Abstract
Aberrant anatomical brain connections in attention-deficit/hyperactivity disorder (ADHD) are reported inconsistently across diffusion weighted imaging (DWI) studies. Based on a pre-registered protocol (Prospero: CRD42021259192), we searched PubMed, Ovid, and Web of Knowledge until 26/03/2022 to conduct a systematic review of DWI studies. We performed a quality assessment based on imaging acquisition, preprocessing, and analysis. Using signed differential mapping, we meta-analyzed a subset of the retrieved studies amenable to quantitative evidence synthesis, i.e., tract-based spatial statistics (TBSS) studies, in individuals of any age and, separately, in children, adults, and high-quality datasets. Finally, we conducted meta-regressions to test the effect of age, sex, and medication-naïvety. We included 129 studies (6739 ADHD participants and 6476 controls), of which 25 TBSS studies provided peak coordinates for case-control differences in fractional anisotropy (FA)(32 datasets) and 18 in mean diffusivity (MD)(23 datasets). The systematic review highlighted white matter alterations (especially reduced FA) in projection, commissural and association pathways of individuals with ADHD, which were associated with symptom severity and cognitive deficits. The meta-analysis showed a consistent reduced FA in the splenium and body of the corpus callosum, extending to the cingulum. Lower FA was related to older age, and case-control differences did not survive in the pediatric meta-analysis. About 68% of studies were of low quality, mainly due to acquisitions with non-isotropic voxels or lack of motion correction; and the sensitivity analysis in high-quality datasets yielded no significant results. Findings suggest prominent alterations in posterior interhemispheric connections subserving cognitive and motor functions affected in ADHD, although these might be influenced by non-optimal acquisition parameters/preprocessing. Absence of findings in children may be related to the late development of callosal fibers, which may enhance case-control differences in adulthood. Clinicodemographic and methodological differences were major barriers to consistency and comparability among studies, and should be addressed in future investigations.
Similar content being viewed by others
Introduction
Attention-deficit/hyperactivity disorder (ADHD) is a neurodevelopmental condition characterized by age-inappropriate inattentive and/or hyperactive-impulsive symptoms [1]. Cognitively, individuals with ADHD may present with deficits in executive functions, such as motor inhibition, attention, and/or working memory [2]. It is commonly diagnosed in childhood, with community prevalence between 2-7% [3], but its impairing symptoms persists in adulthood in up to 65% of cases, and are associated with poor social and occupational outcomes [4]. Co-occurrent disorders, from autism spectrum disorder (ASD) to affective and substance abuse disorders, are often observed [5, 6]. Multiple genetic and environmental factors contribute to ADHD, but it is unclear how they interplay with brain development to produce symptoms and cognitive deficits [2]. Understanding the underlying neuropathophysiology is crucial to develop and tailor behavioral, pharmacological or brain-based treatments.
Meta-analyses of structural and functional neuroimaging studies have identified several case-control differences, but mainly focused on regional alterations [2]. However, brain regions operate as neural networks, and there is increasing evidence that anatomical brain connections are also affected in ADHD [7]. Diffusion-weighted imaging (DWI) is the only non-invasive imaging method that allows us to study the anatomy of brain connections in the living human. It measures the diffusion of water molecules, which in the brain is restricted by structures such as myelin and axons, providing information on the microstructural organization of white matter tracts [8, 9] (Box 1).
The first systematic review of diffusion imaging studies in ADHD was published in 2012 and included 15 studies, mostly in pediatric samples [10]. The meta-analysis of nine of the included studies revealed diffuse alterations mainly affecting fronto-striato-cerebellar connections [10]. The two following meta-analyses, respectively published in 2016 and 2018, included VBA and/or TBSS studies and mainly identified regions of reduced FA in posterior commissural fibers [11, 12]. The separate analysis of TBSS and VBA studies also allowed the identification of regions of increased FA in the corpus callosum (CC) and cingulum [11]. These evidence syntheses were important to elucidate the most consistent findings in ADHD. Still, they did not consider studies using techniques not amenable to meta-analysis, and included a very limited number of studies in adults. Since then, DWI has benefitted from considerable technological advances, and the quality and number of published studies in the field of ADHD has progressively increased. A more comprehensive review with meta-analysis is therefore timely to provide a broader view of the findings, identify the most robust evidence, and highlight methodological considerations. Therefore, we conducted a comprehensive systematic review of DWI studies in ADHD, including a quality assessment of imaging data acquisition, preprocessing and analysis. We then performed, wherever possible, meta-analyses including individuals with ADHD of any age and, separately, children and adults. Finally, we conducted meta-regressions to test the effect of age, sex, and medication-naïve status, followed by a sensitivity analysis including only high-quality datasets.
Methods
This study followed a preregistered protocol (PROSPERO 2021 CRD42021259192) and is reported in line with the 2020 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement [13].
Data sources
We searched the following electronic databases: PubMed (Medline), Ovid databases (Ovid MEDLINE®, EMBASE Classic+EMBASE, PsycINFO), and Web of Knowledge (including Web of Science, Biological Abstracts, BIOSIS, Food science and technology abstracts), without language and date restrictions. The search was first conducted on the 22nd June 2021 and updated on the 26th March 2022. Search terms and syntax for each electronic database are reported in the Supplementary material. The reference lists of previous reviews were hand-searched for any additional eligible studies that could have been missed in the electronic searches.
Identification and selection of studies
First, two authors (VP and TI) independently screened titles and abstracts of all nonduplicated papers and agreed on a final list of studies that proceeded to full-text screening. Then, these two authors independently assessed the eligibility of these studies for the systematic review and meta-analysis. Any discrepancy between the two authors was resolved by a third senior author (SC).
Study selection
Studies were included in the systematic review if they:
-
1.
were peer-reviewed, indicating methodological adequacy, in line with recent meta-analyses [11, 12];
-
2.
recruited individuals diagnosed with ADHD based on the criteria of the Diagnostic and Statistical Manual of Mental Disorders (DSM-III or following editions) or International Classification of Diseases (ICD-9 or 10) and typically developing (TD) controls;
-
3.
collected diffusion imaging data from both ADHD participants and TD controls.
According to our pre-registered protocol, we assessed the feasibility of conducting a meta-analysis of eligible whole-brain studies (i.e., TBSS and VBA studies), provided their number was increased sufficiently (i.e., by 50%), as compared to the last published meta-analysis before the start of this study [11], to justify a new meta-analysis. Additionally, studies were eligible for the meta-analysis only if they compared whole-brain diffusion imaging data (any metric) between individuals with ADHD and TD controls.
Data extraction
Two authors (TI and YL) independently extracted information from the studies selected for the systematic review. Any discrepancy between the two authors was resolved by a third author (VP). Data extracted from all studies in the systemic review included: sample size, demographic and clinical characteristics (i.e., age, sex, total intelligent quotient (IQ), ADHD presentation, comorbidities, and medication-naïve status); analytic method; significant case-control comparisons and associations between diffusion metrics and symptom severity or cognitive performance. Further, data on imaging data acquisition, preprocessing and analysis were extracted for the quality assessment (see below). Authors were contacted for missing data on imaging parameters included in the quality assessment.
For the meta-analyses, we extracted peak coordinates and their effect sizes for FA, MD, AD and RD contrasts. In studies not providing exact effect sizes for peak coordinates, the study threshold for significance was interpreted as the effect size, as in previous reports [11]. In cases where peak coordinates and/or effect sizes were not provided, we contacted the corresponding authors to obtain the missing data. P-values of peak coordinates were converted to t-values using the anisotropic effect size signed differential mapping (AES-SDM) utility (http://www.sdmproject.com/utilities/?show=Statistics) [14]. Peak coordinates are available as Supplementary material.
Quality assessment
In the absence of an established tool to rate the quality of DWI studies, criteria were identified based on published recommendations (listed in Supplementary Table S3). A traffic light system was then implemented, and studies were assigned a low/medium/high-quality rating separately for imaging data acquisition, preprocessing, and analysis, as well as an overall rating based on the worse single rating. Two authors (SL and TTN) independently completed the quality assessment and discrepancies were resolved by a third author (JHZ).
Meta-analysis and meta-regressions
We ran meta-analyses for diffusion metrics that had peak coordinates available from at least five suitable studies. As in previous meta-analyses [11, 12], we used the Signed Differential Mapping (SDM) software, version 6.21 [15] (https://www.sdmproject.com/), to analyze regional differences in tract metrics between ADHD and TD control groups; and used the TBSS template for TBSS studies. Also consistently with prior studies [11, 12], we used a random effects model, and the same statistical threshold that was previously applied (p < 0.005 at the voxel level with an extent threshold of 10 voxels). Meta-analyses were repeated in the pediatric sample (i.e., children/adolescents <18 years) and adults separately; and then in children (<12 years) and adolescents separately (Supplementary Table S2). We also conducted a post-hoc sensitivity analysis only including datasets judged of high quality. Finally, we ran meta-regression analyses to test the linear effects of age, percentage of males and medication-naïve subjects. We chose these variables because they have been associated with variation in white matter characteristics [16, 17]. Further, given that a previous report suggested that stimulant treatment could affect FA measures in children, but not adults [16], we tested the potential confounding effect of treatment exposure in the meta-regression of age.
