluorescent intensity, Unt untreated (no antibiotics).
Since IL-18 in Nlrc4TS mice is intestinally derived16, we directly tested this link by assessing MHCII expression in mice specifically lacking the Il-18 gene in Vil1-expressing IECs. Loss of intestinal Il-18 normalized both serum IL-1816 and MHCII (Fig. 4c). As in Rag1−/− mice, antibody-based blockade of the IL-18R or IL-12 failed to affect SI IEC MHCII expression in Nlrc4TS mice (Supplementary Fig. 3c, d).
We hypothesized that other models associated with excess IL-18 would also increase IEC MHCII. We found that mice with transgenic expression of IL-1818, mice lacking IL-18’s endogenous inhibitor IL-18 binding protein (Il18bp−/− mice)19, and mice with genetic hyperactivity of the Pyrin inflammasome (MEFVV726A/V726A)20 all developed elevation of SI MHCII expression, again solidifying the link between IL-18 overproduction and elevated epithelial MHCII (Fig. 4d–f).
Caspase-1 and gasdermin-D are required for basal serum IL-18 and Nlrc4-inflammasome induced elevations of serum IL-18 and IEC MHCII
Elevated serum IL-18 in Nlrc4TS mice is derived from the intestinal epithelium16. To better understand the mechanisms driving epithelial release of IL-18 and subsequent IEC MHCII induction, we evaluated several strains lacking either intestinal Il-18 or key components of the inflammasome pathway. Notably, mice with WT Nlrc4 specifically lacking Il-18 in intestinal epithelia (Il18fl/fl-ΔIEC) show lower IL-18 levels than Cre-negative “WT” controls, suggesting that basal serum IL-18 is intestinally derived (Fig. 5a).
Serum IL-18 and epithelial MHCII including representative histograms in adult mice of the indicated genotypes (a–c). The dotted lines represent the average gMFI of Gasdermin-D or Caspase-1 heterozygous littermate controls. Terminal ileum Il-18 read counts in co-housed, segmented filamentous bacteria (SFB)-colonized WT, Rag−/− and TCRa−/− mice compared to separately housed TCRa−/− mice. Data are derived from Mao et al. 24 and accessed from GSE86780 (d). Serum IL-18 in mice bearing combined Nlrc4 hyperactivating and Rag1 mutations (e). Combined results of n = 4 (a) or n = 2 (e) independent experiments with n > 2 mice per condition. b, c Representative of n = 2 independent experiments with n = 4–6 mice per genotype. Statistical significance is shown on each graph and is determined by unpaired t test (n = 2 groups) or one-way ANOVA (n = 3 groups). gMFI geometric mean fluorescent intensity, Δ IEC Villin-Cre+.
Multiple inflammasomes, caspases, and cell death pathways are present in IECs21, and activation of the NLRC4 inflammasome in IECs can trigger caspase-1 and caspase-8, both of which can cleave inflammasome substrates22. Serum IL-18 in Casp1−/− mice, even in the presence of the NLRC4TS mutation, was below WT levels, and such mice did not upregulate IEC MHCII (Fig. 5b). This suggests there is little/no contribution of caspase-8 (or any other protease) to either basal or Nlrc4TS-enhanced IL-18 production or subsequent IEC MHCII induction. Identical results were obtained in mice lacking the pore-forming inflammasome substrate gasdermin-D23 (Fig. 5c). Thus, IEC IL-18 production, basal and NLRC4-driven serum IL-18, and spontaneous IL-18 dependent MHCII upregulation require the classical inflammasome components caspase-1 and gasdermin-D.
Rag1 deficiency synergizes with NLRC4 hyperactivity in the elevation of serum IL-18
Paradoxically, IEC Il-18 transcription is slightly decreased in Nlrc4TS mice16. As such, their elevated IL-18 arises due to increased inflammasome-mediated proIL-18 cleavage and export. In locally bred Rag1−/− mice, we suspected that microbial dysbiosis drove elevated Il-18 transcription. Indeed, re-analysis of ileal transcription from Mao et al.24 showed that when SFB-colonized WT, TCRa−/−, and Rag1−/− mice were co-housed, only immunodeficient mice upregulated Il-18; separately housed (SFB-naïve) TCRa−/− mice did not upregulate Il-18 (Fig. 5d)24. We reasoned that if increased serum IL-18 in Rag1−/− mice is the product of increased Il-18 transcription, then combining this mechanism with Nlrc4TS-mediated proIL-18 cleavage should result in (at least) additive IL-18 overproduction. Indeed, Nlrc4TS/WT; Rag1−/− mice had approximately tenfold higher serum IL-18 than either parental strain, supporting the complementary nature of these mechanisms (Fig. 5e).
