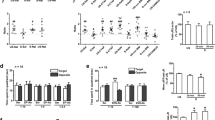
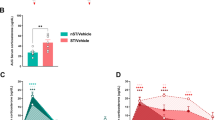
Abstract
Chronic stress causes cognitive deficits, such as impairments in episodic-like hippocampus-dependent memory. Stress regulates an opioid-related neuropeptide named Nociceptin/Orphanin FQ (N/OFQ), the ligand of the G protein-coupled receptor NOP. Since this peptide has deleterious effects on memory, we hypothesized that the N/OFQ system could be a mediator of the negative effects of stress on memory. Chronic stress was mimicked by chronic exposure to corticosterone (CORT). The NOP receptor was either acutely blocked using selective antagonists, or knocked-down specifically in the hippocampus using genetic tools. Long-term memory was assessed in the object recognition (OR) and object location (OL) paradigms. Acute injection of NOP antagonists before learning had a negative impact on memory in naive mice whereas it restored memory performances in the chronic stress model. This rescue was associated with a normalization of neuronal cell activity in the CA3 part of the hippocampus. Chronic CORT induced an upregulation of the N/OFQ precursor in the hippocampus. Knock-down of the NOP receptor in the CA3/Dentate Gyrus region prevented memory deficits in the CORT model. These data demonstrate that blocking the N/OFQ system can be beneficial for long-term memory in a neuroendocrine model of chronic stress. We therefore suggest that NOP antagonists could be useful for the treatment of memory deficits in stress-related disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Raw data are available from the corresponding author upon request.
References
Lupien SJ, McEwen BS, Gunnar MR, Heim C. Effects of stress throughout the lifespan on the brain, behaviour and cognition. Nat Rev Neurosci. 2009;10:434–45.
Lupien SJ, de Leon M, de Santi S, Convit A, Tarshish C, Nair NP, et al. Cortisol levels during human aging predict hippocampal atrophy and memory deficits. Nat Neurosci. 1998;1:69–73.
Darcet F, Mendez-David I, Tritschler L, Gardier AM, Guilloux JP, David DJ. Learning and memory impairments in a neuroendocrine mouse model of anxiety/depression. Front Behav Neurosci. 2014;8:136.
Kim EJ, Pellman B, Kim JJ. Stress effects on the hippocampus: a critical review. Learn Mem. 2015;22:411–6.
Luine V, Villegas M, Martinez C, McEwen BS. Repeated stress causes reversible impairments of spatial memory performance. Brain Res. 1994;639:167–70.
Magarinos AM, Verdugo JM, McEwen BS. Chronic stress alters synaptic terminal structure in hippocampus. Proc Natl Acad Sci USA. 1997;94:14002–8.
Sandi C, Pinelo-Nava MT. Stress and memory: behavioral effects and neurobiological mechanisms. Neural Plast. 2007;2007:78970.
Rock PL, Roiser JP, Riedel WJ, Blackwell AD. Cognitive impairment in depression: a systematic review and meta-analysis. Psychol Med. 2014;44:2029–40.
Miskowiak KW, Ott CV, Petersen JZ, Kessing LV. Systematic review of randomized controlled trials of candidate treatments for cognitive impairment in depression and methodological challenges in the field. Eur Neuropsychopharmacol. 2016;26:1845–67.
Meunier JC, Mollereau C, Toll L, Suaudeau C, Moisand C, Alvinerie P, et al. Isolation and structure of the endogenous agonist of opioid receptor- like ORL1 receptor. Nature. 1995;377:532–5.
Reinscheid RK, Nothacker HP, Bourson A, Ardati A, Henningsen RA, Bunzow JR, et al. Orphanin FQ: a neuropeptide that activates an opioidlike G protein- coupled receptor. Science. 1995;270:792–4.
Mollereau C, Mouledous L. Tissue distribution of the opioid receptor-like (ORL1) receptor. Peptides. 2000;21:907–17.
Anand P, Yiangou Y, Anand U, Mukerji G, Sinisi M, Fox M, et al. Nociceptin/orphanin FQ receptor expression in clinical pain disorders and functional effects in cultured neurons. Pain. 2016;157:1960–9.