Results
Systematic review
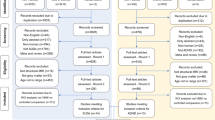
As shown in the PRISMA flow diagram (Fig. 1), from a pool of 956 possibly relevant references, we included 129 studies (96 in children, 25 in adults and 8 including both age groups), for a total of 6739 ADHD participants and 6476 controls. As many studies in pediatric samples included both children and adolescents, in the narrative synthesis we aggregated them under ‘children’ (i.e. <18 years). However, in the meta-analysis, we also considered them separately (Supplementary Table S2). With regards to the imaging data analytic approach, the retained studies used one or a combination of the following: TBSS (43 studies), tractography (38 studies), ROI (22 studies), VBA (16 studies), network/graph analysis (17 studies) or other techniques (e.g., fixel-based analysis) (3 studies). Here, we first summarize the results of these studies as a narrative review, according to the topographical organization of the main brain connections [18]. These can be grouped in projection (cortico-subcortical) pathways, which relay sensory-motor information; association (intra-hemispheric) pathways, which integrate functions of brain regions within the same hemisphere; and commissural (inter-hemispheric) pathways, which support information transfer between the two hemispheres [18]. Associations between diffusion metrics and symptoms/cognitive deficits are summarized in Figs. 2 and 3 and detailed in Supplementary material (page 5). Detailed characteristics of included studies are reported in Tables 1 and 2. Excluded studies are reported, with reasons, in the Supplementary material (page 3 and Supplementary Table S1). We then discuss the quality assessment and present the results of the meta-analyses and meta-regressions.
This figure displays the distinct components of the frontostriatal and thalamo-frontal connections (panel a); cerebellar pathways (panel b); and the corticospinal tract (panel c). Significant associations between tract metrics and cognitive deficits or symptoms are reported. Additional abbreviations: ATR anterior thalamic radiation, FS frontostriatal tract.
This figure shows the three branches of the superior longitudinal fasciculus (SLF) (Panel a); the cingulum (CING), uncinate (UNC), inferior longitudinal fasciculus (ILF) and inferior fronto-occipital fasciculus (IFOF) (Panel b); and the main subdivisions of the corpus callosum (CC) (Panel c). Significant associations between tract metrics and cognitive deficits or symptoms are reported.
Projection pathways
Fronto-striato-thalamic circuits
Most studies focused on fronto-striato-thalamic circuits (Fig. 2). Thirty-two studies reported tract metric alterations within distinct components of the frontostriatal pathways (connecting the frontal white matter to the striatum through the corona radiata and internal capsule) of children with ADHD as compared to controls [19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50]. Reduced FA was reported by seven tractography [23, 24, 27, 29, 36, 41, 45], four TBSS [25, 28, 37, 46], four VBA [19, 32, 40, 49] and two ROI [33, 38] studies. Increased FA was reported by only five studies, which either used a VBA [26, 35, 39] or a TBSS approach [43, 44]. Finally, four studies identified differences between ADHD presentations in children [34, 48, 51, 52]. Considering adult/mixed samples, 10 studies reported diffusion alterations in individuals with ADHD as compared to controls [53,54,55,56,57,58,59,60,61,62]. Of these, five reported reduced FA [55, 57, 58, 60, 62] and only one increased FA [53]. Further, differences in frontostriatal tracts were reported between ADHD persisters (i.e., those with a childhood diagnosis persisting in adulthood) and remitters [63]. Several studies also investigated brain-behavior relationships (Fig. 2).
Considering the thalamus and the anterior thalamic radiation (ATR), 13 studies reported significant case-control differences in children [20,21,22, 28, 30, 43, 50, 64,65,66,67,68,69]. Among these, two reported reduced FA [67, 68], and two increased FA [43, 66]. In adult/mixed samples, alterations were only identified by seven studies [56, 58, 70,71,72,73,74], of which four reported reduced FA [58, 70,71,72] and one both reduced and increased FA [74]. In summary, reduced FA was the most consistently reported alteration within fronto-striato-thalamic circuits, often observed bilaterally. Tract metrics were significantly correlated with both clinical and neuropsychological characteristics (Fig. 2).
Corticospinal tract (CST)
Fifteen studies reported diffusion alterations within the posterior limb of the internal capsule/CST in children with ADHD [25, 37, 38, 44, 48, 66, 67, 75,76,77,78,79,80,81,82]. Among these, reduced FA was noted by four TBSS [25, 37, 67, 75], one tractography [82] and one ROI study [80]. Increased FA was only identified by two TBSS [44, 66] and one tractography study [76]. Only two studies identified alterations in adult/mixed samples and either reported increased [74] or decreased FA in the CST [58]. In sum, the CST has been mainly investigated in children and most studies have reported reduced FA. Diffusion metrics have been associated with clinical symptoms but also task performance (Fig. 2).
Cerebellar pathways
Eight studies reported alterations in the cerebellum or the middle cerebellar peduncle (MCP) in children with ADHD [19, 20, 25, 32, 37, 49, 64, 83]. Among these, reduced FA was identified by three VBA [19, 32, 49], two TBSS [25, 37] and one ROI study [83]. No study reported increased FA. Only two studies identified alterations in the cerebellum/MCP of adults with ADHD [56, 74]. In sum, reduced FA was the most consistently reported alterations within the cerebellum/MCP. Only one study reported reduced FA in the inferior cerebellar peduncle [37]. Tract metrics were significantly associated with symptoms and cognitive performance (Fig. 2).
Commissural pathways
The most investigated commissural pathway was the CC (Fig. 3). We identified 17 studies that reported tract metric alterations in the CC of ADHD children as compared to controls [20, 21, 25, 28, 40, 43, 46, 48, 67, 75, 77, 84,85,86,87,88,89]. Reduced FA was found in seven TBSS studies [25, 28, 46, 67, 75, 84, 88] and in one VBA study [40], especially in the splenium of the CC. ROI studies reported conflicting results, with either increased [20] or decreased FA in the splenium of the CC [85]. Tractography studies reported reduced FA [86, 89]. Differences in diffusion metrics were also noted among ADHD presentations [48, 51] and treated/untreated individuals [87]. Eleven adult/mixed sample studies reported tract metric alterations in the CC [53,54,55, 57, 58, 70, 71, 74, 90,91,92]. Reduced FA was found in four TBSS studies especially in the body and splenium of the CC [55, 58, 70, 91], two VBA [54, 71] and an ROI study [92]. Finally, a tractography study in a mixed pediatric-adult sample reported reduced FA in the splenium of the CC [57], and another reduced FA in callosal fibers [60]. Overall, reduced FA was the most consistently reported alteration, especially in the splenium of the CC. Tract metrics were significantly correlated with both clinical and neuropsychological characteristics (Fig. 3).
Association pathways
Superior longitudinal fasciculus (SLF)
Seventeen studies reported tract metric alterations in the SLF of ADHD children as compared to controls [21, 23, 24, 37, 38, 46, 48, 50, 65, 70, 77, 78, 80, 84, 88, 93, 94]. Among these, reduced FA was observed in five TBSS [37, 46, 70, 84, 88], two tractography [23, 24], and one ROI study [80]. Only a TBSS study reported increased FA [94]. Differences in tract metrics were also observed between ADHD presentations [48, 51] and sexes [88]. Nine adult/mixed sample studies [53, 55, 58, 60, 71, 74, 95,96,97] reported case-control differences in SLF metrics. Of these, three TBSS [58, 70, 95], one tract-based analysis [97], one tractography [60], and one ROI study [96] noted reduced FA in individuals with ADHD as compared to controls. Further, a study combining an ROI and machine learning approach identified both reduced and increased FA in the SLF of adults with ADHD [74]. Sex differences were also observed by a tractography study in a mixed pediatric-adult sample, with lower FA in females [57]. Overall, reduced FA was the most consistently reported alteration, bilaterally or in either hemisphere with comparable frequency. Tract metrics were significantly correlated with both symptom severity and cognitive performance (Fig. 3).
Cingulum bundle
Twenty-one studies in children reported tract alterations in the cingulum of ADHD patients as compared to controls [21,22,23,24, 34, 37,38,39, 47, 48, 50, 66, 67, 69, 78, 88, 94, 98,99,100,101]. Reduced FA was noted by three TBSS [67, 88, 99] and four tractography studies [23, 24, 100, 101]. Increased FA was observed in two TBSS [66, 94] and two VBA studies [39, 98]. Differences were also identified among ADHD presentations [51, 52]. Eight studies in adult/mixed samples reported tract alterations in the cingulum [53, 54, 59, 71, 73, 74, 96, 102]. Reduced FA was noted by two VBA studies [54, 71], one tractography [102] and one ROI study [96]. Conversely, one VBA study reported increased FA [53] and one ROI study both increased and reduced FA [74]. Overall, both increased and reduced FA in the cingulum have been reported, bilaterally or in either hemisphere with equal frequency. Diffusion characteristics have been associated with variation in both clinical and cognitive profiles (Fig. 3).
Uncinate fasciculus
Ten studies in children and three in adults reported tract metric alterations in the uncinate fasciculus of individuals with ADHD as compared to controls [37, 38, 43, 53, 71, 74, 82, 87, 94, 97]. Of these, TBSS studies either reported increased [43, 94] or decreased FA [37, 82] in ADHD children. In adults, two VBA study reported increased FA [53, 71], whilst a tract-based analysis observed reduced FA [97]. Overall, main differences were observed either bilaterally or in the left uncinate, and were associated with inattentive-emotional symptoms, and cognitive deficits (Fig. 3).
Inferior longitudinal fasciculus (ILF)
Thirteen studies reported case-control differences in the ILF of ADHD children versus controls [21, 25, 37, 38, 46, 66, 75, 78, 79, 82, 87, 94, 103]. Reduced FA in the ILF was noted by four TBSS [25, 37, 46, 75], an ROI [103], and two tractography studies [79, 82]. Two studies reported increased FA [66, 94]. Differences were also identified between females and males [88] and between treated and untreated individuals [87]. Five studies in adult/mixed samples reported case-control differences in the ILF [58, 60, 74, 102, 104]. Using different techniques, three of these studies observed reduced FA [58, 102, 104], one increased FA [60], and one both increased and reduced FA [74]. Overall, reduced FA was the most frequently reported tract alteration, either bilaterally or in the left hemisphere, and was often associated with inattention (Fig. 3).
