Abstract
Intact epithelial barrier function is pivotal for maintaining intestinal homeostasis. Current therapeutic developments aim at restoring the epithelial barrier in inflammatory bowel disease. Histone deacetylase (HDAC) inhibitors are known to modulate immune responses and to ameliorate experimental colitis. However, their direct impact on epithelial barrier function and intestinal wound healing is unknown. In human and murine colonic epithelial cell lines, the presence of the HDAC inhibitors Givinostat and Vorinostat not only improved transepithelial electrical resistance under inflammatory conditions but also attenuated the passage of macromolecules across the epithelial monolayer. Givinostat treatment mediated an accelerated wound closure in scratch assays. In vivo, Givinostat treatment resulted in improved barrier recovery and epithelial wound healing in dextran sodium sulphate-stressed mice. Mechanistically, these regenerative effects could be linked to an increased secretion of transforming growth factor beta1 and interleukin 8, paralleled by differential expression of the tight junction proteins claudin-1, claudin-2 and occludin. Our data reveal a novel tissue regenerative property of the pan-HDAC inhibitors Givinostat and Vorinostat in intestinal inflammation, which may have beneficial implications by repurposing HDAC inhibitors for therapeutic strategies for inflammatory bowel disease.
Similar content being viewed by others
Introduction
Inflammatory bowel diseases (IBDs) are chronic relapsing conditions of the gastrointestinal tract. Recent work has highlighted the importance of the intestinal barrier in maintaining the homeostasis between immune cells, epithelial cells and the microbiome.1 The majority of inflammatory bowel diseases can be classified as either Crohn’s disease (CD) or ulcerative colitis (UC). CD is characterised as a transmural inflammation with the potential to affect any part of the gastrointestinal tract, whereas UC only involves the mucosa and is limited to the large intestine.2 Intestinal epithelial cells (IECs) form the main physical barrier, which prevents excessive contact of luminal antigens with intestinal immune cells and thereby protects from unwanted immune reactions. The regulation of intestinal permeability by this functional layer represents a sophisticated balance that is primarily adjusted by the cell-cell connecting tight junction (TJ) proteins as well as by continuous cell renewing.3,4 In addition, IECs contribute to intestinal homeostasis by secreting immunoregulatory and regenerative cytokines including transforming growth factor beta (TGFβ), interleukin-8 (IL-8) and monocyte chemo-attractant protein-1.5,6,7 Genetic and functional studies indicate that disruption of the epithelial barrier and subsequent bacterial translocation represent the initial step of intestinal inflammation.8 Furthermore, IECs are not able to form a functional epithelial monolayer in a chronically inflammatory milieu, since epithelial apoptosis is enhanced and barrier-forming TJ proteins are downregulated, underlining barrier formation and regeneration as critical processes in IBD pathogenesis.9
As mucosal healing such as epithelial protection and restoration of the impaired intestinal barrier has emerged as a key goal for IBD therapy, a combined anti-inflammatory and tissue-regenerating drug therapy is desirable.10 Recently, the interest in butyrate, a short-chain fatty acid and histone deacetylase (HDAC) inhibitor produced by intestinal microbiota, was renewed and its importance of the intestinal homestasis demonstrated.11
Suberoylanilide hydroxamic acid (SAHA) and ITF2357, labelled as Vorinostat and Givinostat, are both hydroxamic acids and pan-HDAC inhibitors.12 While Vorinostat is approved for the treatment of cutaneous T-cell lymphoma,13 Givinostat is currently undergoing clinical trials both in oncology as well as in chronic inflammation (NCT00792467; NCT01761968) and has been granted orphan drug designation in the European Union. Both HDAC inhibitors suppress cancer cell proliferation in vitro14 and reduce experimental tumour growth in vivo.15 Our previous work indicates an anti-inflammatory effect of HDAC inhibition in models of experimental colitis.15,16,17 Furthermore, specific effects on T cells and macrophages have been demonstrated.17,18
HDAC inhibition has been shown to attenuate fibrosis in various models.19,20,21 The effects of HDAC inhibitors on epithelial cells have been discussed controversially and there are limited data on the effects of HDAC inhibitors on intestinal barrier function and regeneration. Ohno et al. described the induction of the regenerative cytokine macrophage inflammatory protein-2 (MIP-2) in IEC under inflammatory conditions in the presence of the HDAC inhibitors sodium (Na)-butyrate or trichostatin A.22 Bordin et al. demonstrated later that Vorinostat and Na-butyrate upregulate TJ protein expression in Rat-1 fibroblasts and HeLa cells,23 which was transferred to IEC by Wang et al.24 Thus, there are data to indicate that HDAC inhibition supports intestinal epithelial barrier restitution. However, the mechanisms and its functional impact have not yet been defined.
Here we analysed effects of HDAC inhibition on the intestinal epithelial barrier under inflammatory conditions. Using in vitro model systems with human and murine epithelial cell lines as well as in vivo dextran sulphate sodium (DSS)-induced colitis as barrier disruption model in mice, a barrier-protective and regenerative effect of HDAC inhibitors mediated by a direct effect on the epithelial cells was identified. Functionally, we revealed an effect of HDAC inhibition on epithelial autocrine transforming growth factor beta1 (TGFβ1) secretion with upstream effects on TJ and IL-8 expression.
Methods
Reagents
Hank’s balanced salt solution (HBSS), phosphate-buffered saline (PBS), Dulbecco’s modified Eagle’s medium (DMEM), DMEM/F12, advanced DMEM/F12 and RPMI were obtained from Thermo Scientific (Schwerte, Germany). Medium was supplemented with 10% foetal calf serum, 50 U/ml penicillin (Biochrom, Berlin, Germany) and 50 µg/ml streptomycin (Biochrom). Cytokines and recombinant proteins were from Peprotech (New Jersey, NJ, USA). Fluorescein isothiocyanate-labelled dextran 4 kDa (FITC-dextran) was obtained from TdB Consultancy (Uppsala, Sweden). All other chemicals were obtained from Sigma-Aldrich (St. Louis, MO, USA) unless indicated otherwise.
HDAC inhibitors
Givinostat (ITF2357) was synthesised by the Chemical Department of Italfarmaco (Cinisello Balsamo, Italy) and processed as described previously.15 Vorinostat (SAHA) was provided by MSD (Haar, Germany). HDAC inhibitors were diluted in sterile distilled water and heated to 95 °C for complete dissolution and stored at −80 °C or kept at room temperature for immediate use in animal models.
Human samples and isolation of IECs
Biopsy specimens were obtained from colonic mucosa at routine colonoscopy in patients with active disease (CD and UC) or disease in remission (CD rem and UC rem) undergoing a diagnostic examination and in healthy controls included in a polyp surveillance programme. In each patient four to six biopsy specimens were taken from the sigmoid colon, using standard biopsy forceps (Olympus, Japan). Patients gave their informed consent prior to surgery. The study was approved by the local ethics commission of the Charité – Universitätsmedizin Berlin (Approval number: EA4/049/15).