IL-18 dependent upregulation of IEC MHCII occurs broadly in response to systemic inflammation and Tritrichomonas colonization
We postulated that other stimuli associated with changes in local intestinal and systemic IL-18 would also influence IEC MHCII expression. Repeated systemic stimulation through toll-like receptor (TLR) nine results in an inflammatory phenotype reminiscent of human macrophage activation syndrome, which increases serum IL-18 and is dependent on an IL-12-IFNg axis16,25,26,27. Repeated CpG administration induced high expression of MHCII on colonic and SI IECs (Fig. 6a).
Serum IL-18 and gMFI of epithelial MHCII, including representative histograms in CpG or Tritrichomonas-treated mice (a). WT mice were injected every other day with CpG (ip) or actively gavaged with Tritrichomonas-enriched stool. Two weeks post treatment initiation, mice were sacrificed and analyzed for colonic and small intestinal epithelial MHCII expression by flow cytometry (a) and serum IL-18 by cytometric bead array (b). MHCII expression on small and large intestinal epithelial cells in littermates with or without intestinal epithelial IL-18 treated with two CpG injections (c) or colonized with Tritrichomonas for 2 weeks (d). Dotted lines represent unstimulated MHCII in Il18fl/fl controls. Each figure is a representative of two independent experiments and each individual mouse is denoted by a datapoint with 3–5 mice per treatment/genotype. Statistical significance is shown on each graph and was determined by unpaired t test (n = 2 groups) or one-way ANOVA (n = 3 groups). TLR toll-like receptor, gMFI geometric mean fluorescent intensity, Unt untreated, TMu Tritrichomonas colonized, ΔIEC Villin-Cre+.
Colonization with the gut-specific commensal protist Tritrichomonas has been associated with intestinal inflammasome activation and lamina propria T-cell IFNg production28,29 as well as Tuft cell activation and induction of type 2 cytokines30. We gavaged WT mice with Tritrichomonas enriched from the cecal contents of a colonized mouse and noted both extensive establishment of Tritrichomonas colonization in recipient animals and robust upregulation of intestinal (particularly colonic) MHCII (Fig. 6a). Interestingly, Tritichomonas colonization did not appreciably elevate serum IL-18 (Fig. 6b), supporting a remarkably gut-specific immune response. Specific deletion of Il-18 in IECs also resulted in decreased IEC MHCII upregulation with either TLR9 stimulation or Tritrichomonas colonization (Fig. 6c, d), particularly in the colon.
Elevated IL-18 precedes induction of IEC MHCII
Interestingly, elevated IL-18 occurred around weaning in Rag1−/− mice (~3–4 weeks of age), preceding MHCII induction (6 weeks of age) (Supplementary Fig. 4a, b). Despite Nlrc4TS/TS mice elevating serum IL-18 as early as 2 days of age16, they too did not upregulate MHCII until 3–4 weeks of age, suggesting an important role for time/weaning in IL-18-induced MHCII expression (Supplementary Fig. 4c). This was largely replicated in ll18bp−/− mice (Supplementary Fig. 4d), although they took slightly longer to achieve stable and high MHCII elevation. In addition, 10-day-old WT pups treated with CpG showed elevated serum IL-18, but not IEC MHCII (Supplementary Fig. 4e).
IFNg is a unique and potent driver of MHCII expression on IECs in vitro
As IECs have little/no expression of the IL-18 or Il-12 receptors (Supplementary Fig. 5a) and elevated IL-18 in many of our models precedes the upregulation of MHCII, we hypothesized that IL-18 did not act directly on epithelial cells to upregulate MHCII31. Stimulation of WT SI organoids with high-dose IL-18 (50 ng/mL) failed to upregulate MHCII, whereas IFNg stimulation did, even at doses as low as 0.1 ng/mL (Supplementary Fig. 5b and data not shown). To determine whether any other stimuli could induce IEC organoid MHCII, we stimulated with various cytokines and pathogen-associated molecules chosen based on their receptor expression in WT IECs16 or their presence within the intestinal microenvironment. Strikingly, only IFNg was able to upregulate MHCII expression on IECs (Supplementary Fig. 5b). Similar stimulation of human fetal small intestine organoids showed upregulation of MHCII with IFNg, but not IL-18, LPS, or CpG stimulation (Supplementary Fig. 5c) supporting IFNg as a primary driver of MHCII expression in both murine and human IECs.
IEC MHCII expression requires IFNg signaling in vivo
In vivo, T-cell transfer colitis and irradiation1,5 require IFNg to upregulate IEC MHCII. We sought to determine whether IFNg was an absolute requirement for IEC MHCII induction in the variety of IL-18-dependent systems described above. Using Ifng−/− bone marrow chimeras and Ifngr1−/− mice, we found that induction of either SI or colonic IEC MHCII in Rag1−/− (Fig. 7a), Nlrc4TS (Fig. 7b), TLR9-stimulated (Fig. 7c), and Tritrichomonas (Fig. 7d) colonized mice required IFNg or the IFNg receptor. Though not comprehensive, these data suggest IFNg is likely to be required for IEC MHCII upregulation regardless of whether the stimulus is acute or chronic, local or systemic.