Ding H, Kiguchi N, Yasuda D, Daga PR, Polgar WE, Lu JJ, et al. A bifunctional nociceptin and mu opioid receptor agonist is analgesic without opioid side effects in nonhuman primates. Sci Transl Med. 2018;10:eaar3483.
Jais A, Paeger L, Sotelo-Hitschfeld T, Bremser S, Prinzensteiner M, Klemm P, et al. PNOC(ARC) neurons promote hyperphagia and obesity upon high-fat-diet feeding. Neuron. 2020;106:1009–25.e10.
Smith MA, Choudhury AI, Glegola JA, Viskaitis P, Irvine EE, de Campos Silva PCC, et al. Extrahypothalamic GABAergic nociceptin-expressing neurons regulate AgRP neuron activity to control feeding behavior. J Clin Invest. 2020;130:126–42.
Hardaway JA, Halladay LR, Mazzone CM, Pati D, Bloodgood DW, Kim M, et al. Central amygdala prepronociceptin-expressing neurons mediate palatable food consumption and reward. Neuron. 2019;102:1037–52.e7.
Parker KE, Pedersen CE, Gomez AM, Spangler SM, Walicki MC, Feng SY, et al. A paranigral VTA nociceptin circuit that constrains motivation for reward. Cell. 2019;178:653–71.e19.
Rodriguez-Romaguera J, Ung RL, Nomura H, Otis JM, Basiri ML, Namboodiri VMK, et al. Prepronociceptin-expressing neurons in the extended amygdala encode and promote rapid arousal responses to motivationally salient stimuli. Cell Rep. 2020;33:108362.
Kallupi M, Carrette LLG, Kononoff J, Solberg Woods LC, Palmer AA, Schweitzer P, et al. Nociceptin attenuates the escalation of oxycodone self-administration by normalizing CeA-GABA transmission in highly addicted rats. Proc Natl Acad Sci USA. 2020;117:2140–8.
Morairty SR, Sun Y, Toll L, Bruchas MR, Kilduff TS. Activation of the nociceptin/orphanin-FQ receptor promotes NREM sleep and EEG slow wave activity. Proc Natl Acad Sci USA. 2023;120:e2214171120.
Andero R, Brothers SP, Jovanovic T, Chen YT, Salah-Uddin H, Cameron M, et al. Amygdala-dependent fear is regulated by Oprl1 in mice and humans with PTSD. Sci Transl Med. 2013;5:188ra73.
Narendran R, Tollefson S, Fasenmyer K, Paris J, Himes ML, Lopresti B, et al. Decreased Nociceptin Receptors Are Related to Resilience and Recovery in College Women Who Have Experienced Sexual Violence: Therapeutic Implications for Posttraumatic Stress Disorder. Biol Psychiatry. 2019;85:1056–1064.
Toll L, Cippitelli A, Ozawa A. The NOP receptor system in neurological and psychiatric disorders: discrepancies, peculiarities and clinical progress in developing targeted therapies. CNS Drugs. 2021;35:591–607.
Zaveri NT. Nociceptin Opioid Receptor (NOP) as a therapeutic target: progress in translation from preclinical research to clinical utility. J Med Chem. 2016;59:7011–28.
Al Yacoub ON, Awwad HO, Zhang Y, Standifer KM. Therapeutic potential of nociceptin/orphanin FQ peptide (NOP) receptor modulators for treatment of traumatic brain injury, traumatic stress, and their co-morbidities. Pharm Ther. 2022;231:107982.
Gavioli EC, Holanda VAD, Calo G, Ruzza C. Nociceptin/orphanin FQ receptor system blockade as an innovative strategy for increasing resilience to stress. Peptides. 2021;141:170548.
Post A, Smart TS, Krikke-Workel J, Dawson GR, Harmer CJ, Browning M, et al. A selective nociceptin receptor antagonist to treat depression: evidence from preclinical and clinical studies. Neuropsychopharmacology. 2016;41:1803–12.