The biopsy specimens were immediately transferred into ice-cold 0.9% NaCl solution and washed three times in HBSS. The mucosa was digested for 10–15 min at 37 °C in 15 ml HBSS containing 2 mM EDTA/1 mM EGTA shaking at 200 rpm. Then colonic crypts were mechanically separated from tissue specimens by repeated pipetting through a steel needle followed by filtration through a 70 µM cellstrainer (Falcon, Becton Dickinson, San Jose, USA). Isolated IECs were washed with RPMI medium and centrifuged twice (400 × g for 10 min). Viability and purity of epithelial cells was confirmed by flow cytometric analysis (FACS Canto II; Becton Dickinson) staining with propidium iodide (Roth, Karlsruhe, Germany) and allophycocyanin-conjugated anti-epithelial cell adhesion molecule (EpCAM, CD326)-specific antibody (9C4; Biolegend, San Diego, USA), respectively, with >95% cells being epithelial cell-specific antigen-positive.25,26 IECs were collected by centrifugation and cell pellet was snap-frozen in liquid nitrogen for later RNA analysis.
Epithelial cell culture, organoid culture and in vitro treatment
The human colorectal cell line T84 (American Type Culture (ATCC) CCL-248) and the murine rectal cell line CMT93 (ATCC CCL-223) were cultured according to the provider’s instructions. Both are epithelial cell lines, which have been widely used for studying intestinal barrier and integrity.27,28 For primary organoid culture, crypts from the small intestine and the colon were isolated from 10- to 15-week-old C57BL/6J mice using 2 mM EDTA/PBS isolation buffer or 2 mM EDTA/chelation buffer (PBS supplemented with 54.9 mM d-sorbitol and 43.4 mM sucrose), respectively, as previously described.29,30 Crypts were transferred into matrigel (BD Biosciences) in 48-well plates and were cultured for a minimum of 6 days to allow formation of established organoids. Crypt culture medium (Advanced DMEM/F12) containing 2 mM l-glutamin (Biochrom), 50 U/ml penicillin (Biochrom), 50 μg/ml streptomycin (Biochrom), 10 mM HEPES (Sigma-Aldrich), 1× N-2 supplement (Thermo Scientific), 1× B-27 supplement (Thermo Scientific), 50 ng/ml recombinant murine epidermal growth factor, 100 ng/ml recombinant murine Noggin, 500 ng/ml recombinant human R-Spondin-1 and 500 mM N-acetyl-l-cystein, was replaced every day. For colon organoid culture, crypt culture medium was additionally supplemented 1:2 with Wnt-3A-conditioned medium obtained from the L Wnt-3A cell line (ATCC® CRL-2647) according to the manufacturer’s instructions. Before experiments, colonic organoids were cultured in Wnt-3A-free crypt culture medium for 4 days to allow differentiation.30 Cells and organoids were pre-treated for 16 h with HDAC inhibitors in various concentrations as indicated. If applicable, tumour necrosis factor alpha (TNFα) and interferon gamma (IFNγ) in indicated concentrations, 2 ng/mL recombinant human TGFβ1, 10 µg/ml monoclonal anti-human TGFβ1 (1D11.16.8 GeneTex, CA, USA) or 1 µM TGFβR1 receptor antagonist SB431542 (Selleckchem, Houston, TX, USA) were added to the cell culture.
Measurement of transepithelial resistance
T84 and CMT93 cells in standard culture medium were plated on permeable transwell polycarbonate filter supports (0.4 µm; 0.6 cm2, Merck Millipore, Billerica, MA) in a 12-well plate grown as monolayers until confluence as previously described.28 On day 10–14 after plating, filters were used for treatment with indicated conditions and the transepithelial electrical resistance (TER) as a marker of ion permeability of epithelial monolayers was measured with a Millicell-ERS voltohmmeter (Merck Millipore) as previously described.24
In vitro permeability assay
Passage of 4 kDa FITC-dextran across the epithelial layer was measured as described previously.31 Cells were grown on permeable supports for 10–14 days in standard medium without phenol red. A unit of 200 µM of dialysed FITC-dextran was added to the apical side. Fluxes were calculated from the amount of FITC-dextran in the basolateral compartment that was measured at 520 nm (Tecan Infinite M200, Tecan, Switzerland). Fluorescence signals were calibrated using defined dilutions in medium. Flux was calculated as the increase in tracer quantity (corrected for dilution) per time unit and filter area.
DSS-induced colitis model
Acute colitis was induced by feeding mice ad libitum with 2.5% DSS, 36,000–50,000 Da, (MP Biomedicals, Santa Ana, USA) dissolved in drinking water from day 1 to 6, followed by 7 days of regular drinking water. Givinostat treatment (5 mg/kg body weight) was initiated simultaneously at the start of water phase via oral gavage in 200 µl PBS twice daily. Clinical colitis parameters were assessed daily and disease score was evaluated as described previously.16 Colon length was measured post mortem. For in vivo wounding experiments, mice were treated as above, but at day 7 lower colonoscopy was performed as described before to obtain biopsies of approximately 800 µm in diameter with biopsy forceps inserted through the endoscope’s working channel (Karl Storz GmbH, Tuttlingen, Germany), thereby inducing a defined mucosal wound.15 Mice underwent additional colonoscopy 2 days later to assess macroscopic wound healing of the biopsy area. Endoscopic procedures surveyed on a colour monitor were recorded digitally (DSR- 20MD; Sony, Cologne, Germany). Mice were sacrificed on day 9.
In vivo permeability assay
On day 6 and 9 of experimental colitis the intestinal permeability to 4 kDa FITC-dextran was assessed as described previously.32 Briefly, 4 h-fasted mice were orally gavaged with dialysed FITC-dextran (250 mg/kg body weight). After 5 h, 200 μl cheek blood was collected into serum tubes (Greiner Bio-one, Frickenhausen, Germany) and serum was separated following the manufacturer’s instruction. Serum was diluted in an equal volume of PBS (pH 7.4) and analysed for FITC-dextran concentration at 520 nm (Tecan Infinite M200). Standard curves obtained from diluted FITC-dextran in non-treated serum/PBS served for calculating the FITC-dextran concentration in the samples.
Apoptosis assay
Cells were grown on filter supports, supernatants were removed and unattached dead cells were collected via centrifugation. Cell extracts were prepared by harvesting cells from the filter in 300 µl of lysis buffer containing 0.2% Triton X-100 followed by 30 min shaking at 4 °C followed by centrifugation (2.500 × g, 10 min, 4 °C). Caspase-3/7 activity in cell extracts was measured using the SensoLyte® Homogeneous AFC Caspase-3/7 assay kit (AnaSpec, Fremont, CA) according to the manufacturer’s protocol. For positive control cells were treated with 100 nM staurosporine overnight. Relative caspase activity was calculated from fluorescence intensity (Tecan Infinite M200).
Statistical analysis
Data are expressed as mean ± SEM. Statistical significance was determined by the indicated test using GraphPad PRISM software, version 6.0 (San Diego, CA, USA). p < 0.05 was considered significant
Additional Methods are described in the supplemental materials.
