Abstract
Depressive disorders are the most burdensome psychiatric disorders worldwide. Although huge efforts have been made to advance treatment, outcomes remain unsatisfactory. Many factors contribute to this gridlock including suboptimal animal models. Especially limited study comparability and replicability due to imprecise terminology concerning depressive-like states are major problems. To overcome these issues, new approaches are needed. Here, we introduce a taxonomical concept for modelling depression in laboratory mice, which we call depression-like syndrome (DLS). It hinges on growing evidence suggesting that mice possess advanced socioemotional abilities and can display non-random symptom patterns indicative of an evolutionary conserved disorder-like phenotype. The DLS approach uses a combined heuristic method based on clinical depression criteria and the Research Domain Criteria to provide a biobehavioural reference syndrome for preclinical rodent models of depression. The DLS criteria are based on available, species-specific evidence and are as follows: (I) minimum duration of phenotype, (II) significant sociofunctional impairment, (III) core biological features, (IV) necessary depressive-like symptoms. To assess DLS presence and severity, we have designed an algorithm to ensure statistical and biological relevance of findings. The algorithm uses a minimum combined threshold for statistical significance and effect size (p value ≤ 0.05 plus moderate effect size) for each DLS criterion. Taken together, the DLS is a novel, biologically founded, and species-specific minimum threshold approach. Its long-term objective is to gradually develop into an inter-model validation standard and microframework to improve phenotyping methodology in translational research.
Similar content being viewed by others
Introduction
Depressive disorders are the second most common, yet most burdensome psychiatric disorders worldwide with regards to years lived with disability [1]. They are responsible for huge socioeconomic and personal costs and associated with increased morbidity and mortality [2, 3]. Trend analyses indicate an increasing prevalence, highlighting its growing global public health burden [4]. While our understanding of biological, clinical, and imaging features and biomarkers is mounting [5], and therapies have improved [6, 7], clinical outcomes remain heterogenous and vary depending on the individual and therapy modality [8, 9]. In fact, only 50–70% of patients recover within one year of diagnosis, while up to 40% experience multiple depressive episodes and 15% suffer a chronic course of disease [10,11,12,13].
A recent meta-analysis of 522 double-blind, randomized placebo-controlled trials confirmed the efficacy of 21 common antidepressants and their superiority over placebo [14]. Nevertheless, beside the serendipitous discovery of ketamine and brexanalone, the last few decades of research have not provided any novel and effective antidepressants [15,16,17]. Many factors contribute to this gridlock, including suboptimal animal models, poor clinical trial design and outdated drug approval processes [17,18,19]. Ever since the first animal models of mental disorders emerged, they have triggered considerable debate about their value and validity [20]. The frequent lack of reproducibility and context-dependency of data derived from animal experiments, especially in preclinical neuropsychiatric research, are issues that remain unsolved [21,22,23,24]. Despite the shortcomings and criticism, the importance of animal models in the fields of neurobiology and psychiatry cannot be denied [16, 20]. However, to move past current limitations and potentiate the impact of animal model-derived data on the psychiatric diagnostic and therapeutic progress, methodological improvements are urgently needed [15]. The obvious discrepancy between an increasing public health need, limited therapeutic dependability and suboptimal preclinical modelling demands critical evaluation of the way depression is conceptualized in clinical, translational, and basic neuropsychiatric research. Foremost, a shared and reliable framework for clinical and preclinical data is necessary. Advances should include innovative, replicable in vivo models and employ a precise and universal terminology concerning depressive-like states in animals [7, 15, 16, 25]. Here, we propose a methodological approach to address this issue for preclinical mouse models in translational neuropsychiatry.
First, we outline the depression criteria of the Diagnostic and Statistical Manual of Mental Disorders 5th version (DSM-5) and in the International Classification of Diseases 11th revision (ICD-11), discuss conceptual pros and cons and present emerging alternative taxonomical approaches. Second, we critically analyze the current use of the term ‘depressive-like’ in contemporary translational research in neuropsychiatry. Third, we propose a novel taxonomical approach for depression and its modelling, which we denominate as depression-like syndrome (DLS) in laboratory mice (Mus musculus). Here, we focus on mice due of their dominant role in translational psychiatric research and behavioural neuroscience, even though the approach per se is also applicable in other rodent species like rats. DLS is a threshold-based syndrome definition, hinging on a combinatory DSM-5/ICD-11 and Research Domain Criteria (RDoC) approach. We examine the potential of DLS as a novel consensus in the field of neuropsychiatry, capable of increasing comparability between models as well as overall face and construct validity. Finally, we devise a blueprint for DLS assessment and describe how it can be integrated into the existing scientific framework.