Epithelial cell gMFI and representative histograms in lethally irradiated Rag1−/− mice reconstituted with either WT or Ifng−/− (gKO) bone marrow (a), NLRC4TS mice with or without Ifngr1 (b), and Ifngr1+/− and Ifngr1−/− littermates treated with CpG (c), or gavaged with Tritrichomonas-enriched stool (d). qPCR showing equal colonization with Tritrichomonas (d, inset). Data show combined results for two independent experiments, except for b, which shows a representative experiment from n = 2, with 3–5 mice per condition. Statistical significance is shown on each graph and was determined by unpaired t test. BMCs bone marrow chimeras, gMFI geometric mean fluorescent intensity, gKO Ifng−/−, TMu Tritrichomonas, Unt untreated (no irradiation or reconstitution).
Since IEC MHCII induction requires IFNg but is not observed prior to weaning, we first confirmed that neonatal IECs express the IFNg receptor (Supplementary Fig. 4f). As murine intestines are not fully populated with lymphocytes until after weaning24,32, we hypothesized that IL-18 acted on IFNg-producing intestinal lymphocytes to increase IEC MHCII. Neither T- nor B-cells are absolutely required for IEC MHCII upregulation, given the robust responses we observed in Rag1−/− mice. Investigation of colonic lamina propria innate lymphoid cells (ILCs) in Rag1−/− mice did not show an increase in the proportion of colonic NK or ILCs1 cells, but rather a shift in their expression of KLRG1 consistent with an activated phenotype (Fig. 8a–c). Ex vivo stimulation with PMA/Ionomycin resulted in dramatic loss of ILC/NK cells, preventing direct confirmation of their IFNg production. By contrast, examination of SI lamina propria of Nlrc4TS mice showed no change in NK/ILC1 populations, but a dramatic increase in both CD4 and CD8 T-cells (Fig. 8d–g). These cells showed a non-significant trend towards more type I differentiation/activation. These data suggest that chronic IL-18 is associated with activation and/or expansion of diverse, and likely redundant, IFNg-competent intestinal lymphocyte populations.
Quantification of lamina propria lymphocytes by flow cytometry. Total live single CD45+ (a) and ILC (b) cell populations in Jackson WT vs. Jackson and Pitt Rag1−/− mice. Representative flow plots and quantification showing KLRG1 NK1.1 double-positive cells (c). T cell and NK cell composition (d, e) including representative flow plots and Tbet+ cells (f) in in-house WT vs. NLRC4TS/TS. Isolated ILCs were stimulated with Brefeldin, PMA and ionomycin for 2.5 h and IFNg expression quantified (g). Summary diagram (h) created with BioRender.com. a–c Representative on n = 2 experiments with three mice per experimental group. d–g Combined results from n = 3 experiments with n = 1–2 mice per group. Statistical significance is shown on each graph and was determined by unpaired t test. Jax Jackson Rag1−/− mice.
Discussion
Intestinal epithelial expression of MHCII was described nearly 50 years ago, and has since then largely existed as a phenomenon associated with specific microbial or inflammatory triggers, or its analysis restricted to specific portions of the intestine33. More recent selective deletion and single-cell technologies have ushered a resurgence of interest in the phenomenon. Though many studies implicate DCs as the main sources of productive intestinal antigen presentation8,9,34, others nevertheless highlight a role for IEC MHCII in epithelial remodeling, GvHD, and infectious colitis1,4,5,11. As our appreciation of the functions of IEC MHCII expands, understanding the factors regulating its expression become increasingly important.
We found that Rag1−/− mice in our colony had elevated serum IL-18 and colonic and SI MHCII expression. IL-18 and IEC MHCII were antibiotic-sensitive in Rag1−/− mice, suggesting the activity of specific commensal mediators. Co-housing commercially sourced Rag1−/− mice (low for IL-18 and MHCII) with those locally bred resulted in a pronounced fecal microbial shift characterized by the increased abundance of Helicobacter, Prevotella, and Mucispirillum (Deferribacteracea) genera. Similar co-housing of WT mice did not induce serum IL-18 or IEC MHCII despite a similar microbial shift, suggesting a role for immunodeficiency in microbial-driven MHCII induction. Though the resolution of this analysis did not permit the identification of specific bacterial species, Helicobacter, Prevotella, and Mucispirillum are all organisms bound strongly by IgA14,15. The interaction of these bacteria and host immunodeficiency may have enabled them to interact more directly with IECs and driven Il-18 transcription. A similar induction of Il-18 was observed in dysbiotic TCRa−/− mice (Fig. 5d), suggesting T-cell functions, such as production of barrier-maintaining cytokines, T-cell dependent antibody responses, or limiting the outgrowth of ILCs could prevent the dysbiosis and IEC MHCII induction observed in Rag1−/− mice.