Results
Human colonic epithelial cells in IBD patients downregulate the expression of HDAC expression in a global manner
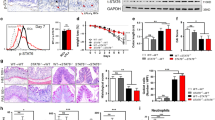
Epithelial cells from colonic biopsies from IBD patients and healthy controls were isolated (>98% purity, Suppl. Fig 1A) and mRNA expression for HDAC1–11 was analysed. As demonstrated in Fig. 1a, eight out of eleven HDAC (HDAC2, 3, 4, 5, 6, 8, 10, 11) were downregulated in the epithelial cells due to ongoing inflammation in these tissues, although overall histone 3 acetylation was not affected (Suppl. Figure 1B). The HDAC expression changes could be confirmed (Fig. 1b), when the patients were divided in UC and CD. Although not statistically significant for all HDAC, due to low sample numbers, this inflammation induced downregulation of most HDAC is also evident when active IBD patients were compared to UC and CD patients in remission, suggesting a counter mechanism to local epithelial tissue damages. Differential expression of Hdac1–11 could be confirmed in vitro in murine small intestine organoid cultures under inflammatory conditions (Suppl. Figure 1C). Even a confluent cell layer of the murine IEC line CMT93 showed similar expression changes of various Hdac under inflammatory conditions on mRNA level, which were confirmed on protein level by western blot comparing cytoplasmic, nuclear as well as membrane fractions (Suppl. Figure 1D/E).
Global downregulation of histone deacetylase (HDAC) mRNA expression in human colonic epithelial cells derived from inflammation. Primary intestinal epithelial cells (IECs) were isolated from mucosal biopsy specimens and mRNA levels of HDAC1–11 were determined by quantitative PCR relative to the healthy controls. a Shown are relative expression levels of HDAC1–11 mRNA in IBD patients with active disease state. Mean values and min to max whiskers of n = 7 (control) and n = 9 (IBD); Mann-Whitney U-test **p < 0.01, ***p < 0.001 versus healthy control. b Shown are relative expression levels of HDAC1–11 mRNA in patients with active disease state (CD and UC) and with disease in remission (CD rem and UC rem). Mean values and ranges of n = 7 (control), n = 4 (CD), n = 7 (CD rem), n = 5 (UC) and n = 2 (UC rem). Mann-Whitney U-test *p < 0.05, **p < 0.01 versus healthy control or versus remission control. CD Crohn’s disease, Ctrl control, IBD inflammatory bowel disease, rem disease in remission, UC ulcerative colitis
HDAC inhibition protects epithelial barrier function under inflammatory conditions in vitro
To discover, if the demonstrated downregulation of HDAC supports barrier integrity and regeneration, an in vitro model system based on changes in the TER of polarised epithelial cell monolayers was applied. Pan-HDAC inhibition was induced by treatment with two hydroxamic acids, already approved by the Food and Drug Administration (Vorinostat) and/or proven their safety in several clinical studies (Givinostat). Two IEC lines of human (T84) and murine (CMT93) origin were used, both of which developed a specific TER after 10–14 days of cultivation on permeable supports (T84: 3000 ± 500 Ω cm2; CMT93: 400 ± 50 Ω cm2).9,27 Incubation with increasing concentrations of the pan-HDAC inhibitor Givinostat led to an additional increase of TER in T84 and CMT93 cells up to 130 and 250%, respectively (Fig. 2a).
Stabilisation of epithelial barrier function under physiological and inflammatory conditions by histone deacetylase (HDAC) inhibition. Shown are data from confluent T84 and CMT93 monolayer on permeable transwell supports. a Cells were incubated in the presence of Givinostat in increasing concentrations for 48 h and transepithelial electrical resistance (TER) was measured. Mean values ± SEM of n = 6–10/condition from three independent experiments; Kruskal-Wallis test with Dunnett’s test **p < 0.01, ***p < 0.001 versus the vehicle control. b Cells were incubated in the presence of tumour necrosis factor alpha (TNFα) and interferon gamma (IFNγ) as indicated and TER was determined after 24 and 48 h. c Cells were pre-incubated for 16 h with either Givinostat (T84: 1 µM; CMT93: 0.6 µM) or Vorinostat (4 µM) followed by addition of TNFα and IFNγ (2 ng/ml each for T84; 5 ng/ml each for CMT93) indicated by the vertical dotted line. The TER was measured after 24 and 48 h and is shown relative to the TER of vehicle control. Mean values ± SEM of n = 4–18/condition from three independent experiments; one-way analysis of variance with Dunnett’s test **p < 0.01, ***p < 0.001 versus TNFα/IFNγ-treated vehicle control
As reported previously,33 the TER decreases under inflammatory conditions, here mimicked by the addition of TNFα and IFNγ (Fig. 2b). Based on the dose-dependent levels of barrier impairment, reflected by TER decrease, medium TNFα and IFNγ concentrations of 2 and 5 ng/ml each were chosen for T84 and CMT93 cells, respectively, for all subsequent experiments. To evaluate the effect of HDAC inhibition in this epithelial barrier model, cells were pre-incubated for 16 h with the pan-HDAC inhibitors Givinostat or Vorinostat followed by the addition of TNFα and IFNγ. During the time course of the experiment, changes in the TER were monitored daily. The TER of controls was stable throughout the experiment (t0: 2952 ± 291 Ω cm2 and t72h: 2854 ± 352 Ω cm2 for T84; t0: 457 ± 93 Ω cm2 and t72h: 428 ± 42 Ω cm2 for CMT93). HDAC inhibition with either Givinostat or Vorinostat maintained the TER under pro-inflammatory conditions in both cell lines suggesting a protective mechanism on the epithelial barrier function (Fig. 2c).
We next aimed to identify whether the maintenance of the epithelial barrier function by HDAC inhibition was associated with a reduced passage of macromolecules across the epithelial layer. To this end, we performed a unidirectional marker flux experiment by adding 0.2 mM of 4 kDa FITC-dextran to the apical site of the monolayer. FITC-dextran could pass the monolayer only in the presence of TNFα and IFNγ but not under physiological conditions (Fig. 3a, b). In concordance with the TER data above, pre-incubation with either HDAC inhibitor resulted in a reduced flux of FITC-dextran compared to non-treated cells under inflammatory conditions. This was indicated by a lowered concentration of FITC-dextran in the basolateral compartment throughout the time course of the experiment (Fig. 3a) and by a significantly reduced absolute flux of FITC-dextran (Fig. 3b). This effect was paralleled by decreased induction of apoptosis in the HDAC inhibitor treated groups under inflammatory conditions (Fig. 3c).
Histone deacetylase (HDAC) inhibition induces a reduction of macromolecular flux in colonic epithelial cell lines. T84 and CMT93 monolayers were grown to confluence on permeable transwell supports. Cells were pre-incubated for 16 h with Givinostat (T84: 1 µM; CMT93: 0.6 µM) or with Vorinostat (4 µM) followed by addition of tumour necrosis factor alpha (TNFα) and interferon gamma (IFNγ; 2 ng/ml each for T84; 5 ng/ml each for CMT93) for 24 h. a Transepithelial flux assay was conducted with 200 µM of fluorescein isothiocyanate (FITC)-dextran, added to the apical side of the monolayer. Basolateral FITC-dextran concentrations for all sequential time points were calculated from FITC fluorescence values and were baseline corrected for untreated controls. Mean values ± SEM of n = 4/condition of one experiment; Mann-Whitney U-test *p < 0.05 versus TNFα/IFNγ-treated vehicle control. b Shown are values of the total FITC-dextran flux over time, calculated from single concentration values as described in the Methods section. Mean values ± SEM of n = 4/condition; Mann-Whitney U-test *p < 0.05 versus TNFα/IFNγ-treated vehicle control. c The caspase-3/7 activity is shown relative to vehicle control. Mean values ± SEM of n = 6/condition from three independent experiments (T84) or n = 2/condition (CMT93); two-way analysis of variance with Tukey’s test **p < 0.01, ***p < 0.001 versus vehicle control or versus TNFα/IFNγ-treated vehicle control
Thus, HDAC inhibition ameliorates epithelial damage, thereby protecting barrier function under inflammatory conditions.