Inferior fronto-occipital fasciculus (IFOF)
Eleven studies in pediatric samples reported case-control differences in the IFOF [21, 43, 46, 48, 67, 75, 78, 79, 82, 84, 103]. Reduced FA was noted in four TBSS studies [46, 67, 75, 84], an ROI [103], and two tractography studies [79, 82]. Increased FA was only reported by one TBSS study [43]. Six studies reported case-control differences in the IFOF in adult or mixed children-adult samples [53, 58, 71, 74, 97, 104]. Of these, three studies using different techniques reported reduced FA [58, 74, 97] and one both reduced and increased FA [71]. Sex differences were also noted in a mixed pediatric-adult sample [57]. Overall, as for the ILF, reduced FA was the most frequently reported tract alteration, either bilaterally or in the left hemisphere, and was associated with inattention and emotional problems (Fig. 3).
Quality assessment
Among the included studies, 68.7% were judged to be of low overall quality, mainly due to factors related to pre-processing (rated low-quality in 54% of studies) and/or acquisition parameters (rated low-quality in 46% of reports). Low-quality ratings for preprocessing were mainly due to lack of motion correction/quality checks; those for acquisition to the use of non-isotropic voxels or lack of information. The quality assessment of individual studies is reported in Supplementary Tables S4 and 5.
Meta-analyses and meta-regressions
Meta-analysis of TBSS studies comparing ADHD versus controls
Of the 43 TBSS studies included in the systematic review, 25 had available peak coordinates for case-control differences in FA and 18 in MD from non-duplicated datasets (see Supplementary Table S2). Therefore, these studies were included in two separate meta-analyses. The first meta-analysis included 25 TBSS studies, for a total of 32 datasets (26 in children and 6 in adults), comparing FA between 1348 individuals with ADHD and 1354 TD controls. As shown in Fig. 4 and Supplementary Table S6, the SDM analysis identified two clusters showing reduced FA values in the ADHD group compared with the TD control group. The right splenium of the CC, extending to the posterior cingulum, showed the most significant effect size and cluster extent (Fig. 4). The right body of CC exhibited the second-largest effect size and cluster extent. No clusters exhibited increased FA values in the ADHD group as compared with TD controls. The second meta-analysis included 18 studies, for a total of 23 datasets (19 in children and 4 in adults), comparing MD between 1051 participants with ADHD and 1101 controls, and did not identify any significant clusters. Due to the limited number of studies reporting peak coordinates for AD and RD (three and four, respectively), it was not possible to carry out meta-analyses for these metrics. Finally, the systematic review identified 16 studies that used a VBA approach. However, among these, only one study with available peak coordinates was not included in the previous review by Aoki et al., 2018 [11]. Therefore, we did not re-run the meta-analysis based on our pre-published protocol.
The meta-analysis of TBSS studies comparing fractional anisotropy (FA) between individuals with ADHD (any age) and typically developing (TD) controls showed that individuals with ADHD had lower FA in the splenium of the corpus callosum (CC), extending to the posterior cingulum, and in the body of the CC. Peak coordinates are reported in Supplementary Table S6.
Meta-analyses of TBSS studies in children and adult samples
The meta-analyses of TBSS studies investigating case-control differences in FA in the pediatric sample (<18 years) showed no significant clusters. Similarly, we did not observe significant clusters when we separated children (<12 years) and adolescents. By contrast, the meta-analysis including only adult studies identified five clusters showing reduced FA in individuals with ADHD as compared to controls (Supplementary Table S7). The right splenium of the CC exhibited the largest effect size and cluster extent, and the right ATR showed the second-largest effect size and cluster extent. No clusters exhibited increased FA values in adults with ADHD as compared to controls. No significant cluster was identified when repeating the meta-analysis on case-control comparisons in MD separately in children and adults.
Meta-analysis of high-quality studies
In the sensitivity analysis considering only the 6 TBSS datasets rated of high quality (Supplementary Table S2), we did not observe significant differences in FA between ADHD and TD controls.
Meta-regression analyses
We ran three meta-regressions including the 25 TBSS studies (32 datasets) comparing FA between ADHD and TD controls. The first meta-regression tested the linear influence of age and identified two clusters with significant negative associations. In the right splenium and body of the CC, the differences between groups in reduced FA amplified with advancing age (Supplementary Table S8). No clusters displayed significant positive associations with age. These findings held after including treatment exposure as additional regressor (Supplementary Table S9). Meta-regressions accounting for the percentage of medication-naïve participants or the percentage of males included 23 studies (30 datasets) and 24 studies (31 datasets), respectively. We observed that ratios of medication-naïve/medicated individuals and males/females had no significant influence on FA.
Discussion
We conducted the most comprehensive systematic review of DWI studies in the field of ADHD, meta-analyzing studies amenable to quantitative synthesis. The systematic review identified widespread alterations (mainly reduced FA) in individuals with ADHD as compared to controls, mostly in the fronto-striatal pathways, cingulum, and CC. The meta-analysis of TBBS studies included 32 datasets (of which six in adults) and identified the most consistent FA reduction in the right splenium (extending to the posterior cingulum), followed by the body, of the CC. Meta-regressions showed that these effects were not affected by sex or exposure to ADHD medication. However, lower FA was related to older age, and case-control differences did not survive in the pediatric meta-analysis. Conversely, the meta-analysis in adults mainly identified reduced FA in the right splenium of the CC and ATR.
Alterations in the splenium and the body of the CC are in line with their roles in supporting cognitive and motor functions affected in ADHD. The splenium of the CC connects the occipital, temporal and posterior parietal lobes of the two hemispheres [105] and has been associated with visuospatial information transfer, processing speed, IQ, and behavior [106]. Posterior cortical regions underpin attention and fronto-parietal cognitive control networks [107, 108], and thus contribute to cognitive functions, such as attention, working memory and executive control, which are commonly impaired in ADHD [109, 110]. Further, the body of the CC primarily connects premotor, supplementary and primary motor cortices between the two hemispheres, and contributes to the modulation of motor activity [105]. Our findings are in line with previous meta-analyses of structural magnetic resonance imaging (MRI) data reporting consistent volumetric reductions in the splenium of the CC in individuals with ADHD as compared to controls [111, 112]; although more recent meta- and mega-analyses observed significant case-control differences only in children [113, 114]. Further, functional MRI meta-analyses have identified reduced parietal and temporal activations during cognitive control, attention and timing tasks [115,116,117,118]. Of note, the cluster located in the splenium of the CC extended to the cingulum, which connects regions subserving the default mode network (DMN) [119]. There is fMRI evidence for a poor anticorrelation between the DMN and task-positive networks, such as the fronto-parietal control and ventral attentive networks [120, 121]. This, according to the default mode interference theory of ADHD [122], may cause lapses of attention during cognitively demanding tasks [123, 124]. Notably, a prior study reported that white matter disruption in the splenium and body of the CC was associated with decreased resting-state functional connectivity in the DMN posterior cingulate cortex [125]. Taken together, our findings and prior studies suggest that altered anatomical connectivity within the splenium and body of the CC may disrupt the function of brain networks supporting cognitive and motor functions affected in ADHD or their interaction with the DMN.
Importantly, our findings not only confirm but also extend those of previous meta-analyses of DWI studies. Although the first meta-analysis combining nine VBA and TBSS studies reported more consistent alterations within fronto-striato-cerebellar connections, subsequent meta-analyses mainly identified altered FA within commissural fibers [11, 12, 126]. The first included ten TBSS studies and reported reduced FA in the splenium of the CC, right sagittal stratum and left tapetum, extending to the cingulum, ILF and IFOF [12]. The second performed two separate meta-analyses, including 12 TBSS and 13 VBA studies respectively. It confirmed a consistent reduced FA in the CC, in addition to the ILF, IFOF and SLF. However, the meta-analysis of VBA studies also identified regions of increased FA, e.g., in the midcingulate and anterior CC [11]. Finally, a more recent meta-analysis combining 24 TBSS and VBA studies confirmed a consistent FA reduction in the splenium of the CC, extending to the body and right posterior corona radiata [126]. The high consistency among the more recent meta-analyses may be due to their largely overlapping pool of included studies (e.g., almost 90% of articles included in Zhao et al., 2022 [126] were included in Aoki et al., 2018 [11]). Our findings from 32 TBSS datasets further support the consistently reported callosal alterations in ADHD extending to the cingulum. Different mechanisms may lead to reduced FA, such as altered myelination, axonal density/diameter or fiber crossing [127, 128]. As FA is a composite measure, we encourage future studies to consistently report additional metrics, e.g. AD and RD, as this may help understand whether white matter alterations are primary (e.g. related to myelination) or secondary to those in the gray matter from which the tract origins from (e.g. number of neurons). Both these mechanisms have been suggested as plausible in relation to ADHD pathophysiology [113, 129]. Further, both genetic and environmental factors may potentially contribute to the observed alterations. For instance, a recent genome-wide association (GWA) meta-analysis in ADHD identified, among others, genes related to myelination [130]. However, their patter of methylation, which may reflect environmental adversity, affect ADHD symptoms trajectories [131]. Thus, genetic and environmental factors may interplay to cause white matter abnormalities, which in turn are associated with ADHD symptoms. Overall, there are likely multiple alternative pathophysiologic pathways underpinning ADHD and brain alterations are not necessarily causal to symptoms, but might be co-occurrent manifestations or consequence of ADHD behaviors.