Ubaldi M, Cannella N, Borruto AM, Petrella M, Micioni Di Bonaventura MV, Soverchia L, et al. Role of nociceptin/orphanin FQ-NOP receptor system in the regulation of stress-related disorders. Int J Mol Sci. 2021;22:12956.
Berthele A, Platzer S, Dworzak D, Schadrack J, Mahal B, Buttner A, et al. [3H]-nociceptin ligand-binding and nociceptin opioid receptor mrna expression in the human brain. Neuroscience. 2003;121:629–40.
Neal CR Jr, Mansour A, Reinscheid R, Nothacker HP, Civelli O, Akil H, et al. Opioid receptor-like (ORL1) receptor distribution in the rat central nervous system: comparison of ORL1 receptor mRNA expression with (125)I- [(14)Tyr]-orphanin FQ binding. J Comp Neurol. 1999;412:563–605.
Neal CR Jr, Mansour A, Reinscheid R, Nothacker HP, Civelli O, Watson SJ Jr. Localization of orphanin FQ (nociceptin) peptide and messenger RNA in the central nervous system of the rat. J Comp Neurol. 1999;406:503–47.
Ozawa A, Brunori G, Mercatelli D, Wu J, Cippitelli A, Zou B, et al. Knock-in mice with NOP-eGFP receptors identify receptor cellular and regional localization. J Neurosci. 2015;35:11682–93.
Mouledous L. The nociceptin/orphanin FQ System and the Regulation of Memory. Handb Exp Pharm. 2019;254:259–78.
Higgins GA, Kew JN, Richards JG, Takeshima H, Jenck F, Adam G, et al. A combined pharmacological and genetic approach to investigate the role of orphanin FQ in learning and memory. Eur J Neurosci. 2002;15:911–22.
Sandin J, Georgieva J, Schott PA, Ogren SO, Terenius L. Nociceptin/orphanin FQ microinjected into hippocampus impairs spatial learning in rats. Eur J Neurosci. 1997;9:194–7.
Fornari RV, Soares JC, Ferreira TL, Moreira KM, Oliveira MG. Effects of nociceptin/orphanin FQ in the acquisition of contextual and tone fear conditioning in rats. Behav Neurosci. 2008;122:98–106.
Goeldner C, Reiss D, Wichmann J, Kieffer BL, Ouagazzal AM. Activation of nociceptin opioid peptide (NOP) receptor impairs contextual fear learning in mice through glutamatergic mechanisms. Neurobiol Learn Mem. 2009;91:393–401.
Mamiya T, Noda Y, Nishi M, Takeshima H, Nabeshima T. Nociceptin system plays a role in the memory retention: involvement of naloxone benzoylhydrazone binding sites. Neuroreport. 1999;10:1171–5.
Rekik K, Faria Da Silva R, Colom M, Pacifico S, Zaveri NT, Calo G, et al. Activation of nociceptin/orphanin FQ receptors inhibits contextual fear memory reconsolidation. Neuropharmacology. 2017;125:39–49.
Goeldner C, Reiss D, Wichmann J, Meziane H, Kieffer BL, Ouagazzal AM. Nociceptin receptor impairs recognition memory via interaction with NMDA receptor-dependent mitogen-activated protein kinase/extracellular signal-regulated kinase signaling in the hippocampus. J Neurosci. 2008;28:2190–8.
Devine DP, Hoversten MT, Ueda Y, Akil H. Nociceptin/orphanin FQ content is decreased in forebrain neurones during acute stress. J Neuroendocr. 2003;15:69–74.
Nativio P, Pascale E, Maffei A, Scaccianoce S, Passarelli F. Effect of stress on hippocampal nociceptin expression in the rat. Stress. 2012;15:378–84.
Green MK, Devine DP. Nociceptin/orphanin FQ and NOP receptor gene regulation after acute or repeated social defeat stress. Neuropeptides. 2009;43:507–14.
Reiss D, Wolter-Sutter A, Krezel W, Ouagazzal AM. Effects of social crowding on emotionality and expression of hippocampal nociceptin/orphanin FQ system transcripts in mice. Behav Brain Res. 2007;184:167–73.