HDAC inhibition controls the regulation of TJ proteins under inflammatory conditions
Since barrier function and TER in particular highly depend on TJ and TJ-related proteins,21,23 the expression level of selected TJ proteins were analysed in total cell lysates in our experimental barrier model. This analysis revealed increased expression of the TJ proteins occludin and claudin-1 in the presence of HDAC inhibitors under inflammatory conditions. In parallel, a significant downregulation of the paracellular channel-forming claudin-2 protein was demonstrated. Expression levels of claudin-3 and claudin-5 were not affected by HDAC inhibition at all. In accordance with the expression of the marker proteins E-cadherin, MMP9 and vimentin, epithelial-mesenchymal transition (EMT) does not play a role in this setting (Fig. 4, Suppl. Figure 2).
Effect of histone deacetylase (HDAC) inhibition on tight junction expression. Confluent T84 monolayer on permeable transwell supports were pre-incubated for 16 h with Givinostat (1 µM) followed by addition of tumour necrosis factor alpha (TNFα) and interferon gamma (IFNγ; 2 ng/ml each) for 48 h. Total cell lysates were used for the detection of claudin-1, -2, -3, -5, occludin, E-cadherin, MMP9, vimentin and acetylated histone 3 (acetylH3) by western blot analysis, β-actin served as loading control. Left panels show representative blots; right panels indicate the corresponding ratios of band intensities in arbitrary units (AU) relative to β-actin. Mean values ± SEM of n = 4–10/condition from three independent experiments; one-way analysis of variance with Dunnett’s test *p < 0.05 versus TNFα/IFNγ-treated vehicle control
These findings further support the concept that HDAC inhibition stabilises barrier function by shifting expression levels of TJ proteins.
HDAC inhibition enhances epithelial wound healing
Since the integrity of the intestinal epithelial barrier under inflammatory conditions is shaped by maintenance but also barrier regeneration, we investigated the impact of HDAC inhibition on the migratory capacity of IEC. Here, a classical scratch assay served as an in vitro wound healing model in the presence or absence of Givinostat. For the human epithelial cell line T84, a highly significant increase in wound closure was observed in the presence of Givinostat after short (2 days)- and long-time cultivation periods (6 days) (Fig. 5). The CMT93 cells turned out to be unsuitable for this assay due to their high proliferation rate (data not shown). In contrast to the IEC, fibroblasts, here represented by the human fibroblast cell line NIH/3T3, showed reduced migration upon Givinostat treatment (Suppl. Figure 3). These data indicate that HDAC inhibition acts directly on epithelial cells to promote epithelial regeneration.
Givinostat-induced acceleration of wound closure and cell migration in colonic epithelial cells. Confluent T84 monolayers were cultured in serum-deprived medium and were wounded using a pipette tip. In vitro scratch closure was monitored over a 6 days culture period in the presence or absence of Givinostat. Left panels show representative pictures of the scratch assay at the indicated time points (×100 magnification). Right panels show the corresponding data in per cent of wound closure calculated by the open area at indicated time points relative to day 0. Mean values ± SEM of n = 23–33/condition from three independent experiments; unpaired t-test ***p < 0.001 versus vehicle-treated control
TGFβ1 is a key modulator of epithelial regeneration
TGFβ was previously described to mediate epithelial regeneration and thus enhances barrier function,34 while secreted by the epithelial cells themselves. Exposure of IEC to the pro-inflammatory milieu in the described in vitro barrier model resulted in a substantial increase of TGFβ1 mRNA expression, followed by a profound dose-dependent increase in the presence of Givinostat in both cell lines, hence suggesting a possible mode of action for an improved epithelial regeneration (Fig. 6a, b). This was confirmed, by a dose-dependent increase in secreted TGFβ1 in both cell lines, when treated with Givinostat under inflammatory conditions (Fig. 6c). The dose-dependent increase in protein expression upon Givinostat treatment could similarly be shown for the TGFβ-dependent regenerative cytokine IL-8 and the murine analogue MIP-2, respectively (Suppl. Figure 4A/B). Other inflammatory cytokines could not be detected or remained stable as, for example, IL-6. (Suppl. Figure 4C).
Histone deacetylase (HDAC) inhibition causes enhanced transforming growth factor beta1 (TGFβ1) expression in colonic epithelial cell lines under inflammatory conditions. a Confluent T84 (left panel) and CMT93 (right panel) monolayer were pre-incubated for 16 h with either Givinostat (T84: 1 µM; CMT93: 0.6 µM) or with Vorinostat (4 µM) followed by the addition of tumour necrosis factor alpha (TNFα) and interferon gamma (IFNγ; T84: 2 ng/ml each; CMT93: 5 ng/ml each) for 24 h. Cells were lysed and mRNA levels of TGFB1/Tgfb1 were determined by quantitative PCR relative to the vehicle control. Mean ± SEM of n = 4–6/condition from two independent experiments; two-way analysis of variance (ANOVA) with Holm-Sidak’s test *p < 0.05, **p < 0.01, ***p < 0.001 versus the vehicle control or TNFα/IFNγ-treated vehicle control. b As in a but monolayer were pre-incubated with increasing concentrations of Givinostat as indicated. Mean value ± SEM of n = 3–4/condition. c As in a but monolayer were pre-incubated with Givinostat as indicated for 24 h and secreted TGFβ1 levels were determined in the supernatant using the reporter cell line MFB-F11 with subsequent measurement of alkaline phosphatase (SEAP) activity as described in the Methods section. Mean value ± SEM of n = 3/condition; one-way ANOVA with Dunnett’s test *p < 0.05 versus the TNFα/IFNγ-treated vehicle control. d T84 monolayer were treated as indicated in a but for analysis of TGFβ1 effects, cells were either pre-incubated for 16 h with the TGFβR1 inhibitor SB431542 (1 µM) or with recombinant TGFβ1 (2 ng/ml). The transepithelial electrical resistance (TER) was measured after 24 and 48 h and is expressed relative to the TER of vehicle control. Mean values ± SEM of n = 4/condition from two independent experiments. Mann-Whitney U-test at t = 24 h and t = 48 h *p < 0.05 versus TNFα/IFNγ + Givinostat-treated cells or versus TNFα/IFNγ-treated vehicle control, respectively. e Confluent T84 monolayers were cultured in serum-deprived medium and were wounded using a pipette tip. In vitro scratch closure was monitored during culturing for 6 days in the presence or absence of Givinostat. For analysis of TGFβ1 effects, cells were additionally treated with anti-TGFβ1 antibody (αTGFβ1) (10 µg/ml), with the TGFβR1 inhibitor SB431542 (1 µM) or with recombinant TGFβ1 (2 ng/ml) as indicated. Bars show the corresponding data in per cent of wound closure calculated by the open area at indicated time points relative to day 0. Mean values ± SEM of n = 6–26/condition from two independent experiments; two-way ANOVA with Holm-Sidak’s test ***p < 0.001 versus vehicle control
To further validate the impact of TGFβ1 on barrier protection and regeneration, we either blocked the main TGFβ1 receptor (TGFβ1R) via the chemical inhibitor SB43154 or added recombinant TGFβ1 as a regenerative agent. As indicated, inhibition of TGFβ1R completely abolished the otherwise prominent effect of Givinostat on the TER, while TGFβ1 alone, when added instead of the HDAC inhibitor, closely mimicked the effect of Givinostat (Fig. 6d). The same held true for the scratch assay as an in vitro wound healing experiment. Here the blocking of TGFβ1R inhibited Givinostat-triggered wound healing, whereas the addition of TGFβ1 led to accelerated wound healing comparable to Givinostat treatment (Fig. 6e).