As reported by Chen et al. [12], we also observed a negative association between FA and age, with lower FA values in older individuals. However, in this prior meta-analysis, findings in the splenium of the CC survived in the pediatric meta-analysis, in contrast to our study. The absence of significant findings in our meta-analysis restricted to children is somehow unexpected given that prior structural MRI meta- and mega-analyses reported significant case-control volumetric and morphometric differences in children but not in adults with ADHD [113, 114]. For instance, reduced surface area and cortical thickness were identified in fronto-cingulate-temporo-occipital regions of ADHD children, but not in adults, compared to controls [114]. However, we believe that our findings could provide new important insight into the pathophysiology of ADHD across the lifespan. A possible explanation of differences between prior and our results might be related to the distinct developmental trajectories of the gray and white matter. While gray matter structural measures increase and reach their peak in childhood (~age 2 for cortical thickness; ~age 6 for gray matter volume; and ~11-12 years for surface area/cerebral volume) and then decrease in a curvilinear fashion [132]; FA in the CC increases in a curvilinear fashion with age, peaking between 21 and 29 years [133, 134]. Hence a delayed, less steep increase in FA with age in ADHD compared to healthy subjects would be reflected in larger case-control differences in older subjects; whilst a less steep decrease in gray matter measures would be more pronounced in childhood. The reduction in FA we observed in the splenium and body of the CC of individuals with ADHD could hence reflect a delay in white matter maturation, parallel to that observed in posterior cortices [135], which are interhemispherically connected by the posterior CC. However, longitudinal studies investigating developmental trajectories are needed to clarify the course of brain structure and connectivity alterations in ADHD.
We also observed a lack of spatial convergence in the sensitivity analysis only including high-quality studies. These findings need to be interpreted with caution because, although the quality assessment was based on published recommendations, there is not an available, established gold standard quality rating tool for DWI studies. Further, this meta-analysis only included six studies. Nevertheless, these findings suggest that the case-control differences identified in DWI meta-analyses might be influenced by non-optimal acquisition parameters or preprocessing. Future studies should be encouraged to follow the existing recommendations on imaging data acquisition, preprocessing, and analysis (referenced in Supplementary Table S3). For example, studies should (1) opt for isotropic voxels to avoid bias; (2) include sufficient gradient directions to ensure rotation invariance and improve precision; (3) spread multiple gradients unweighted among weighted volumes in the scan for similar noise profiles; (4) report sufficient acquisition and preprocessing information; (5) include additional scans for distortion correction; and (6) perform data quality control on the motion and report motion thresholds. Further, research in ADHD may benefit from methodological advances in the field. For instance, higher encoding resolution may improve sensitivity to signal and biophysical properties [136]; acceleration techniques may improve applications to younger populations [137]; and newer preprocessing methods may handle more types of artefacts [138]. Overall, methodological developments may improve the study quality and reliability of findings, thus fostering our understanding of brain microstructure and connectivity in ADHD.
Prior meta-analyses of ADHD studies in other imaging modalities also reported no significant spatial convergence [139, 140], and related this to methodological differences among studies and the high clinical and neurobiological heterogeneity of ADHD. In support to the latter observation, our systematic review indicates that differences may exist between ADHD presentations, females and males, treated versus untreated individuals, and participants with/without comorbidities. Further, a recent meta-analysis has shown that FA reduction in the splenium of the CC is a common feature of both ADHD and ASD, although the latter is characterized by additional white matter alterations in frontostriatal pathways [126]. These findings, together with the limited converging results in the whole sample meta-analysis and the absence of significant results in the pediatric meta-analysis, should encourage future studies to extend their investigations beyond case-control differences and determine whether subgroups of individuals with ADHD could be discerned based on white matter characteristics. The need to parse neuroanatomic heterogeneity has also been raised by studies in other MRI modalities [141], as this may improve our understanding of ADHD pathophysiology. Further, the stratification of the heterogenous ADHD population based on differences in the underlying neuroanatomy may pave the way to the development of new more targeted treatments (Parlatini et al., under review).
Limitations
Some limitations of this work should be considered, mostly related to limitations of the included studies. For instance, most studies included in the narrative review used traditional tensor-based methods, and only a minority included other imaging (e.g., q-ball) and analysis (e.g., graph theory) techniques. We encourage the use of state-of-the-art methods as this can also advance our understanding of ADHD. As in previous meta-analyses, we used a coordinate-based approach rather than the original t-statistic maps, as these are not publicly available, but this may limit the accuracy of the results [14]. We conducted meta-analyses of TBSS studies but did not replicate the one of VBA studies, according to our pre-published protocol, as we could only have included one additional study compared to Aoki et al. [11]. This reflects the general tendency in the current literature towards the use of TBSS as compared to VBA approaches. As in this previous work [11], we preferred not to combine TBSS and VBA approaches in a single meta-analysis because this would violate the assumption under the null hypothesis that the expected FA differences are equal at every voxel, and because there is evidence that the two approaches may produce non-converging findings [11]. We conducted separate meta-analyses of FA and MD, but could not investigate other diffusion metrics (e.g., RD and AD) due to the limited number of studies reporting them. We contacted authors for missing data, but peak coordinates were not available for eight studies. Our meta-analysis included a much greater number of studies than prior syntheses, which enhances robustness of findings; however, we encourage future investigations to provide full details of their results to limit potential reporting bias.
The studies included in the systematic review/meta-analyses mostly recruited small samples and were heterogeneous in terms of clinicodemographic characteristics. They included varying proportions of males and females and different ADHD presentations. Most studies included subjects previously exposed to medication, which can represent a potential confounder in connectivity measurements [16]. Our meta-regression analysis did not show any significant effect of previous exposure to stimulants; however, longitudinal studies in large samples are needed to disentangle the effects of development and treatment. Further, only few studies restricted the recruitment to comorbidity-free individuals. Most included different disorders or did not provide information, thus we were unable to control for comorbidities. This heterogeneity may have potentially influenced consistency among studies and our findings. For instance, about one fifth of individuals with ADHD also have ASD [142]. However, comorbidity was not officially allowed until the DSM-5 was published (2013) [1]; therefore, the most recent studies and those in pediatric samples may be enriched with comorbid cases. This may also have potentially contributed to the lack of convergent findings in our pediatric submeta-analysis. For instance, recent meta-analyses highlighted both shared and specific alterations in individuals with ADHD and ASD [126, 143]. One of these identified reduced FA in the CC of both ADHD and ASD individuals, but also clusters of increased FA in those with ASD [126]. This may have confounded and could be responsible for the negative findings in children with ADHD. Finally, comorbidities, such as affective and substance use disorders, are more common in adults with ADHD [5] and could also have confounded the results. Restricting recruitment to comorbidity-free individuals does not reflect daily clinical practice and limits our understanding of the biological basis of brain disorders. An alternative for future studies is that of taking a dimensional approach, in line with the National Institute of Mental Health Research Domain Criteria (RDoC) framework [5, 144]. In our meta-analysis, we considered the potential effect of sex and treatment exposure on the findings. However, the number of studies comparing ADHD presentations was limited, thus we could not meta-analyze them, and this should be subject of future investigations. Finally, we separately analyzed studies in children and adults, which allowed us to identify adult-specific alterations; however, the number of studies in the adult population was limited, thus findings should be interpreted with caution. Nevertheless, the association we observed between callosal FA and age, as well as findings from previous structural MRI studies, should encourage the longitudinal investigation of differences in the developmental trajectories of the white and gray matter in ADHD.
Conclusions
Clinicodemographic and methodological differences among studies are major barriers to our understanding of the neurobiology of ADHD. Future studies are needed to disentangle the potential biological differences related to sex, age, presentations, and comorbidities. They may also investigate tracts that have so far received less attention, such as the cerebellar peduncles, especially in adults. Finally, methodological improvements are recommended, especially optimizing imaging parameters, controlling and reporting for head motion, as well as efforts to enhance the comparability among studies, e.g., by using anatomical atlases to identify ROIs and tracts. While currently DWI studies do not have direct clinical implications in the field of ADHD [145], our findings may help future studies to stratify individuals with ADHD according to the underlying pathophysiology, which may guide the development of more tailored treatments. To this purpose, future study may also benefit from the combination of multiple imaging approaches, e.g., fMRI, functional Near Infrared Spectroscopy (fNIRS), and positron emission tomography (PET), as these can provide complementary information on functional and metabolic changes associated with ADHD.
References
American Psychiatric Association. DSM-5 Task Force. Diagnostic and statistical manual of mental disorders: DSM-5. 5th ed. Washington, D.C.: American Psychiatric Association; 2013. xliv, 947.
Faraone SV, Banaschewski T, Coghill D, Zheng Y, Biederman J, Bellgrove MA, et al. The World Federation of ADHD International Consensus Statement: 208 Evidence-based conclusions about the disorder. Neurosci Biobehav Rev. 2021;128:789–818.
Sayal K, Prasad V, Daley D, Ford T, Coghill D. ADHD in children and young people: prevalence, care pathways, and service provision. Lancet Psychiatry. 2018;5:175–86.
Faraone SV, Biederman J, Mick E. The age-dependent decline of attention deficit hyperactivity disorder: a meta-analysis of follow-up studies. Psychol Med. 2006;36:159–65.
Katzman MA, Bilkey TS, Chokka PR, Fallu A, Klassen LJ. Adult ADHD and comorbid disorders: clinical implications of a dimensional approach. BMC Psychiatry. 2017;17:302.
Gnanavel S, Sharma P, Kaushal P, Hussain S. Attention deficit hyperactivity disorder and comorbidity: a review of literature. World J Clin Cases. 2019;7:2420–6.
Gonzalez-Madruga K, Staginnus M, Fairchild G. Alterations in structural and functional connectivity in ADHD: implications for theories of ADHD. Curr Top Behav Neurosci. 2022;57:445–81.