Zhang Y, Gandhi PR, Standifer KM. Increased nociceptive sensitivity and nociceptin/orphanin FQ levels in a rat model of PTSD. Mol Pain. 2012;8:76.
Vitale G, Ruggieri V, Filaferro M, Frigeri C, Alboni S, Tascedda F, et al. Chronic treatment with the selective NOP receptor antagonist [Nphe 1, Arg 14, Lys 15]N/OFQ-NH 2 (UFP-101) reverses the behavioural and biochemical effects of unpredictable chronic mild stress in rats. Psychopharmacol Berl. 2009;207:173–89.
David DJ, Samuels BA, Rainer Q, Wang JW, Marsteller D, Mendez I, et al. Neurogenesis-dependent and -independent effects of fluoxetine in an animal model of anxiety/depression. Neuron. 2009;62:479–93.
Andero R. Nociceptin and the nociceptin receptor in learning and memory. Prog Neuropsychopharmacol Biol Psychiatry. 2015;62:45–50.
Sandin J, Ogren SO, Terenius L. Nociceptin/orphanin FQ modulates spatial learning via ORL-1 receptors in the dorsal hippocampus of the rat. Brain Res. 2004;997:222–33.
Adem A, Madjid N, Kahl U, Holst S, Sadek B, Sandin J, et al. Nociceptin and the NOP receptor in aversive learning in mice. Eur Neuropsychopharmacol. 2017;27:1298–307.
Delaney G, Dawe KL, Hogan R, Hunjan T, Roper J, Hazell G, et al. Role of nociceptin/orphanin FQ and NOP receptors in the response to acute and repeated restraint stress in rats. J Neuroendocr. 2012;24:1527–41.
D’Oliveira da Silva F, Azevedo Neto J, Sturaro C, Guarino A, Robert C, Gavioli EC, et al. The NOP antagonist BTRX-246040 increases stress resilience in mice without affecting adult neurogenesis in the hippocampus. Neuropharmacology. 2022;212:109077.
Fulford AJ. Endogenous nociceptin system involvement in stress responses and anxiety behavior. Vitam Horm. 2015;97:267–93.
Zhang Y, Simpson-Durand CD, Standifer KM, Nociceptin/orphanin FQ. peptide receptor antagonist JTC-801 reverses pain and anxiety symptoms in a rat model of post-traumatic stress disorder. Br J Pharm. 2015;172:571–82.
Conrad CD. A critical review of chronic stress effects on spatial learning and memory. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34:742–55.
Finsterwald C, Alberini CM. Stress and glucocorticoid receptor-dependent mechanisms in long-term memory: from adaptive responses to psychopathologies. Neurobiol Learn Mem. 2014;112:17–29.
McEwen BS, Bowles NP, Gray JD, Hill MN, Hunter RG, Karatsoreos IN, et al. Mechanisms of stress in the brain. Nat Neurosci. 2015;18:1353–63.
Chao OY, Nikolaus S, Yang YM, Huston JP. Neuronal circuitry for recognition memory of object and place in rodent models. Neurosci Biobehav Rev. 2022;141:104855.
Brown ES. Effects of glucocorticoids on mood, memory, and the hippocampus. Treatment and preventive therapy. Ann N Acad Sci. 2009;1179:41–55.
Barker GR, Warburton EC. When is the hippocampus involved in recognition memory? J Neurosci. 2011;31:10721–31.
Cohen SJ, Munchow AH, Rios LM, Zhang G, Asgeirsdottir HN, Stackman RW Jr. The rodent hippocampus is essential for nonspatial object memory. Curr Biol. 2013;23:1685–90.
Rebola N, Carta M, Mulle C. Operation and plasticity of hippocampal CA3 circuits: implications for memory encoding. Nat Rev Neurosci. 2017;18:208–20.
Stefanelli T, Bertollini C, Luscher C, Muller D, Mendez P. Hippocampal somatostatin interneurons control the size of neuronal memory ensembles. Neuron. 2016;89:1074–85.
Topolnik L, Tamboli S. The role of inhibitory circuits in hippocampal memory processing. Nat Rev Neurosci. 2022;23:476–92.