Strengthening the link between elevated IL-18 and IEC MHCII, we found that NLRC4 inflammasome hyperactivity drove IEC-dependent IL-18 overproduction and upregulated MHCII and related genes16. We also observed robust IEC MHCII expression on SI, but not colonic cells in other known IL-18 overproducing mouse strains (Il18Tg+/wt, Il18bp−/− and MEFVV726A/V726A), suggesting differential regulation of SI and colon IECs, and confirming a strong link between IL-18 and IEC MHCII. IEC MHCII was IL-18 dependent, as blocking IL-18 signaling or deleting Il-18 from IECs significantly decreased MHCII expression in these models.
In NLRC4 hyperactive mice, both elevated serum IL-18 and IEC MHCII depended on IEC production of IL-18, but not microbial factors, suggesting that IL-18 connects intestinal inflammatory stimulation to IEC MHCII16. As in myeloid cells, this intestinal IL-18 was cleaved and released via a classical inflammasome pathway that required caspase-1 and gasdermin-D. While other inflammasome components, particularly caspase-8, have been identified as downstream targets of NLRC421,22, our results leave little room for a contribution by other proteases. Even WT mice depended on caspase-1, gasdermin-D, and IEC IL-18 for maintaining basal serum IL-18, demonstrating a requirement for homeostatic intestinal inflammasome activity. Other inflammasome-nucleating proteins present in IECs, like NLRP6 or AIM2, may also be important contributors to basal IL-1835. We also identified an impressive synergy between factors driving IEC Il-18 transcription (e.g., dysbiosis and immunodeficiency) and factors promoting IEC inflammasome activation.
We consistently observed differences in the regulation of colonic vs. SI IEC MHCII such that genetically-mediated excess of IL-18 or NLRC4 inflammasome activation predominantly affected SI IECs, whereas Tritrichomonas colonization, TLR9 stimulation, and Rag1 deficiency/dysbiosis altered both colonic and SI MHCII. We also observed occasional spontaneous increases in WT SI, but not colonic, IEC MHCII expression. SI MHCII may be more sensitive to transient stimuli (e.g., SFB colonization8,13,36) and also more durable, as blockade of IL-18 or IL-12 via systemic antibody injection was able to alter colonic but not SI IEC expression (Fig. 3 and Supplementary Fig. 3). Likewise, genetic ablation of IEC Il-18 had a more profound effect on colonic vs. SI IEC MHCII expression (particularly in the TLR9 and Tritrichomonas systems). These regional differences in IEC MHCII regulation could be for several reasons: (1) greater feedback inhibition by microbial products in the colon leading to less sensitivity/durability of MHCII expression, (2) a greater abundance of IL-18 responsive/IFNg-producing lymphocytes in the SI, or (3) other mechanisms36,37.
In various systems, we found that IEC MHCII induction did not occur until about the time of weaning, typically days to weeks after elevation of systemic IL-18. Weaning is a dynamic time in the murine intestine, witnessing an increase in exposure to food antigens, a window where waning luminal IgA from ingested breast milk is not yet replaced by endogenous IgA, a temporary flash of intestinal STAT3 signals, and an influx of intestinal lymphocytes24,32. Food and microbial signals may not be necessary for IEC upregulation of MHCII, as lymphocytes enter the human intestine in utero and human IECs can upregulate MHCII around 18 weeks gestation2,38,39. We speculate that the influx and effector differentiation of lymphocytes competent to produce IFNg may be crucial for driving IEC MHCII in response to local stimuli, particularly IL-18. Obviously, IFNg production by T-cells is not absolutely required given robust IEC MHCII upregulation in dysbiotic Rag1−/− mice. Indeed, we observed activation of colonic NK1.1-expressing ILCs in locally bred, but not Jackson, Rag-deficient mice. Our analysis was not able to distinguish NK from ILC1 cells, but both express the IL-18 receptor and are competent to produce IFNg. In NLRC4TS mice, the most likely producers of IFNg appeared to be the dramatically expanded population of (largely) Tbet+ CD4 and CD8 T-cells in the lamina propria. Thus, both T-cells and ILCs may be sufficient IFNg producers for the induction of IEC MHCII.
Our mouse and human enteroid studies not only supported an indirect role for IL-18 in driving IEC MHCII, they expanded upon previous findings1,2,5,40 to suggest that IFNg was uniquely capable of inducing IEC MHCII. Why IFNg would drive IEC MHCII even under homeostatic conditions, and why upregulation of IEC MHCII is the unique domain of IFNg, remain mysterious. The latter may relate to a greater diversity of cytokine- and pattern recognition-receptors on classical APCs vs. IECs, or to the differential usage of promoters driving the MHC Class II Transactivator (CIITA)5,41. All CIITA promoters are IFNg-responsive, but promoter IV is more commonly used in non-hematopoietic cells and specifically regulates IFNg-inducible CIITA expression42.
Overall, these data link the diverse mechanisms contributing to intestinal IL-18 to local production of IFNg and geographically diverse upregulation of IEC MHCII (Fig. 8h). They provide a uniquely broad picture of the regulation of this energetically costly process, but questions pertaining to the significance of IEC MHCII in barrier integrity, host defense, and tolerance induction remain open. As these functions gain greater clarity, understanding IEC MHCII regulation will be critical for identifying and correcting pathology related to its dysregulation and exploiting this process to treat intestinal disease.