Basser PJ, Mattiello J, LeBihan D. Estimation of the effective self-diffusion tensor from the NMR spin echo. J Magn Reson Ser B. 1994;103:247–54.
Basser PJ, Mattiello J, LeBihan D. MR diffusion tensor spectroscopy and imaging. Biophysical J. 1994;66:259–67.
Van Ewijk H, Heslenfeld DJ, Zwiers MP, Buitelaar JK, Oosterlaan J. Diffusion tensor imaging in attention deficit/hyperactivity disorder: a systematic review and meta-analysis. Neurosci Biobehav Rev. 2012;36:1093–106.
Aoki Y, Cortese S, Castellanos FX. Research review: diffusion tensor imaging studies of attention-deficit/hyperactivity disorder: meta-analyses and reflections on head motion. J Child Psychol Psychiatry. 2018;59:193–202.
Chen L, Hu X, Ouyang L, He N, Liao Y, Liu Q, et al. A systematic review and meta-analysis of tract-based spatial statistics studies regarding attention-deficit/hyperactivity disorder. Neurosci Biobehav Rev. 2016;68:838–47.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
Radua J, Mataix-Cols D, Phillips ML, El-Hage W, Kronhaus DM, Cardoner N, et al. A new meta-analytic method for neuroimaging studies that combines reported peak coordinates and statistical parametric maps. Eur Psychiatry. 2012;27:605–11.
Albajes-Eizagirre A, Solanes A, Vieta E, Radua J. Voxel-based meta-analysis via permutation of subject images (PSI): theory and implementation for SDM. Neuroimage. 2019;186:174–84.
Bouziane C, Filatova OG, Schrantee A, Caan MWA, Vos FM, Reneman L. White matter by diffusion MRI following methylphenidate treatment: a randomized control trial in males with attention-deficit/hyperactivity disorder. Radiology. 2019;293:186–92.
Simmonds DJ, Hallquist MN, Asato M, Luna B. Developmental stages and sex differences of white matter and behavioral development through adolescence: a longitudinal diffusion tensor imaging (DTI) study. Neuroimage. 2014;92:356–68.
Catani M dSM. Atlas of human brain connections. Oxford: Oxford University Press; 2012.
Ashtari M, Kumra S, Bhaskar SL, Clarke T, Thaden E, Cervellione KL, et al. Attention-deficit/hyperactivity disorder: a preliminary diffusion tensor imaging study. Biol Psychiatry. 2005;57:448–55.
Acer N, Dolu N, Zararsiz G, Dogan MS, Gumus K, Ozmen S, et al. Anatomical characterization of ADHD using an atlas-based analysis: a diffusion tensor imaging study. EuroBiotech J. 2017;1:46–56.
Adisetiyo V, Tabesh A, Di Martino A, Falangola MF, Castellanos FX, Jensen JH, et al. Attention-deficit/hyperactivity disorder without comorbidity is associated with distinct atypical patterns of cerebral microstructural development. Hum Brain Mapp. 2014;35:2148–62.
Beare R, Adamson C, Bellgrove MA, Vilgis V, Vance A, Seal ML, et al. Altered structural connectivity in ADHD: a network based analysis. Brain Imaging Behav. 2017;11:846–58.
Chiang H-L, Chen Y-J, Lo Y-C, Tseng W-YI, Gau SS-F. Altered white matter tract property related to impaired focused attention, sustained attention, cognitive impulsivity and vigilance in attention-deficit/ hyperactivity disorder. J Psychiatry Neurosci. 2015;40:325–35.
Chiang HL, Chen YJ, Shang CY, Tseng WYI, Gau SSF. Different neural substrates for executive functions in youths with ADHD: a diffusion spectrum imaging tractography study. Psychological Med. 2016;46:1225–38.
Chuang T-C, Wu M-T, Huang S-P, Weng M-J, Yang P. Diffusion tensor imaging study of white matter fiber tracts in adolescent attention-deficit/hyperactivity disorder. Psychiatry Res Neuroimaging. 2013;211:186–7.
Davenport ND, Karatekin C, White T, Lim KO. Differential fractional anisotropy abnormalities in adolescents with ADHD or schizophrenia. Psychiatry Res. 2010;181:193–8.
De Zeeuw P, Mandl RC, Hulshoff Pol HE, Van Engeland H, Durston S. Decreased frontostriatal microstructural organization in attention deficit/hyperactivity disorder. Hum Brain Mapp. 2012;33:1941–51.
Francx W, Llera A, Mennes M, Zwiers MP, Faraone SV, Oosterlaan J, et al. Integrated analysis of gray and white matter alterations in attention-deficit/hyperactivity disorder. NeuroImage Clin. 2016;11:357–67.
Gau SS, Tseng WL, Tseng WY, Wu YH, Lo YC. Association between microstructural integrity of frontostriatal tracts and school functioning: ADHD symptoms and executive function as mediators. Psychological Med. 2015;45:529–43.
Griffiths KR, Braund TA, Kohn MR, Clarke S, Williams LM, Korgaonkar MS. Structural brain network topology underpinning ADHD and response to methylphenidate treatment. Transl Psychiatry. 2021;11:150.
Hong SB, Zalesky A, Park S, Yang YH, Park MH, Kim B, et al. COMT genotype affects brain white matter pathways in attention-deficit/hyperactivity disorder. Hum Brain Mapp. 2015;36:367–77.
Kobel M, Bechtel N, Specht K, Klarhofer M, Weber P, Scheffler K, et al. Structural and functional imaging approaches in attention deficit/hyperactivity disorder: does the temporal lobe play a key role?. Psychiatry Res Neuroimaging. 2010;183:230–6.
Jacobson LA, Peterson DJ, Rosch KS, Crocetti D, Mori S, Mostofsky SH. Sex-based dissociation of white matter microstructure in children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2015;54:938–46.
Lei D, Ma J, Du X, Shen G, Jin X, Gong Q. Microstructural abnormalities in the combined and inattentive subtypes of attention deficit hyperactivity disorder: a diffusion tensor imaging study. Sci Rep. 2014;4:6875.
Li Q, Sun J, Guo L, Zang Y, Feng Z, Huang X, et al. Increased fractional anisotropy in white matter of the right frontal region in children with attention-deficit/hyperactivity disorder: a diffusion tensor imaging study. Neuroendocrinol Lett. 2010;31:747–53.
Lin HY, Gau SSF, Huang-Gu SL, Shang CY, Wu YH, Tseng WYI. Neural substrates of behavioral variability in attention deficit hyperactivity disorder: based on ex-Gaussian reaction time distribution and diffusion spectrum imaging tractography. Psychological Med. 2014;44:1751–64.
Nagel BJ, Bathula D, Herting M, Schmitt C, Kroenke CD, Fair D, et al. Altered white matter microstructure in children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2011;50:283–92.
Pavuluri MN, Yang S, Kamineni K, Passarotti AM, Srinivasan G, Harral EM, et al. Diffusion tensor imaging study of white matter fiber tracts in pediatric bipolar disorder and attention-deficit/hyperactivity disorder. Biol Psychiatry. 2009;65:586–93.
Peterson DJ, Ryan M, Rimrodt SL, Cutting LE, Denckla MB, Kaufmann WE, et al. Increased regional fractional anisotropy in highly screened attention-deficit hyperactivity disorder (ADHD). J Child Neurol. 2011;26:1296–302.
Qiu M-G, Ye Z, Li Q-Y, Liu G-J, Xie B, Wang J. Changes of brain structure and function in ADHD children. Brain Topogr. 2011;24:243–52.
Shang CY, Wu YH, Gau SS, Tseng WY. Disturbed microstructural integrity of the frontostriatal fiber pathways and executive dysfunction in children with attention deficit hyperactivity disorder. Psychological Med. 2013;43:1093–107.
Silk TJ, Vilgis V, Adamson C, Chen J, Smit L, Vance A, et al. Abnormal asymmetry in frontostriatal white matter in children with attention deficit hyperactivity disorder. Brain Imaging Behav. 2016;10:1080–9.
Tamm L, Barnea-Goraly N, Reiss AL. Diffusion tensor imaging reveals white matter abnormalities in Attention-Deficit/Hyperactivity Disorder. Psychiatry Res. 2012;202:150–4.
Unsel-Bolat G, Baytunca MB, Kardas B, Ipci M, Inci Izmir SB, Ozyurt O, et al. Diffusion tensor imaging findings in children with sluggish cognitive tempo comorbid Attention Deficit Hyperactivity Disorder. Nord J Psychiatry. 2020;74:620–6.
Wu Y-H, Gau SS-F, Lo Y-C, Tseng W-YI. White matter tract integrity of frontostriatal circuit in attention deficit hyperactivity disorder: association with attention performance and symptoms. Hum Brain Mapp. 2014;35:199–212.
Wu Z-M, Bralten J, Cao Q-J, Hoogman M, Zwiers MP, An L, et al. White matter microstructural alterations in children with ADHD: categorical and dimensional perspectives. Neuropsychopharmacology. 2017;42:572–80.
Zhan C, Liu Y, Wu K, Gao Y, Li X. Structural and functional abnormalities in children with attention-deficit/hyperactivity disorder: a focus on subgenual anterior cingulate cortex. Brain Connect. 2017;7:106–14.
Wu ZM, Wang P, Liu L, Liu J, Cao XL, Sun L, et al. ADHD-inattentive versus ADHD-Combined subtypes: a severity continuum or two distinct entities? A comprehensive analysis of clinical, cognitive and neuroimaging data. J Psychiatr Res. 2022;149:28–36.
Choi J, Lim MH, Lee C, Park JK, Son J, Shim SH, et al. Comparison of diffusion tensor imaging in attention deficit hyperactivity disorder boys with or without comorbid tic disorders. J Korean Neuropsychiatr Assoc. 2008;47:493–502.