Ikeda K, Watanabe M, Ichikawa T, Kobayashi T, Yano R, Kumanishi T. Distribution of prepro-nociceptin/orphanin FQ mRNA and its receptor mRNA in developing and adult mouse central nervous systems. J Comp Neurol. 1998;399:139–51.
Law J, Ibarguen-Vargas Y, Belzung C, Surget A. Decline of hippocampal stress reactivity and neuronal ensemble coherence in a mouse model of depression. Psychoneuroendocrinology. 2016;67:113–23.
Tomar A, McHugh TJ. The impact of stress on the hippocampal spatial code. Trends Neurosci. 2022;45:120–32.
Fee C, Banasr M, Sibille E. Somatostatin-positive gamma-aminobutyric acid interneuron deficits in depression: cortical microcircuit and therapeutic perspectives. Biol Psychiatry. 2017;82:549–59.
Prevot T, Sibille E. Altered GABA-mediated information processing and cognitive dysfunctions in depression and other brain disorders. Mol Psychiatry. 2021;26:151–67.
Cavallini S, Marino S, Beani L, Bianchi C, Siniscalchi A. Nociceptin inhibition of acetylcholine efflux from different brain areas. Neuroreport. 2003;14:2167–70.
Uezu K, Sano A, Sei H, Toida K, Houtani T, Sugimoto T, et al. Enhanced hippocampal acetylcholine release in nociceptin-receptor knockout mice. Brain Res. 2005;1050:118–23.
Watanabe Y, Gould E, Cameron HA, Daniels DC, McEwen BS. Phenytoin prevents stress- and corticosterone-induced atrophy of CA3 pyramidal neurons. Hippocampus. 1992;2:431–5.
Woolley CS, Gould E, McEwen BS. Exposure to excess glucocorticoids alters dendritic morphology of adult hippocampal pyramidal neurons. Brain Res. 1990;531:225–31.
Caradonna SG, Einhorn NR, Saudagar V, Khalil H, Petty GH, Lihagen A, et al. Corticosterone induces discrete epigenetic signatures in the dorsal and ventral hippocampus that depend upon sex and genotype: focus on methylated Nr3c1 gene. Transl Psychiatry. 2022;12:109.
Zhang Y, Schalo I, Durand C, Standifer KM. Sex differences in nociceptin/orphanin FQ peptide receptor-mediated pain and anxiety symptoms in a preclinical model of post-traumatic stress disorder. Front Psychiatry. 2018;9:731.
Witkin JM, Wallace TL, Martin WJ. Therapeutic approaches for NOP receptor antagonists in neurobehavioral disorders: clinical studies in major depressive disorder and alcohol use disorder with BTRX-246040 (LY2940094). Handb Exp Pharm. 2019;254:399–415.
Acknowledgements
We thank the CBI-Anexplo mouse facility. We also thank the LITC imaging platform of Toulouse TRI, and Sébastien Gauzin from the Mouse Behavioral Core of CBI-Anexplo for support. We thank Dr Catherine Mollereau for critical reading of the manuscript.
Funding
This work was supported by the Centre National de la Recherche Scientifique (to LM), the Research Center on Animal Cognition (to LM), the French Ministry of Research (to FD), NIH grants, R21DA034929, and P50MH119467 (to MB).
Author information
Authors and Affiliations
Contributions
Conceptualization: FD, BG, LM; Methodology: FD, FC, MB, LM; Resources: CP, MB; Formal analysis: FD, BG, LM; Investigation: FD, CR, EL, LM; Visualization: FD; Writing—Original Draft: FD, LM; Writing—Review & Editing: FD, MB, BG, FC, LM; Funding Acquisition: FD, MB, LM; Supervision: FC, LM.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
D’Oliveira da Silva, F., Robert, C., Lardant, E. et al. Targeting Nociceptin/Orphanin FQ receptor to rescue cognitive symptoms in a mouse neuroendocrine model of chronic stress. Mol Psychiatry (2023). https://doi.org/10.1038/s41380-023-02363-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41380-023-02363-x