Materials and methods
Mice
All mice were bred and manipulated under specific pathogen-free conditions in 12-h light-dark cycles with food and water available ad libitum. All procedures were approved by the University of Pittsburgh animal care and use committee. All mice are on a C57Bl/6 background and originated from Jackson Laboratories, except the Il-18Tg+/wt (Dr. Tomoaki Hoshino, Kurume University), MEFVV726A/V726A mice (Drs. Daniel Kastner and Jae Jin Chae, NHGRI), Il18bp−/− (Knockout Mouse Project), Il18fl/fl (Dr. Richard Flavell, Yale University) and the NLRC4TS/TS 16,18,19,20,43. Ifng−/− bone marrow was a generous gift from Drs. Dario Vignali and Warren Shlomchik (University of Pittsburgh). All treatments were conducted on 6 weeks or older mice, except where noted.
Murine manipulation studies
Epithelial and Lamina Propria Isolations and Flow Cytometry
To obtain intestinal scrapes, mice were sacrificed and their colon and ~10 cm of small intestine (~5 cm distal to the stomach) were removed, colons were flushed with PBS, and organs flayed open. Sections were rinsed with PBS and the epithelial surfaces scraped with a sterile slide into cold DMEM with 2% FCS and 2 mM EDTA. Supplementary Fig. 6 shows the flow gating strategy for the colonic epithelium. Lamina propria cells were isolated according to described protocols44 and stimulated for 2.5 h with PMA/ionomycin and Brefeldin A to study cytokine secretion. Filtered single cells suspensions were stained with anti-mouse antibodies against CD3(OTK3), CD4(RM4-5), CD8b(YTS 156.7.7), CD45.2(104), CD90(53-2.1), B220(RA3-6B2), Epcam(G8.8), Fixable Live Dead Stain (Thermofisher, Waltham, MA), IFNg(XMG1.2), KLRG1(2F1), NK1.1 (pk136), MHCII(M5/114.152), RORgT(2B2), Tbet(4B10), TCRb(H57-597), TCRgd(eBioGL3), and/or unlabeled CD16/32(93) for 30–60 min at 4 °C. Cells were fixed prior flow and/or the addition of intracellular antibodies. Samples were run on an LSR Fortessa (BD Biosciences, San Jose, CA) and data analyzed using FlowJo v10.6 (Treestar, Ashland, OR) software. All antibodies were purchased from eBiosciences (San Diego, CA) or Biolegend (San Diego, CA).
CpG
CpG1826 (IDT Technologies) was injected at a dose of 2.5 mg/kg intraperitoneally (ip) as done previously26. A total of 2–5 CpG injections, spaced 2 days apart, were given with takedown 24 h after the last injection. For the pup experiments, 5-day-old mice were injected daily with 0.125 mg CpG for 4 days and sacrificed on day 5.
Antibiotic treatments
Adult mice were transferred to fresh cage bases and given drinking water supplemented with metronidazole (1 g L−1), ampicillin (1 g L−1), neomycin (1 g L−1) and vancomycin (0.5 g L−1) for 3 weeks. Control and antibiotic water were sweetened with saccharine 6 g/500 mL.
IL-12/IL-18 blockade
Adult Rag1−/− and Nlrc4TS mice were injected ip with 0.25–0.5 mg/mouse Anti-IL-18R Clone 9E6 (IL-1F4(mu):10235, Genentech, San Francisco, CA) or 0.2–0.4 mg/mouse (Anti-Mouse IL-12p40 Clone 17.8, Bio X Cell, Lebanon, NH) every 3 days for 2 weeks. To account for variability in MHCII fluorescence between the IL-18-R block experiments, MHCII gMFI was calculated as a percentage of the average of the isotype-treated controls in each experiment.
Tritrichomonas treatment, Isolation, and qPCR
Tritrichomonas was identified during routine import testing at the University of Pittsburgh and confirmed to be T. muris or musculis based on qPCR amplification45. Cecal stool from colonized mice was isolated and Tritrichomonas were enriched by centrifugation as done previoulsy29 prior to gavage. Mice were sacrificed and analyzed for IEC MHCII and serum IL-18 2 weeks post colonization.
BMCs
Rag1−/− mice were lethally irradiated with 1100 cGy (split dose, 2 h apart) in an XRAD320 (Precision Xray Inc) irradiator. The following day mice were reconstituted with 4–6 million WT or IFNg−/− bone marrow cells. Intestinal scrapes were collected 4–6 weeks post treatment initiation.
Serum IL-18 measurements
Serum was collected from terminal bleeds and analyzed using a BD Cytometric Bead Array according to the manufacturer’s instructions, run on the BD LSR II, and analyzed using FCAP Array Software (BD Biosciences, v. 3.0, San Jose, CA).