Wu W, McAnulty G, Hamoda HM, Sarill K, Karmacharya S, Gagoski B, et al. Detecting microstructural white matter abnormalities of frontal pathways in children with ADHD using advanced diffusion models. Brain Imaging Behav. 2020;14:981–97.
Ercan ES, Suren S, Bacanli A, Yazici KU, Calli C, Ardic UA, et al. Altered structural connectivity is related to attention deficit/hyperactivity subtypes: A DTI study. Psychiatry Res: Neuroimaging. 2016;256:57–64.
Hong S-B, Zalesky A, Fornito A, Park S, Yang Y-H, Park M-H, et al. Connectomic disturbances in attention-deficit/hyperactivity disorder: a whole-brain tractography analysis. Biol Psychiatry. 2014;76:656–63.
Chaim TM, Zhang T, Zanetti MV, Da Silva MA, Louza MR, Doshi J, et al. Multimodal magnetic resonance imaging study of Treatment-Naive Adults with Attention-Deficit/Hyperactivity Disorder. PLoS One. 2014;9:e110199.
Gehricke JG, Kruggel F, Thampipop T, Alejo SD, Tatos E, Fallon J, et al. The brain anatomy of attention-deficit/hyperactivity disorder in young adults - a magnetic resonance imaging study. PLoS One. 2017;12:e0175433.
Onnink AM, Zwiers MP, Hoogman M, Mostert JC, Dammers J, Kan CC, et al. Deviant white matter structure in adults with attention-deficit/hyperactivity disorder points to aberrant myelination and affects neuropsychological performance. Prog Neuro Psychopharmacol Biol Psychiatry. 2015;63:14–22.
Sidlauskaite J, Caeyenberghs K, Sonuga-Barke E, Roeyers H, Wiersema JR. Whole-brain structural topology in adult attention-deficit/hyperactivity disorder: Preserved global - disturbed local network organization. NeuroImage Clin. 2015;9:506–12.
Tung YH, Lin HY, Chen CL, Shang CY, Yang LY, Hsu YC, et al. Whole brain white matter tract deviation and idiosyncrasy from normative development in autism and ADHD and unaffected siblings link with dimensions of psychopathology and cognition. Am J Psychiatry. 2021;178:730–743.
van Ewijk H, Heslenfeld DJ, Zwiers MP, Faraone SV, Luman M, Hartman CA, et al. Different mechanisms of white matter abnormalities in attention-deficit/hyperactivity disorder: a diffusion tensor imaging study. J Am Acad Child Adolesc Psychiatry. 2014;53:790–9.
Wang B, Wang GS, Wang X, Cao R, Xiang J, Yan T, et al. Rich-club analysis in adults with ADHD connectomes reveals an abnormal structural core network. J Atten Disord. 2021;25:1068–79.
Chiang HL, Yang LK, Chen YJ, Hsu YC, Lo YC, Tseng WYI, et al. Altered white-matter tract property in adults with attention-deficit hyperactivity disorder. Neuroscience. 2022;487:78–87.
Li D, Li T, Niu Y, Xiang J, Cao R, Liu B, et al. Reduced hemispheric asymmetry of brain anatomical networks in attention deficit hyperactivity disorder. Brain Imaging Behav. 2019;13:669–84.
Schweren LJ, Hartman CA, Zwiers MP, Heslenfeld DJ, Franke B, Oosterlaan J, et al. Stimulant treatment history predicts frontal-striatal structural connectivity in adolescents with attention-deficit/hyperactivity disorder. Eur Neuropsychopharmacol. 2016;26:674–83.
Luo YY, Halperin JM, Li XB. Anatomical substrates of symptom remission and persistence in young adults with childhood attention deficit/hyperactivity disorder. Eur Neuropsychopharmacol. 2020;33:117–25.
Çelik Z, Çolak Ç, Di Biase MA, Zalesky A, Zorlu N, Bora E, et al. Structural connectivity in adolescent synthetic cannabinoid users with and without ADHD. Brain Imaging Behav. 2020;14:505–14.
Lawrence KE, Levitt JG, Loo SK, Ly R, Yee V, O'Neill J, et al. White matter microstructure in subjects with attention-deficit/hyperactivity disorder and their siblings. J Am Acad Child Adolesc Psychiatry. 2013;52:431–40.
Svatkova A, Nestrasil I, Rudser K, Fine JG, Bledsoe J, Semrud-Clikeman M. Unique white matter microstructural patterns in ADHD presentations-A diffusion tensor imaging study. Hum Brain Mapp. 2016;37:3323–36.
Wu ZM, Llera A, Hoogman M, Cao QJ, Zwiers MP, Bralten J, et al. Linked anatomical and functional brain alterations in children with attention-deficit/hyperactivity disorder. Neuroimage Clin. 2019;23:101851.
Xia S, Li X, Kimball AE, Kelly MS, Lesser I, Branch C. Thalamic shape and connectivity abnormalities in children with attention-deficit/hyperactivity disorder. Psychiatry Res. 2012;204:161–7.
Zhou X, Lin Q, Gui Y, Wang Z, Liu M, Lu H. Multimodal MR images-based diagnosis of early adolescent attention-deficit/hyperactivity disorder using multiple kernel learning. Front Neurosci. 2021;15:710133.
Bouziane C, Caan MWA, Tamminga HGH, Schrantee A, Bottelier MA, de Ruiter MB, et al. ADHD and maturation of brain white matter: A DTI study in medication naive children and adults. Neuroimage Clin. 2018;17:53–9.
Konrad A, Dielentheis TF, El Masri D, Bayerl M, Fehr C, Gesierich T, et al. Disturbed structural connectivity is related to inattention and impulsivity in adult attention deficit hyperactivity disorder. Eur J Neurosci. 2010;31:912–9.
Kölle M, Mackert S, Heckel K, Philipsen A, Ulrich M, Grön G. Lower fractional anisotropy of the corticothalamic tract and increased response time variability in adult patients with ADHD. J Psychiatry Neurosci. 2022;47:E99–e108.
Li D, Cui X, Yan T, Liu B, Zhang H, Xiang J, et al. Abnormal rich club organization in hemispheric white matter networks of ADHD. J Atten Disord. 2021;25:1215–29.
Chaim-Avancini TM, Doshi J, Zanetti MV, Erus G, Silva MA, Duran FLS, et al. Neurobiological support to the diagnosis of ADHD in stimulant-naïve adults: pattern recognition analyses of MRI data. Acta Psychiatr Scand. 2017;136:623–36.
Ameis SH, Lerch JP, Taylor MJ, Lee W, Viviano JD, Pipitone J, et al. A diffusion tensor imaging study in children with ADHD, autism spectrum disorder, OCD, and matched controls: Distinct and non-distinct white matter disruption and dimensional brain-behavior relationships. Am J Psychiatry. 2016;173:1213–22.
Bu X, Yang C, Liang K, Lin Q, Lu L, Zhang L, et al. Quantitative tractography reveals changes in the corticospinal tract in drug-naïve children with attention-deficit/hyperactivity disorder. J Psychiatry Neurosci. 2020;45:134–41.
Chiang H-L, Hsu Y-C, Shang C-Y, Tseng W-YI, Gau SS-F. White matter endophenotype candidates for ADHD: a diffusion imaging tractography study with sibling design. Psychological Med. 2020;50:1203–13.
Douglas PK, Gutman B, Anderson A, Larios C, Lawrence KE, Narr K, et al. Hemispheric brain asymmetry differences in youths with attention-deficit/hyperactivity disorder. Neuroimage Clin. 2018;18:744–52.
Fuelscher I, Hyde C, Anderson V, Silk TJ. White matter tract signatures of fiber density and morphology in ADHD. Cortex. 2021;138:329–40.
Hamilton LS, Levitt JG, O'Neill J, Alger JR, Luders E, Phillips OR, et al. Reduced white matter integrity in attention-deficit hyperactivity disorder. NeuroReport. 2008;19:1705–8.
Hyde C, Fuelscher I, Sciberras E, Efron D, Anderson VA, Silk T. Understanding motor difficulties in children with ADHD: A fixel-based analysis of the corticospinal tract. Prog Neuropsychopharmacol Biol Psychiatry. 2021;105:110125.
Graziano PA, Garic D, Dick AS. Individual differences in white matter of the uncinate fasciculus and inferior fronto-occipital fasciculus: possible early biomarkers for callous-unemotional behaviors in young children with disruptive behavior problems. J Child Psychol Psychiatry. 2022;63:19–33.
Bechtel N, Kobel M, Penner I-K, Klarhofer M, Scheffler K, Opwis K, et al. Decreased fractional anisotropy in the middle cerebellar peduncle in children with epilepsy and/or attention deficit/hyperactivity disorder: a preliminary study. Epilepsy Behav. 2009;15:294–8.
Pastura G, Doering T, Gasparetto EL, Mattos P, Araújo AP. Exploratory analysis of diffusion tensor imaging in children with attention deficit hyperactivity disorder: evidence of abnormal white matter structure. Atten Defic Hyperact Disord. 2016;8:65–71.
Cao Q, Sun L, Gong G, Lv Y, Cao X, Shuai L, et al. The macrostructural and microstructural abnormalities of corpus callosum in children with attention deficit/hyperactivity disorder: a combined morphometric and diffusion tensor MRI study. Brain Res. 2010;1310:172–80.
Lin Q, Bu X, Chen H, Liang Y, Wang W, Yi Y, et al. Sex differences in microstructural alterations in the corpus callosum tracts in drug-naïve children with ADHD. Brain Imaging Behav. 2022;16:1592–604.
de Luis-Garcia R, Cabus-Pinol G, Imaz-Roncero C, Argibay-Quinones D, Barrio-Arranz G, Aja-Fernandez S, et al. Attention deficit/hyperactivity disorder and medication with stimulants in young children: a DTI study. Prog Neuro Psychopharmacol Biol Psychiatry. 2015;57:176–84.