This increased TGFβ1 expression could be confirmed in primary cells by Givinostat and Vorinostat treatment of intestinal epithelial organoids in an inflammatory milieu. Tgfb1 mRNA expression was increased up to threefold in small intestine organoids, while there were no morphological differences between treatment groups and viability was not affected after HDAC inhibitor treatment (Fig. 7a, b). Also in colon organoids, Givinostat led to an increase in Tgfb1 transcription (Fig. 7c). The same holds true for the Mip-2 expression in small intestinal as well as colon organoids (Suppl. Figure 4D).
Histone deacetylase (HDAC) inhibition causes enhanced transforming growth factor β1 (TGFβ1) expression in primary murine intestinal organoids under inflammatory conditions. a Murine small intestine organoids and c colon organoids on day 6 were pre-incubated for 16 h with either 4 µM Givinostat or Vorinostat followed by the addition of 10 ng/ml tumour necrosis factor alpha (TNFα) and interferon gamma (IFNγ) each for 6 h. Organoids were lysed and mRNA levels of Tgfb1 were determined by quantitative PCR relative to the TNFα/IFNγ-treated vehicle control (left panel). Mean values ± SEM, n = 2–3/condition with three independent experiments, two-way analysis of variance with Holm-Sidak’s test *p < 0.05, ***p < 0.001 versus vehicle control. b Viability assay for three-dimensional organoids was performed in the presence of 4 µM Givinostat or Vorinostat for 2 days with or without treatment with 10 ng/ml TNFα and IFNγ for 6 or 30 h. Mean values ± SEM, n = 3/condition. Lower panels show representative pictures of untreated and Givinostat-treated organoids (×100 magnification, scale bar 50 µm)
Taken together, these experiments reveal a critical role for the induction of TGFβ1 by Givinostat as a major mechanism to exert the demonstrated regenerative effects.
HDAC inhibition promotes recovery via epithelial regeneration in experimental colitis
Having shown that HDAC inhibition maintains the intestinal epithelial barrier function in IEC and enhances epithelial regeneration, we aimed to explore whether these in vitro findings equally apply in vivo. As model system, the recovery phase of DSS-induced colitis was chosen. For this, colitis was induced by administration of 2.5% DSS for 6 days followed by initiation of drinking water and Givinostat (10 mg/kg body weight/day) treatment via oral gavage. In order to evaluate barrier leakage and barrier restitution, day 6 as the peak of barrier disruption and day 9 as time point of recovery were chosen for comparison (Fig. 8a). In a second setting, mucosal wounds were induced locally in the colon of mice and wound healing was monitored by lower endoscopy (Fig. 8b).
Givinostat treatment promotes intestinal regeneration in vivo. Mice were exposed to 2.5% dextran sulphate sodium (DSS) in the drinking water for 6 consecutive days followed by 7 days of regular drinking water. Mice were weight matched and Givinostat treatment (10 mg/kg body weight) was initiated at the end of day 6 and continued until the end of experiment. a, b The experimental setup of DSS-induced colitis and subsequent recovery is illustrated. c Stool consistency, bleeding and weight loss were scored separately and were summarised as clinical score as described in the Methods section. d Weight was determined daily and expressed as percentage of initial body weight at day 6. e At the end of experiment, colon length was measured. c–e Mean values ± SEM of n = 2 (vehicle), n = 8 (DSS + vehicle) and n = 9 (DSS + Givinostat). Mann-Whitney U-test *p < 0.05, **p < 0.01 versus DSS-treated vehicle control. f To assess intestinal permeability, 4 kDa fluorescein fluorescein isothiocyanate (FITC)-dextran (250 mg/kg body weight) was administered orally (day 6 and day 9) and serum concentrations were determined 5 h later. Mean values ± SEM of n = 2 (d6 vehicle), n = 2 (d6 Givinostat), n = 8 (d6 DSS + vehicle), n = 9 (d6 DSS + Givinostat), n = 6 (d9 vehicle), n = 6 (d9 Givinostat), n = 19 (d9 DSS + vehicle) and n = 23 (d9 DSS + Givinostat); Kruskal-Wallis test with Dunnett’s test **p < 0.01. The experiment was repeated three times. g To assess wound healing in vivo, colonic mucosa was locally wounded during endoscopy in a DSS-induced colitis model as indicated in b and mucosal healing of wounded sites was reported after 3 days of Givinostat treatment via lower endoscopy. Shown are 8 representative pictures out of 40 (2 wounds per mouse with n = 5/group)
Givinostat treatment significantly reduced the recovery time as indicated by a faster and earlier increase in body weight. This was equally reflected by amelioration of the clinical colitis score suggesting accelerated healing from the DSS-induced barrier disruption (Fig. 8c, d). In line, colon shortening, a surrogate marker for the degree of disease severity, was significantly ameliorated in the Givinostat-treated group (Fig. 8e). To analyse the barrier integrity specifically, mice were gavaged with 4 kDa FITC-dextran and the fluorescence intensity in the serum was measured. After 6 days of DSS exposure, an expected increase in intestinal permeability for FITC-dextran was observed. Remarkably, at day 9 Givinostat treatment resulted in an almost complete recovery, comparable to non-DSS-treated control mice, while DSS-exposed vehicle-treated mice did not reveal any signs of recovery regarding the integrity of the intestinal barrier (Fig. 8f, Suppl. Figure 5). Moreover, significantly accelerated healing of local wounding sites, inflicted by endoscopic clamps, was observed in DSS-treated mice after 3 days of Givinostat treatment. Wounds of Givinostat-treated mice were in later wound healing stages as DSS-treated vehicle control as indicated by less visible blood, less fibrin accumulation, reduced hypervascularisation and reduced overall wound size (Fig. 8g).
These in vivo findings demonstrate that HDAC inhibitor treatment accelerates intestinal epithelial recovery after both, DSS-induced barrier disruption and mechanical wounding, and hence are in line with our in vitro data.
Discussion
In the present study, we described a novel, direct effect of the HDAC inhibitors Givinostat and Vorinostat on epithelial barrier function and regeneration in intestinal inflammation. With our in vitro systems, we demonstrated for the first time a stabilisation of barrier formation and epithelial integrity mediated by HDAC inhibitors, which resulted in a protection against barrier disruption and transepithelial leakage under inflammatory conditions. Here HDAC inhibition acted directly on the IEC and promoted tissue repair via epithelial cell restitution and accelerated migration. In vivo, Givinostat treatment increased mucosal healing and intestinal regeneration after DSS-induced barrier disruption and mechanical wounding. Mechanistically, this effect was linked to a dose-dependent increase in the expression of the regenerative cytokines TGFβ1 and IL-8 and subsequently to reduced apoptosis and a differential expression of the TJ proteins claudin-1 and claudin-2 as well as occludin.