Organoid cultures
Murine
WT organoid cultures were generated from SI samples according to the manufacturer’s protocol (Stem Cell Technologies, Vancouver, Canada) in IntestiCult™ Organoid Growth Medium. Organoids were routinely passaged and maintained in Matrigel domes every 7–10 days. For TLR and cytokine stimulation, mature organoids were seeded at a density of ~300 organoids/well in a 12 well dish. Following organoid budding (3–5 days post passage), the media was removed and replaced with TLR/cytokine supplemented media and the samples incubated at 37 °C for 3.5 days. Organoids were removed, stained, and analyzed as described above for the IEC scrapes. All murine cytokines were purchased from Peprotech (Rockyhill, NJ) and used at a concentration of 10 ng/mL, apart from IL-18 (MBL International, 50 ng/mL) and IL-2 (Miltenyi, Germany). See Supplementary Table 1 for concentration and source of the TLR ligands analyzed.
Human
SI enteroids were isolated from surgically resected fetal (<24 weeks) SI tissue according to described protocols46,47. Samples were procured from the Health Science Tissue Bank of UPMC Magee-Womens Hospital in accordance with the guidelines set forth by the University of Pittsburgh Ethics Committee. Briefly, 100 crypts were plated in Matrigel in 48 well plates containing a 50:50 vol:vol ratio of human intestinal stem cells media (plus 50 mM EGF)47 and WRN-conditioned media48. Three days post plating the media was removed and replaced with TLR/Cytokines supplemented media (same concentrations as the murine organoids). Organoids were harvested 3.5 days post stimulation according to the methods described in ref. 40 and stained and analyzed for IEC MHCII surface expression.
Co-housing and 16S sequence analysis
Three- to five-week-old Rag1−/− mice bred locally or purchased commercially (Jackson labs) and/or WT mice were co-housed for 5 weeks prior to sacrifice. Cecal stool samples were collected at the time of death and frozen until processing. Fecal DNA was extracted using the QiaAMP Stool DNA Mini Kit (Qiagen, Germantown, MD) according to the manufacturer’s instructions and sequencing performed by Microbiome Insights (University of British Columbia, Canada). Microbiome informatics were performed by AHPB using QIIME2 2020.249. Raw sequences were quality-filtered and denoised with DADA250. Amplicon sequence variants (ASVs) were aligned with mafft and used to construct a phylogeny with fasttree251,52. Alpha diversity metrics (observed OTUs), beta diversity metrics (Jaccard Similarity) and Principal Coordinate Analysis were estimated after samples were rarefied to 63,000 (subsampled without replacement) sequences per sample. Taxonomy was assigned to ASVs using naive Bayes taxonomy classifier against the Greengenes 18_8 99% OTUs reference sequences53. All plots were made with publicly available R packages. Full sequence data are available upon request.
Analysis of publicly available data sets
Publicly available data sets were obtained from the Gene Expression Omnibus. Briefly, terminal ileum Il-18 read counts in co-housed, SFB-colonized WT, Rag−/− and TCRa−/− mice were obtained from Mao et al. and accessed from GSE8678024. Normalized read counts from bulk RNA-seq of FACS-sorted IECs from neonatal intestine derived from mice 4 h after subcutaneous injection of saline54. t-SNE plot (perplexity = 25) colored by inferred cell type or gene expression of select murine cytokine receptors from Haber et al. 31 visualized in Single-Cell Expression Atlas.
Statistics
Unpaired Student’s t tests or one-way ANOVAs with Tukey post correction were performed in GraphPad Prism (Version 8.3.1) as identified in the figure legends. Results were considered significant if p < 0.05.
References
Koyama, M. et al. MHC class II antigen presentation by the intestinal epithelium initiates graft-versus-host disease and is influenced by the microbiota. Immunity 51, 885–898 (2019).
Wosen, J. E., Mukhopadhyay, D., Macaubas, C. & Mellins, E. D. Epithelial MHC class II expression and its role in antigen presentation in the gastrointestinal and respiratory tracts. Front Immunol. 9, 2144 (2018).
Hershberg, R. M. et al. Intestinal epithelial cells use two distinct pathways for HLA class II antigen processing. J. Clin. Investig. 100, 204–215 (1997).
Jamwal, D. R. et al. Intestinal epithelial expression of MHCII determines severity of chemical, T cell-induced, and infectious colitis in mice. Gastroenterology 159, 1342–1356.e1346 (2020).
Thelemann, C. et al. Interferon-gamma induces expression of MHC class II on intestinal epithelial cells and protects mice from colitis. PLoS One 9, e86844 (2014).
Bekiaris, V., Persson, E. K. & Agace, W. W. Intestinal dendritic cells in the regulation of mucosal immunity. Immunol. Rev. 260, 86–101 (2014).
Grainger, J. R., Askenase, M. H., Guimont-Desrochers, F., da Fonseca, D. M. & Belkaid, Y. Contextual functions of antigen-presenting cells in the gastrointestinal tract. Immunol. Rev. 259, 75–87 (2014).