King JB, Yurgelun-Todd D, Stoeckel A, Di Muzio JM, Lopez-Larson MP. Sex differences in white matter integrity in youths with attention-deficit/hyperactivity disorder: A pilot study. Front Neurosci. 2015;9:232.
Langevin LM, Macmaster FP, Crawford S, Lebel C, Dewey D. Common white matter microstructure alterations in pediatric motor and attention disorders. J Pediatrics. 2014;164:1157–64.e1.
Bode MK, Lindholm P, Kiviniemi V, Moilanen I, Ebeling H, Veijola J, et al. DTI abnormalities in adults with past history of attention deficit hyperactivity disorder: a tract-based spatial statistics study. Acta Radiol. 2015;56:990–6.
Ohta H, Aoki YY, Itahashi T, Kanai C, Fujino J, Nakamura M, et al. White matter alterations in autism spectrum disorder and attention-deficit/hyperactivity disorder in relation to sensory profile. Mol Autism. 2020;11:13.
Dramsdahl M, Westerhausen R, Haavik J, Hugdahl K, Plessen KJ. Adults with attention-deficit/hyperactivity disorder - a diffusion-tensor imaging study of the corpus callosum. Psychiatry Res. 2012;201:168–73.
Francx W, Zwiers MP, Mennes M, Oosterlaan J, Heslenfeld D, Hoekstra PJ, et al. White matter microstructure and developmental improvement of hyperactive/impulsive symptoms in attention-deficit/hyperactivity disorder. J Child Psychol Psychiatry. 2015;56:1289–97.
Silk TJ, Vance A, Rinehart N, Bradshaw JL, Cunnington R. White-matter abnormalities in attention deficit hyperactivity disorder: a diffusion tensor imaging study. Hum Brain Mapp. 2009;30:2757–65.
Cortese S, Imperati D, Zhou J, Proal E, Klein RG, Mannuzza S, et al. White matter alterations at 33-year follow-up in adults with childhood attention-deficit/hyperactivity disorder. Biol Psychiatry. 2013;74:591–8.
Makris N, Buka SL, Biederman J, Papadimitriou GM, Hodge SM, Valera EM, et al. Attention and executive systems abnormalities in adults with childhood adhd: A DT-MRI study of connections. Cereb Cortex. 2008;18:1210–20.
Shaw P, Sudre G, Wharton A, Weingart D, Sharp W, Sarlls J. White matter microstructure and the variable adult outcome of childhood attention deficit hyperactivity disorder. Neuropsychopharmacology. 2015;40:746–54.
Chen L, Huang X, Lei D, He N, Hu X, Chen Y, et al. Microstructural abnormalities of the brain white matter in attention-deficit/hyperactivity disorder. J Psychiatry Neurosci. 2015;40:280–7.
Basay BK, Buber A, Basay O, Alacam H, Ozturk O, Suren S, et al. White matter alterations related to attention-deficit hyperactivity disorder and COMT val158met polymorphism: Children with valine homozygote attention-deficit hyperactivity disorder have altered white matter connectivity in the right cingulum (cingulate gyrus). Neuropsychiatr Dis Treat. 2016;12:969–81.
Wang L, Fan GG, Yu B, Ma HW, Pan XX, Guo QY. Correlated study between reaction time and fractional anisotropy for ADHD children. J Med Imaging Technol. 2008;24:489–92.
Stephens K, Silk TJ, Anderson V, Hazell P, Enticott PG, Sciberras E. Associations between limbic system white matter structure and socio-emotional functioning in children with ADHD + ASD. J Autism Dev Disord. 2021;51:2663–72.
Versace A, Jones NP, Joseph HM, Lindstrom RA, Wilson TK, Lima Santos JP, et al. White matter abnormalities associated with ADHD outcomes in adulthood. Mol Psychiatry. 2021;26:6655–65.
Lee S, Jeong B, Go HJ, Yang PS, Kwon MJ, Choi J. Occipito-tempo- ral connectivity in medication-naïve ADHD children: preliminary diffusion tensor imaging study. J Korean Neuropsychiatr Assoc. 2009;48:344–51.
Konrad A, Dielentheis TF, El Masri D, Dellani PR, Stoeter P, Vucurevic G, et al. White matter abnormalities and their impact on attentional performance in adult attention-deficit/hyperactivity disorder. Eur Arch Psychiatry Clin Neurosci. 2012;262:351–60.
Hofer S, Frahm J. Topography of the human corpus callosum revisited-comprehensive fiber tractography using diffusion tensor magnetic resonance imaging. Neuroimage. 2006;32:989–94.
Blaauw J, Meiners LC. The splenium of the corpus callosum: embryology, anatomy, function and imaging with pathophysiological hypothesis. Neuroradiology. 2020;62:563–85.
Yeo BT, Krienen FM, Sepulcre J, Sabuncu MR, Lashkari D, Hollinshead M, et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol. 2011;106:1125–65.
Sanefuji M, Craig M, Parlatini V, Mehta MA, Murphy DG, Catani M, et al. Double-dissociation between the mechanism leading to impulsivity and inattention in Attention Deficit Hyperactivity Disorder: a resting-state functional connectivity study. Cortex. 2017;86:290–302.
Ramos AA, Hamdan AC, Machado L. A meta-analysis on verbal working memory in children and adolescents with ADHD. Clin Neuropsychol. 2020;34:873–98.
Pievsky MA, McGrath RE. The neurocognitive profile of attention-deficit/hyperactivity disorder: a review of meta-analyses. Arch Clin Neuropsychol. 2018;33:143–57.
Valera EM, Faraone SV, Murray KE, Seidman LJ. Meta-analysis of structural imaging findings in attention-deficit/hyperactivity disorder. Biol Psychiatry. 2007;61:1361–9.
Hutchinson AD, Mathias JL, Banich MT. Corpus callosum morphology in children and adolescents with attention deficit hyperactivity disorder: a meta-analytic review. Neuropsychology. 2008;22:341–9.
Hoogman M, Bralten J, Hibar DP, Mennes M, Zwiers MP, Schweren LSJ, et al. Subcortical brain volume differences in participants with attention deficit hyperactivity disorder in children and adults: a cross-sectional mega-analysis. Lancet Psychiatry. 2017;4:310–9.
Hoogman M, Muetzel R, Guimaraes JP, Shumskaya E, Mennes M, Zwiers MP, et al. Brain imaging of the cortex in ADHD: a coordinated analysis of large-scale clinical and population-based samples. Am J Psychiatry. 2019;176:531–42.
Norman LJ, Carlisi C, Lukito S, Hart H, Mataix-Cols D, Radua J, et al. Structural and functional brain abnormalities in attention-deficit/hyperactivity disorder and obsessive-compulsive disorder: a comparative meta-analysis. JAMA Psychiatry. 2016;73:815–25.
Lukito S, Norman L, Carlisi C, Radua J, Hart H, Simonoff E, et al. Comparative meta-analyses of brain structural and functional abnormalities during cognitive control in attention-deficit/hyperactivity disorder and autism spectrum disorder. Psychol Med. 2020;50:894–919.
Hart H, Radua J, Nakao T, Mataix-Cols D, Rubia K. Meta-analysis of functional magnetic resonance imaging studies of inhibition and attention in attention-deficit/hyperactivity disorder: exploring task-specific, stimulant medication, and age effects. JAMA Psychiatry. 2013;70:185–98.
Hart H, Radua J, Mataix-Cols D, Rubia K. Meta-analysis of fMRI studies of timing in attention-deficit hyperactivity disorder (ADHD). Neurosci Biobehav Rev. 2012;36:2248–56.
Laird AR, Eickhoff SB, Li K, Robin DA, Glahn DC, Fox PT. Investigating the functional heterogeneity of the default mode network using coordinate-based meta-analytic modeling. J Neurosci. 2009;29:14496–505.
Bozhilova NS, Michelini G, Kuntsi J, Asherson P. Mind wandering perspective on attention-deficit/hyperactivity disorder. Neurosci Biobehav Rev. 2018;92:464–76.
Sripada C, Kessler D, Fang Y, Welsh RC, Prem Kumar K, Angstadt M. Disrupted network architecture of the resting brain in attention-deficit/hyperactivity disorder. Hum Brain Mapp. 2014;35:4693–705.
Sonuga-Barke EJ, Castellanos FX. Spontaneous attentional fluctuations in impaired states and pathological conditions: a neurobiological hypothesis. Neurosci Biobehav Rev. 2007;31:977–86.
Castellanos FX, Margulies DS, Kelly C, Uddin LQ, Ghaffari M, Kirsch A, et al. Cingulate-precuneus interactions: a new locus of dysfunction in adult attention-deficit/hyperactivity disorder. Biol Psychiatry. 2008;63:332–7.
Sutcubasi B, Metin B, Kurban MK, Metin ZE, Beser B, Sonuga-Barke E. Resting-state network dysconnectivity in ADHD: A system-neuroscience-based meta-analysis. World J Biol Psychiatry. 2020;21:662–72.
Sharp DJ, Beckmann CF, Greenwood R, Kinnunen KM, Bonnelle V, De Boissezon X, et al. Default mode network functional and structural connectivity after traumatic brain injury. Brain. 2011;134:2233–47.
Zhao Y, Yang L, Gong G, Cao Q, Liu J. Identify aberrant white matter microstructure in ASD, ADHD and other neurodevelopmental disorders: a meta-analysis of diffusion tensor imaging studies. Prog Neuropsychopharmacol Biol Psychiatry. 2022;113:110477.