The intestinal permeability is a delicate balance, tightly regulated by epithelial regeneration and the expression of junctional proteins on IEC. Pro-inflammatory cytokines that are induced in IBD (e.g. TNFα and IFNγ) induce epithelial cell apoptosis and affect the expression of epithelial TJ proteins in IEC, resulting in the disruption of intestinal epithelial barrier function.9,33,35 Simultaneously, a downregulation of most (eight out of eleven) HDAC could be demonstrated in epithelial cells isolated from UC and CD patients when compared to healthy controls. As this could be confirmed even in patients in remission compared to active IBD, this broad downregulation of HDAC might indicate a counter mechanism to the inflammation-induced tissue destruction and barrier disruption.
In our in vitro studies on polarised epithelial cells grown on transwell filters, the presence of HDAC inhibitors induced an increase in TER, thus a reduction of ion permeability, without an effect on junction proteins (data not shown). Interestingly, under inflammatory conditions, the positive effect on the TER was even more pronounced and the transepithelial flux of the 4 kDa macromolecule FITC-dextran was reduced. Here HDAC inhibition resulted in reduced apoptosis paralleled by higher expression of the barrier-forming protein claudin-1 and of occludin and downregulation of channel-forming claudin-2. While inflammatory cytokines that are upregulated in human IBD induce a loss of barrier-protective junctional proteins (e.g. E-cadherin, claudin-1 and occludin),9 the expression and junctional localisation of channel-forming TJ proteins such as claudin-2, is elevated in both UC and CD.36,37 Since claudin-2, also described as the “leaky gut claudin”, forms a paracellular channel for ions, our data link the reduced ion permeability, reflected by the TER under inflammatory conditions, to a shift in TJ expression towards barrier-protective TJ proteins after HDAC inhibitor treatment.
Regarding effects of other HDAC inhibitors, Na-butyrate has been described to decrease FITC-dextran flux and increase TER via enhanced claudin-1 transcription in an IEC-6-derived cell line or by repressing claudin-2 expression in T84 cells under normal conditions.24,38 Both findings are in line with our results regarding Givinostat or Vorinostat on T84 cells under inflammatory conditions. Moreover, we could identify significantly upregulated occludin as an additional potential mediator of barrier stabilisation among other prominent TJ proteins (claudin-3 and claudin-5), which are not affected. Occludin expression is reported to be reduced in patient samples in which claudin-2 levels are elevated, suggesting both as IBD-associated structural alterations of the epithelial barrier.36
Notably, we could not observe a regulation of TJ expression in untreated cells under inflammatory conditions but an induction of apoptosis, which also enables FITC-dextran to pass the barrier. Here, treatment with a HDAC inhibitor reduced cell apoptosis in IEC within the epithelial layer on the filter membrane. Moreover, occludin seems to be involved in barrier regulation for macromolecules in intestinal cell lines, since its knockdown results in increased permeability.39 Hence, TJ modulation by claudin-1, claudin-2 and occludin expression along with protection from apoptosis contribute to the observed attenuation of FITC-dextran flux under inflammatory conditions in our model.
Altogether, these data support the hypothesis that HDAC inhibition mediates a structural rearrangement of the epithelial barrier leading to protection against inflammation-related tissue damage.
Further on, we provide evidence for accelerated cell migration of epithelial cell lines in vitro in addition to the barrier protecting-effect. In the performed wound healing assay the IEC, as the single cell component, can only be influenced in an autocrine manner and not by surrounding immune cells. This underlines the direct effect of the HDAC inhibitors on IEC to promote wound repair via epithelial cell restitution and migration, independent from the known anti-inflammatory effect on lamina propria cells in vivo.
EMT, as an initial part of the wound healing process,40,41 has also been shown to be regulated by HDAC inhibitors: Na-butyrate and valproic acid induced EMT in colon carcinoma cell lines, including enhanced migration. Remarkably, these effects were augmented in the presence of TGFβ1.21 TGFβ1 has equally been described to induce EMT in a lung epithelial cell line paralleled by an upregulation of claudin-1 and vimentin and a downregulation of E-cadherin.42 In our in vitro model we could observe no regulation of E-cadherin, vimentin or MMP9, implicating that EMT likely plays a negligible role for the regenerative effects of HDAC inhibition.
Acute DSS-induced colitis model served as model for regeneration after barrier disruption in order to translate our in vitro findings to an in vivo system.43 DSS treatment leads to the destruction of the epithelial monolayer, introducing a barrier disruption and a subsequent inflammation in the colon, making this model ideal to confirm our in vitro data of epithelial barrier regeneration under inflammatory conditions. Earlier studies from our group highlighted the anti-inflammatory potency of HDAC inhibitor treatment during experimental colitis resulting in a reduced disease severity as well as suppression of pro-inflammatory cytokine secretion.15 Here we observed that Givinostat therapy during the re-epithelialisation phase promoted regeneration and colonic healing, resulting in an improved clinical score, clearly reduced macromolecular leakage and visible mucosal healing of wounded sites. While it is impossible to exclude effects of HDAC inhibition on other cells involved, we here provide evidence for the accelerated regeneration process, in particular for the reconstituted intestinal epithelial barrier. At least, an additional direct effect of HDAC inhibition regarding cell migration and wound healing on fibroblasts, as critical part of the stroma, could be excluded in vitro, which is in line with previous studies.19,44 Last, to prove the mode of action in primary epithelial cells, we treated murine epithelial organoids with Givinostat and Vorinostat under inflammatory conditions and could confirm the primary effect of the induction of TGFβ1 expression.
Epithelial healing emerges as a finely tuned process. Here, IECs receive various signals from the microenvironment, such as TGFβ or IL-8, which are both significantly upregulated in our in vitro system and activate master transcription factors, such as nuclear factor-kappaB and STAT3 leading to survival and proliferation.45,46 TGFβ is a well-described regenerative factor and mandatory for the regulation of mucosal healing.47 In CMT93 cells TGFβ was shown to attenuate the damage of epithelial monolayer and changes in TER caused by TNFα and IFNγ. Notably, claudin-1, but not occludin or claudin-2, has been described to be regulated by TGFβ signalling in T84 cells, independent from proliferation.34 In addition, our data indicate that TGFβ1 production is not only upregulated under pro-inflammatory conditions, but demonstrate a striking dose-dependent increase of TGFβ1 for both cell lines in the presence of HDAC inhibitors. Accordingly, addition of the TGFβR1 antagonist SB431542 reversed the effects of Givinostat, whereas treatment with TGFβ1 impressively reproduced these effects, revealing TGFβ1 as key mediator of enhanced epithelial regeneration induced by Givinostat or Vorinostat. Consistently, TGFβ has been tightly connected to barrier integrity, and has previously been identified as a central regulator of epithelial restitution in IBD,45,48 mediating mucosal healing in vitro and in vivo.47 Additionally, TGFβ1 regulates the expression of adhesion molecules, such as E-cadherin or Notch ligands in lymphocytes, which are also therapeutic targets for IBD49 and support barrier formation in T84 cells via activation of ERK/MAPK/SMAD signalling. This includes an upregulation of claudin-1 and an increase in TER.34 In DSS-induced colitis, it has been shown that TGFβ prevents IEC from apoptosis,50 an effect we could also show in the presence of a HDAC inhibitor in our model under pro-inflammatory conditions. In addition, accumulation of extracellular matrix proteins in intestinal strictures, as seen in chronic inflammation and fibrosis, were associated with increased TGFβ level.51 With TGFβ as a critical factor in IBD and a prerequisite for mucosal wound healing, we propose the increased TGFβ1 expression in our model system as the main mode of action for the demonstrated regenerative capacity of Givinostat and Vorinostat. Enhanced TGFβ signalling is paralleled by reduced apoptosis, reassembling of TJ protein expression towards promotion of tight cell-cell connections, enhanced migration and even increased IL-8 expression of epithelial cells, all effects directly correlated to barrier integrity and regeneration as summarised in Fig. 9
Illustration of the proposed mechanism. Histone deacetylase (HDAC) inhibitor treatment (left side) compared to normal condition (right side) supports epithelial barrier restitution and regeneration by induction of transforming growth factor beta1 (TGFβ1) production in intestinal epithelial cells, leading to accelerated migration, reduced apoptosis and epithelial tight junction modulation
As IECs form the structural basis for an intact mucosal barrier and repeated intestinal epithelial damage with disruption of the intestinal barrier function is considered as a central disease inducing and maintaining mechanism in IBD, mucosal healing is the current aim of treatment in clinical practice. Our results identified the HDAC inhibitors Givinostat and Vorinostat, which are already part of numerous clinical trials or are approved drugs, respectively,13 are not merely inhibitors of basic cell functions like proliferation or cytokine production,52 but rather act as inducers of beneficial processes such as tissue regeneration and barrier protection. Together with their confirmed anti-inflammatory capacity, HDAC inhibitors offer an attractive mode of action and hence represent promising candidates for the treatment of IBD.