Goto, Y. et al. Segmented filamentous bacteria antigens presented by intestinal dendritic cells drive mucosal Th17 cell differentiation. Immunity 40, 594–607 (2014).
Maggio-Price, L. et al. Lineage targeted MHC-II transgenic mice demonstrate the role of dendritic cells in bacterial-driven colitis. Inflamm. Bowel Dis. 19, 174–184 (2013).
Hepworth, M. R. et al. Innate lymphoid cells regulate CD4+ T-cell responses to intestinal commensal bacteria. Nature 498, 113–117 (2013).
Biton, M. et al. T helper cell cytokines modulate intestinal stem cell renewal and differentiation. Cell 175, 1307–1320 (2018). e1322.
Ting, J. P. & Trowsdale, J. Genetic control of MHC class II expression. Cell 109, S21–S33 (2002). Suppl.
Umesaki, Y., Okada, Y., Matsumoto, S., Imaoka, A. & Setoyama, H. Segmented filamentous bacteria are indigenous intestinal bacteria that activate intraepithelial lymphocytes and induce MHC class II molecules and fucosyl asialo GM1 glycolipids on the small intestinal epithelial cells in the ex-germ-free mouse. Microbiol Immunol. 39, 555–562 (1995).
Bunker, J. J. & Bendelac, A. IgA responses to microbiota. Immunity 49, 211–224 (2018).
Palm, N. W. et al. Immunoglobulin A coating identifies colitogenic bacteria in inflammatory bowel disease. Cell 158, 1000–1010 (2014).
Weiss, E. S. et al. Interleukin-18 diagnostically distinguishes and pathogenically promotes human and murine macrophage activation syndrome. Blood 131, 1442–1455 (2018).
Poznanski, S. M. et al. Combined Stimulation with Interleukin-18 and Interleukin-12 Potently Induces Interleukin-8 Production by Natural Killer Cells. J. Innate Immun. 9, 511–525 (2017).
Hoshino, T. et al. Cutting edge: IL-18-transgenic mice: in vivo evidence of a broad role for IL-18 in modulating immune function. J. Immunol. 166, 7014–7018 (2001).
Girard-Guyonvarc’h, C. et al. Unopposed IL-18 signaling leads to severe TLR9-induced macrophage activation syndrome in mice. Blood 131, 1430–1441 (2018).
Chae, J. J. et al. Gain-of-function Pyrin mutations induce NLRP3 protein-independent interleukin-1beta activation and severe autoinflammation in mice. Immunity 34, 755–768 (2011).
Vance, R. E. The NAIP/NLRC4 inflammasomes. Curr. Opin. Immunol. 32, 84–89 (2015).
Rauch, I. et al. NAIP-NLRC4 inflammasomes coordinate intestinal epithelial cell expulsion with eicosanoid and IL-18 release via activation of caspase-1 and -8. Immunity 46, 649–659 (2017).
Van Opdenbosch, N. & Lamkanfi, M. Caspases in cell death, inflammation, and disease. Immunity 50, 1352–1364 (2019).
Mao, K. et al. Innate and adaptive lymphocytes sequentially shape the gut microbiota and lipid metabolism. Nature 554, 255–259 (2018).
Canna, S. W. et al. Interferon-gamma mediates anemia but is dispensable for fulminant toll-like receptor 9-induced macrophage activation syndrome and hemophagocytosis in mice. Arthritis Rheum. 65, 1764–1775 (2013).
Behrens, E. M. et al. Repeated TLR9 stimulation results in macrophage activation syndrome-like disease in mice. J. Clin. Investig. 121, 2264–2277 (2011).
Tsoukas, P. et al. Interleukin-18 and cytotoxic impairment are independent and synergistic causes of murine virus-induced hyperinflammation. Blood 136, 2162–2174 (2020).
Chudnovskiy, A. et al. Host-protozoan interactions protect from mucosal infections through activation of the inflammasome. Cell 167, 444–456.e414 (2016).
Escalante, N. K. et al. The common mouse protozoa Tritrichomonas muris alters mucosal T cell homeostasis and colitis susceptibility. J. Exp. Med 213, 2841–2850 (2016).
Schneider, C. et al. A metabolite-triggered tuft. Cell-ILC2 Circuit Drives Small Intestinal Remodeling. Cell 174, 271–284.e214 (2018).
Haber, A. L. et al. A single-cell survey of the small intestinal epithelium. Nature 551, 333–339 (2017).
Al Nabhani, Z. et al. A weaning reaction to microbiota is required for resistance to immunopathologies in the adult. Immunity 50, 1276–1288.e1275 (2019).
Skoskiewicz, M. J., Colvin, R. B., Schneeberger, E. E. & Russell, P. S. Widespread and selective induction of major histocompatibility complex-determined antigens in vivo by gamma interferon. J. Exp. Med 162, 1645–1664 (1985).