Jones DK, Knosche TR, Turner R. White matter integrity, fiber count, and other fallacies: the do’s and don’ts of diffusion MRI. Neuroimage. 2013;73:239–54.
Beaulieu C. The basis of anisotropic water diffusion in the nervous system - a technical review. NMR Biomed. 2002;15:435–55.
Lesch KP. Editorial: can dysregulated myelination be linked to ADHD pathogenesis and persistence? J Child Psychol Psychiatry. 2019;60:229–31.
Demontis D, Walters RK, Martin J, Mattheisen M, Als TD, Agerbo E, et al. Discovery of the first genome-wide significant risk loci for attention deficit/hyperactivity disorder. Nat Genet. 2019;51:63–75.
Walton E, Pingault JB, Cecil CA, Gaunt TR, Relton CL, Mill J, et al. Epigenetic profiling of ADHD symptoms trajectories: a prospective, methylome-wide study. Mol Psychiatry. 2017;22:250–6.
Bethlehem RAI, Seidlitz J, White SR, Vogel JW, Anderson KM, Adamson C, et al. Brain charts for the human lifespan. Nature. 2022;604:525–33.
Lebel C, Caverhill-Godkewitsch S, Beaulieu C. Age-related regional variations of the corpus callosum identified by diffusion tensor tractography. Neuroimage. 2010;52:20–31.
Lebel C, Beaulieu C. Longitudinal development of human brain wiring continues from childhood into adulthood. J Neurosci. 2011;31:10937–47.
Shaw P, Eckstrand K, Sharp W, Blumenthal J, Lerch JP, Greenstein D, et al. Attention-deficit/hyperactivity disorder is characterized by a delay in cortical maturation. Proc Natl Acad Sci USA. 2007;104:19649–54.
Kamiya K, Hori M, Aoki S. NODDI in clinical research. J Neurosci Methods. 2020;346:108908.
Auerbach EJ, Xu J, Yacoub E, Moeller S, Ugurbil K. Multiband accelerated spin-echo echo planar imaging with reduced peak RF power using time-shifted RF pulses. Magn Reson Med. 2013;69:1261–7.
Tax CMW, Bastiani M, Veraart J, Garyfallidis E, Okan Irfanoglu M. What’s new and what’s next in diffusion MRI preprocessing. Neuroimage. 2022;249:118830.
Cortese S, Aoki YY, Itahashi T, Castellanos FX, Eickhoff SB. Systematic review and meta-analysis: resting-state functional magnetic resonance imaging studies of attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2021;60:61–75.
Samea F, Soluki S, Nejati V, Zarei M, Cortese S, Eickhoff SB, et al. Brain alterations in children/adolescents with ADHD revisited: a neuroimaging meta-analysis of 96 structural and functional studies. Neurosci Biobehav Rev. 2019;100:1–8.
Li T, van Rooij D, Roth Mota N, Buitelaar JK, Group EAW, Hoogman M, et al. Characterizing neuroanatomic heterogeneity in people with and without ADHD based on subcortical brain volumes. J Child Psychol Psychiatry. 2021;62:1140–9.
Hollingdale J, Woodhouse E, Young S, Fridman A, Mandy W. Autistic spectrum disorder symptoms in children and adolescents with attention-deficit/hyperactivity disorder: a meta-analytical review. Psychol Med. 2020;50:2240–53.
Di X, Azeez A, Li X, Haque E, Biswal BB. Disrupted focal white matter integrity in autism spectrum disorder: a voxel-based meta-analysis of diffusion tensor imaging studies. Prog Neuropsychopharmacol Biol Psychiatry. 2018;82:242–8.
Heidbreder R. ADHD symptomatology is best conceptualized as a spectrum: a dimensional versus unitary approach to diagnosis. Atten Defic Hyperact Disord. 2015;7:249–69.
Castellanos FX. A biased perspective on brain imaging of ADHD. Am J Psychiatry. 2021;178:694–700.
Bookstein FL. "Voxel-based morphometry" should not be used with imperfectly registered images. NeuroImage. 2001;14:1454–62.
Sage CA, Van Hecke W, Peeters R, Sijbers J, Robberecht W, Parizel P, et al. Quantitative diffusion tensor imaging in amyotrophic lateral sclerosis: revisited. Hum brain Mapp. 2009;30:3657–75.
Zhang H, Avants BB, Yushkevich PA, Woo JH, Wang S, McCluskey LF, et al. High-dimensional spatial normalization of diffusion tensor images improves the detection of white matter differences: an example study using amyotrophic lateral sclerosis. IEEE Trans Med imaging. 2007;26:1585–97.
Catani M. Diffusion tensor magnetic resonance imaging tractography in cognitive disorders. Curr Opin Neurol. 2006;19:599–606.
Gentili C, Messerotti Benvenuti S, Lettieri G, Costa C, Cecchetti L. ROI and phobias: the effect of ROI approach on an ALE meta-analysis of specific phobias. Hum Brain Mapp. 2019;40:1814–28.
Muller VI, Cieslik EC, Laird AR, Fox PT, Radua J, Mataix-Cols D, et al. Ten simple rules for neuroimaging meta-analysis. Neurosci Biobehav Rev. 2018;84:151–61.
Smith SM, Jenkinson M, Johansen-Berg H, Rueckert D, Nichols TE, Mackay CE, et al. Tract-based spatial statistics: voxelwise analysis of multi-subject diffusion data. NeuroImage. 2006;31:1487–505.
Dell’Acqua F, Simmons A, Williams SC, Catani M. Can spherical deconvolution provide more information than fiber orientations? Hindrance modulated orientational anisotropy, a true-tract specific index to characterize white matter diffusion. Hum Brain Mapp. 2013;34:2464–83.
Alexander AL, Lee JE, Lazar M, Field AS. Diffusion tensor imaging of the brain. Neurotherapeutics. 2007;4:316–29.
Basser PJ, Pierpaoli C. Microstructural and physiological features of tissues elucidated by quantitative-diffusion-tensor MRI. J Magn Reson Ser B 1996;111:209–19.
Catani M, Dell’Acqua F. Mapping white matter pathways with diffusion imaging tractography: focus on neurosurgical applications. In: Duffau H, editor. Brain mapping: from neural basis of cognition to surgical applications. Vienna: Springer Vienna; 2011. p. 61–75.
Lebel C, Deoni S. The development of brain white matter microstructure. Neuroimage. 2018;182:207–18.
Forkel SJ, Friedrich P, Thiebaut de Schotten M, Howells H. White matter variability, cognition, and disorders: a systematic review. Brain Struct Funct. 2022;227:529–44.
Acknowledgements
We thank the following authors for providing us with additional information on the included studies: Dr Ball, Prof Berlim de Mello, Dr Doring, Prof Stevens, Dr Lee, Prof Li, Dr Mazzetti, Dr Pastura, Dr Silk, Dr Wolfers, Dr Wu, and Dr Zhou.
Funding
KR and SC were supported by the Efficacy and Mechanism Evaluation (EME) programme, an MRC and NIHR partnership (project ref: NIHR130077) and by the NIHR Programme Grant (NIHR203684), and SC by NIHR203035, NIHR128472, and RP-PG-0618-20003. KR and DGM were supported by the National Institute for Health Research (NIHR) and the UK Department of Health via the National Institute for Health Research (NIHR) Biomedical Research Centre (BRC) for Mental Health at South London and the Maudsley NHS Foundation Trust and Institute of Psychiatry, Psychology and Neuroscience, King’s College London. DGM was funded by two IMI initiatives (EU AIMS and AIMS-2-TRIALS) under grant agreements no.115300 and no. 777394. The design, management, analysis and reporting of the study are independent from the funders.
Author information
Authors and Affiliations
Contributions
VP: Conceptualization, Data curation, Investigation, Methodology, Project administration, Validation, Visualization, Writing - first draft and Writing - review & editing. TI: Data curation, Formal analysis, Investigation, Methodology, Validation, Visualization, Writing - first draft and Writing - review & editing. YL: Data curation, Investigation, and Writing - review & editing. SL: Methodology, Validation and Writing - first draft. TTN: Methodology, Validation and Writing - review & editing. YYA: Validation and Writing - review & editing. SJF: Investigation and Writing - review & editing. MC: Conceptualization, Writing - first draft and Writing - review & editing. KR: Conceptualization, Writing – first draft and Writing - review & editing. JHZ: Methodology, Validation, and Writing - review & editing. DGM: Resources, Supervision and Writing - review & editing. SM: Conceptualization, Methodology, Supervision, and Writing - review & editing.
Corresponding author
Ethics declarations
Competing interests
SC declares honoraria and reimbursement for travel and accommodation expenses for lectures from the following non-profit associations: Association for Child and Adolescent Central Health (ACAMH), Canadian ADHD Alliance Resource (CADDRA), and British Association of Pharmacology (BAP), for educational activity on ADHD. KR has received a grant from TAKEDA pharmaceuticals for a project not related to the current work and honoraria for consulting from SUPERNUS. The other authors have no conflicts of interest to declare.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Parlatini, V., Itahashi, T., Lee, Y. et al. White matter alterations in Attention-Deficit/Hyperactivity Disorder (ADHD): a systematic review of 129 diffusion imaging studies with meta-analysis. Mol Psychiatry 28, 4098–4123 (2023). https://doi.org/10.1038/s41380-023-02173-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-023-02173-1
This article is cited by
-
Attention-deficit/hyperactivity disorder
Nature Reviews Disease Primers (2024)
-
Cortical alterations associated with lower response to methylphenidate in adults with ADHD
Nature Mental Health (2024)
-
Poor response to methylphenidate is associated with a smaller dorsal attentive network in adult Attention-Deficit/Hyperactivity Disorder (ADHD)
Translational Psychiatry (2023)