References
Martini, E., Krug, S. M., Siegmund, B., Neurath, M. F. & Becker, C. Mend your fences: the epithelial barrier and its relationship with mucosal immunity in inflammatory bowel disease. Cell. Mol. Gastroenterol. Hepatol. 4, 33–46 (2017).
Satsangi, J., Silverberg, M. S., Vermeire, S. & Colombel, J. F. The Montreal classification of inflammatory bowel disease: controversies, consensus, and implications. Gut 55, 749–753 (2006).
Vetrano, S. et al. Unique role of junctional adhesion molecule-a in maintaining mucosal homeostasis in inflammatory bowel disease. Gastroenterology 135, 173–184 (2008).
Hering, N. A., Fromm, M. & Schulzke, J. D. Determinants of colonic barrier function in inflammatory bowel disease and potential therapeutics. J. Physiol. 590, 1035–1044 (2012).
Eckmann, L. et al. Differential cytokine expression by human intestinal epithelial cell lines: regulated expression of interleukin 8. Gastroenterology 105, 1689–1697 (1993).
Reinecker, H. C. et al. Monocyte-chemoattractant protein 1 gene expression in intestinal epithelial cells and inflammatory bowel disease mucosa. Gastroenterology 108, 40–50 (1995).
Ciacci, C., Lind, S. E. & Podolsky, D. K. Transforming growth factor beta regulation of migration in wounded rat intestinal epithelial monolayers. Gastroenterology 105, 93–101 (1993).
McGuckin, M. A., Eri, R., Simms, L. A., Florin, T. H. & Radford-Smith, G. Intestinal barrier dysfunction in inflammatory bowel diseases. Inflamm. Bowel Dis. 15, 100–113 (2009).
Bruewer, M. et al. Proinflammatory cytokines disrupt epithelial barrier function by apoptosis-independent mechanisms. J. Immunol. 171, 6164–6172 (2003).
Atreya, R. & Neurath, M. F. Current and future targets for mucosal healing in inflammatory bowel disease. Visc. Med. 33, 82–88 (2017).
Scott, N. A. et al. Antibiotics induce sustained dysregulation of intestinal T cell immunity by perturbing macrophage homeostasis. Sci. Transl. Med. 10 (2018).
Dinarello, C. A., Fossati, G. & Mascagni, P. Histone deacetylase inhibitors for treating a spectrum of diseases not related to cancer. Mol. Med. 17, 333–352 (2011).
West, A. C. & Johnstone, R. W. New and emerging HDAC inhibitors for cancer treatment. J. Clin. Invest. 124, 30–39 (2014).
Armeanu, S. et al. Apoptosis on hepatoma cells but not on primary hepatocytes by histone deacetylase inhibitors valproate and ITF2357. J. Hepatol. 42, 210–217 (2005).
Glauben, R. et al. Histone deacetylases: novel targets for prevention of colitis-associated cancer in mice. Gut 57, 613–622 (2008).
Glauben, R. et al. Histone hyperacetylation is associated with amelioration of experimental colitis in mice. J. Immunol. 176, 5015–5022 (2006).
de Zoeten, E. F., Wang, L., Sai, H., Dillmann, W. H. & Hancock, W. W. Inhibition of HDAC9 increases T regulatory cell function and prevents colitis in mice. Gastroenterology 138, 583–594 (2010).
Glauben, R., Sonnenberg, E., Wetzel, M., Mascagni, P. & Siegmund, B. Histone deacetylase inhibitors modulate interleukin 6-dependent CD4 + T cell polarization in vitro and in vivo. J. Biol. Chem. 289, 6142–6151 (2014).
Consalvi, S. et al. Preclinical studies in the mdx mouse model of duchenne muscular dystrophy with the histone deacetylase inhibitor givinostat. Mol. Med. 19, 79–87 (2013).
Ota, C. et al. Histone deacetylase inhibitor restores surfactant protein-C expression in alveolar-epithelial type II cells and attenuates bleomycin-induced pulmonary fibrosis in vivo. Exp. Lung Res. 41, 422–434 (2015).
Ji, M. et al. HDAC inhibitors induce epithelial-mesenchymal transition in colon carcinoma cells. Oncol. Rep. 33, 2299–2308 (2015).
Ohno, Y., Lee, J., Fusunyan, R. D., MacDermott, R. P. & Sanderson, I. R. Macrophage inflammatory protein-2: chromosomal regulation in rat small intestinal epithelial cells. Proc. Natl Acad. Sci. USA 94, 10279–10284 (1997).
Bordin, M., D’Atri, F., Guillemot, L. & Citi, S. Histone deacetylase inhibitors up-regulate the expression of tight junction proteins. Mol. Cancer Res. 2, 692–701 (2004).
Wang, H. B., Wang, P. Y., Wang, X., Wan, Y. L. & Liu, Y. C. Butyrate enhances intestinal epithelial barrier function via up-regulation of tight junction protein Claudin-1 transcription. Dig. Dis. Sci. 57, 3126–3135 (2012).
Litvinov, S. V., Velders, M. P., Bakker, H. A., Fleuren, G. J. & Warnaar, S. O. Ep-CAM: a human epithelial antigen is a homophilic cell-cell adhesion molecule. J. Cell Biol. 125, 437–446 (1994).
Teijaro, J. R. et al. Endothelial cells are central orchestrators of cytokine amplification during influenza virus infection. Cell 146, 980–991 (2011).
Flynn, A. N. & Buret, A. G. Tight junctional disruption and apoptosis in an in vitro model of Citrobacter rodentium infection. Microb. Pathog. 45, 98–104 (2008).
Lissner, D. et al. Monocyte and M1 macrophage-induced barrier defect contributes to chronic intestinal inflammation in IBD. Inflamm. Bowel Dis. 21, 1297–1305 (2015).