Stagg, A. J. Intestinal dendritic cells in health and gut inflammation. Front Immunol. 9, 2883 (2018).
Ratsimandresy, R. A., Indramohan, M., Dorfleutner, A. & Stehlik, C. The AIM2 inflammasome is a central regulator of intestinal homeostasis through the IL-18/IL-22/STAT3 pathway. Cell Mol. Immunol. 14, 127–142 (2017).
Bowcutt, R. et al. Heterogeneity across the murine small and large intestine. World J. Gastroenterol. 20, 15216–15232 (2014).
Olivares-Villagomez, D. & Kaer, V. L. Intestinal intraepithelial lymphocytes: sentinels of the mucosal barrier. Trends Immunol. 39, 264–275 (2018).
Stras, S. F. et al. Maturation of the HUman Intestinal Immune System Occurs Early in Fetal Development. Dev. Cell 51, 357–373.e355 (2019).
Torow, N., Marsland, B. J., Hornef, M. W. & Gollwitzer, E. S. Neonatal mucosal immunology. Mucosal Immunol. 10, 5–17 (2017).
Wosen, J. E. et al. Human intestinal enteroids model MHC-II in the gut epithelium. Front Immunol. 10, 1970 (2019).
Pai, R. K., Askew, D., Boom, W. H. & Harding, C. V. Regulation of class II MHC expression in APCs: roles of types I, III, and IV class II transactivator. J. Immunol. 169, 1326–1333 (2002).
Morris, A. C., Beresford, G. W., Mooney, M. R. & Boss, J. M. Kinetics of a gamma interferon response: expression and assembly of CIITA promoter IV and inhibition by methylation. Mol. Cell Biol. 22, 4781–4791 (2002).
Nowarski, R. et al. Epithelial IL-18 equilibrium controls barrier function in colitis. Cell 163, 1444–1456 (2015).
Hall, J. A. et al. Essential role for retinoic acid in the promotion of CD4(+) T cell effector responses via retinoic acid receptor alpha. Immunity 34, 435–447 (2011).
Howitt, M. R. et al. Tuft cells, taste-chemosensory cells, orchestrate parasite type 2 immunity in the gut. Science 351, 1329–1333 (2016).
Drummond, C. G. et al. Enteroviruses infect human enteroids and induce antiviral signaling in a cell lineage-specific manner. Proc. Natl Acad. Sci. USA 114, 1672–1677 (2017).
Sato, T. et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology 141, 1762–1772 (2011).
Miyoshi, H. & Stappenbeck, T. S. In vitro expansion and genetic modification of gastrointestinal stem cells in spheroid culture. Nat. Protoc. 8, 2471–2482 (2013).
Bolyen, E. et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 37, 852–857 (2019).
Callahan, B. J. et al. DADA2: high-resolution sample inference from Illumina amplicon data. Nat. Methods 13, 581–583 (2016).
Katoh, K., Misawa, K., Kuma, K. & Miyata, T. MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res 30, 3059–3066 (2002).
Price, M. N., Dehal, P. S. & Arkin, A. P. FastTree 2-approximately maximum-likelihood trees for large alignments. PLoS One 5, e9490 (2010).
McDonald, D. et al. An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea. ISME J. 6, 610–618 (2012).
Van Winkle, J. A., Constant, D. A., Li, L. & Nice, T. J. Selective interferon responses of intestinal epithelial cells minimize tumor necrosis factor alpha cytotoxicity. J Virol. 94, e00603–e00620 (2020).
Acknowledgements
S.C., C.S., and V.D. were supported by NICHD R01HD098428. L.V.D.K. and A.H.P.B. were supported by autoimmunity and immunopathology T32 (5T32AI089443-11). T.H. was supported by the Kenneth Rainin Foundation and R01DK120697. We also thanks Drs. Daniel Kastner and Jae Jin Chae (NHGRI); Dario Vignali, Warren Shlomchik, and Sarah Gaffen (University of Pittsburgh); Tomoaki Hoshino (Kurume University); and Richard Flavell (Yale University) for sharing key reagents.
Author information
Authors and Affiliations
Contributions
S.C. and L.V.D.K. planned and analyzed the experiments, which were conducted by L.V.D.K., C.S., V.D., E.W., J.V., L.Y., and S.C. A.H.P.B. performed the 16S analysis and is supervised by T.H. The manuscript was written by L.V.D.K. and S.C. and all authors reviewed and approved the manuscript prior to submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplemental information
Rights and permissions
About this article
Cite this article
Van Der Kraak, L.A., Schneider, C., Dang, V. et al. Genetic and commensal induction of IL-18 drive intestinal epithelial MHCII via IFNγ. Mucosal Immunol 14, 1100–1112 (2021). https://doi.org/10.1038/s41385-021-00419-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41385-021-00419-1
This article is cited by
-
Biological and clinical roles of IL-18 in inflammatory diseases
Nature Reviews Rheumatology (2024)