Grabinger, T. et al. Ex vivo culture of intestinal crypt organoids as a model system for assessing cell death induction in intestinal epithelial cells and enteropathy. Cell Death Dis. 5, e1228 (2014).
Sato, T. et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology 141, 1762–1772 (2011).
Sanders, S. E., Madara, J. L., McGuirk, D. K., Gelman, D. S. & Colgan, S. P. Assessment of inflammatory events in epithelial permeability: a rapid screening method using fluorescein dextrans. Epithelial Cell Biol. 4, 25–34 (1995).
Volynets, V. et al. Assessment of the intestinal barrier with five different permeability tests in healthy C57BL/6J and BALB/cJ mice. Dig. Dis. Sci. 61, 737–746 (2016).
Fish, S. M., Proujansky, R. & Reenstra, W. W. Synergistic effects of interferon gamma and tumour necrosis factor alpha on T84 cell function. Gut 45, 191–198 (1999).
Howe, K. L., Reardon, C., Wang, A., Nazli, A. & McKay, D. M. Transforming growth factor-beta regulation of epithelial tight junction proteins enhances barrier function and blocks enterohemorrhagic Escherichia coli O157:H7-induced increased permeability. Am. J. Pathol. 167, 1587–1597 (2005).
Heller, F. et al. Interleukin-13 is the key effector Th2 cytokine in ulcerative colitis that affects epithelial tight junctions, apoptosis, and cell restitution. Gastroenterology 129, 550–564 (2005).
Zeissig, S. et al. Changes in expression and distribution of claudin 2, 5 and 8 lead to discontinuous tight junctions and barrier dysfunction in active Crohn’s disease. Gut 56, 61–72 (2007).
Randall, K. et al. Claudin-2 expression levels in ulcerative colitis: development and validation of an in-situ hybridisation assay for therapeutic studies. PLoS ONE 11, e0162076 (2016).
Zheng, L. et al. Microbial-derived butyrate promotes epithelial barrier function through IL-10 receptor-dependent repression of claudin-2. J. Immunol. 199, 2976–2984 (2017).
Al-Sadi, R. et al. Occludin regulates macromolecule flux across the intestinal epithelial tight junction barrier. Am. J. Physiol. Gastrointest. Liver Physiol. 300, G1054–G1064 (2011).
Ahmed, N. et al. Molecular pathways regulating EGF-induced epithelio-mesenchymal transition in human ovarian surface epithelium. Am. J. Physiol. Cell. Physiol. 290, C1532–C1542 (2006).
Yan, C. et al. Epithelial to mesenchymal transition in human skin wound healing is induced by tumor necrosis factor-alpha through bone morphogenic protein-2. Am. J. Pathol. 176, 2247–2258 (2010).
Oyanagi, J., Ogawa, T., Sato, H., Higashi, S. & Miyazaki, K. Epithelial-mesenchymal transition stimulates human cancer cells to extend microtubule-based invasive protrusions and suppresses cell growth in collagen gel. PLoS ONE 7, e53209 (2012).
Bernasconi, E. et al. Granulocyte-macrophage colony-stimulating factor elicits bone marrow-derived cells that promote efficient colonic mucosal healing. Inflamm. Bowel Dis. 16, 428–441 (2010).
Conforti, F. et al. The histone deacetylase inhibitor, romidepsin, as a potential treatment for pulmonary fibrosis. Oncotarget 8, 48737–48754 (2017).
Dignass, A. U. & Podolsky, D. K. Cytokine modulation of intestinal epithelial cell restitution: central role of transforming growth factor beta. Gastroenterology 105, 1323–1332 (1993).
Gatla, H. R. et al. Histone deacetylase (HDAC) inhibition induces IkappaB kinase (IKK)-dependent interleukin-8/CXCL8 expression in ovarian cancer cells. J. Biol. Chem. 292, 5043–5054 (2017).
Beck, P. L. et al. Transforming growth factor-beta mediates intestinal healing and susceptibility to injury in vitro and in vivo through epithelial cells. Am. J. Pathol. 162, 597–608 (2003).
Ihara, S., Hirata, Y. & Koike, K. TGF-beta in inflammatory bowel disease: a key regulator of immune cells, epithelium, and the intestinal microbiota. J. Gastroenterol. 52, 777–787 (2017).
Zhang, N. & Bevan, M. J. Transforming growth factor-beta signaling controls the formation and maintenance of gut-resident memory T cells by regulating migration and retention. Immunity 39, 687–696 (2013).
Sakuraba, H., Ishiguro, Y., Yamagata, K., Munakata, A. & Nakane, A. Blockade of TGF-beta accelerates mucosal destruction through epithelial cell apoptosis. Biochem. Biophys. Res. Commun. 359, 406–412 (2007).
Di Sabatino, A. et al. Transforming growth factor beta signalling and matrix metalloproteinases in the mucosa overlying Crohn’s disease strictures. Gut 58, 777–789 (2009).
Leoni, F. et al. The antitumor histone deacetylase inhibitor suberoylanilide hydroxamic acid exhibits antiinflammatory properties via suppression of cytokines. Proc. Natl Acad. Sci. USA 99, 2995–3000 (2002).
Acknowledgements
We are grateful to Inka Freise and Britta Jebautzke for excellent technical assistance. We thank Paolo Mascagni (Italfarmaco, Cinisello Balsamo, Italy) for providing us with ITF2357 (Givinostat). This work is part of the PhD thesis of Marie Friedrich. This work was supported by the Helmholtz Alliance “Preclinical Comprehensive Cancer Center (PCCC)” of the Helmholtz Society, the Deutsche Forschungsgemeinschaft (SI 749/5-3 and SI 749/7-1) and the Deutsche Krebshilfe (70112011). Further funding was received from the Academic Grant´s Committee der Charité – Universitätsmedizin Berlin, the Berlin Institute of Health, the Sonnenfeld Stiftung and DFG funding through the Berlin-Brandenburg School for Regenerative Therapies GSC 203.
Author information
Authors and Affiliations
Contributions
The study was designed by R.G., B.S. and M.F.. M.F., L.G., M.G., R.R., C.W., J.K., W.H. and F.S. performed experiments and the associated analyses. K.S. and W.W. performed immunohistochemistry. M.F., L.G., R.G. and B.S. wrote the manuscript. All authors have seen and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Friedrich, M., Gerbeth, L., Gerling, M. et al. HDAC inhibitors promote intestinal epithelial regeneration via autocrine TGFβ1 signalling in inflammation. Mucosal Immunol 12, 656–667 (2019). https://doi.org/10.1038/s41385-019-0135-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41385-019-0135-7
This article is cited by
-
The crucial role of SETDB1 in structural and functional transformation of epithelial cells during regeneration after intestinal ischemia reperfusion injury
Histochemistry and Cell Biology (2024)
-
Unlocking the potential of targeting histone-modifying enzymes for treating IBD and CRC
Clinical Epigenetics (2023)
-
Regulation of chromatin organization during animal regeneration
Cell Regeneration (2023)
-
Epigenetic histone modulation contributes to improvements in inflammatory bowel disease via EBI3
Cellular and Molecular Life Sciences (2020)
-
Pioglitazone Attenuates Experimental Colitis-Associated Hyperalgesia through Improving the Intestinal Barrier Dysfunction
Inflammation (2020)