Depression in the DSM-5 and ICD-11
The nosological entity labelled depression has been around since ancient times [26, 27]. Contemporary clinical taxonomy of mental disorders, as seen in the DSM-5 and ICD-11, primarily follows a symptom-based approach (Table 1). The classifications define psychiatric entities by appearance of select, yet only partially disease-specific phenomenological criteria (i.e., symptoms) over a certain period. These definitions often incorporate exclusion stipulations to allow discrimination between entities with a shared symptomatology. Conceptually, the classifications are similar, yet depression criteria differ slightly [28,29,30].
The DSM-5 defines depression as daily presence of five out of nine possible symptoms for a minimum of two weeks, for nearly all the time. One symptom must be depressed mood or strongly diminished interest/pleasure in daily activities. Disease severity is determined by overall symptom number, individual distress and social as well as occupational functional impairment. In addition, the symptoms should not be better explained by physiological effects of a substance or another medical condition [29, 31,32,33]. To assess functional impairment, the Global Assessment of Functioning Scale (GAF) has been developed based on the DSM criteria. It is a severity estimate score of psychiatric conditions ranging from 0 to 100, which accounts for overall symptom severity and functional impairment [34, 35].
The more recently updated ICD-11, on the other hand, defines a depressive episode by daily occurrence of five out of ten symptoms for a minimum of two weeks. The symptoms are similar to the DSM-5, except for the additional symptom hopelessness, which was included because of its reliability to discriminate between depressed and healthy subjects [28, 36]. As with the DSM-5, one symptom must be depressed mood or strongly diminished interest or pleasure in daily activities. Analogous to the DSM-5 exclusion stipulation, the symptoms should not be better explained by the effects of a substance, other medical condition, or bereavement. Disease severity is based on symptom intensity and overall functional impairment. In comparison to earlier ICD editions, the latest edition provides better representation of symptom heterogeneity, global and clinical applicability as well as an improved discrimination of diseases with overlapping symptomatology [28, 32, 37,38,39].
Overall, the ICD-11 depression criteria and severity estimates are closely related to the DSM-5 [28, 29]. The revision has aided the alignment of the two major classification systems and is therefore meeting the call for conceptual consistency in mental health taxonomy [40]. Due to this similarity, the ICD-11 and DSM-5 share most of their strengths and weaknesses in the context of depression [16, 28, 41, 42].
Novel alternatives to established clinical taxonomies
Although expert-consensus classifications like the DSM-5 and ICD-11 are an integral part of current clinical practice, they have triggered considerable debate [40, 43,44,45,46]. For the DSM-5, some of the more disputed features are its weak scientific foundation, arbitrary disease definitions, categorical disease approach and the associated loss of clinical data, insufficient representation of within-diagnosis heterogeneity, limited reliability and validity, negligence of frequent comorbidities, failure in establishing diagnosis-selective treatments, undependable severity assessments and predilection for false positive diagnosis [31, 40, 41, 47,48,49,50]. In contemporary clinical and scientific practice, the boundary between mental health and disease is arbitrary. Categorical distinctions are based on vague symptom clusters, which were established based on historical, and now debatable, empirical pillars [40, 41, 43, 46,47,48].
Given the limitations of the DSM-5 and ICD-11 [51], novel taxonomical approaches attempt to reduce arbitrariness and uncertainty in neuropsychiatry. For example, the ongoing BeCOME study uses deep phenotyping and a multi-omics approach to establish a biologically grounded classification of mental disorders [52]. Meanwhile, the Hierarchical Taxonomy of Psychopathology (HiTOP) follows an empirical bottom-up approach and defines psychiatric entities according to covariation of psychopathological symptoms, quantitively and based on available evidence. It proverbially aims to overhaul the DSM-5 to improve reliability, validity, and clinical utility [47, 48, 53]. Even though the HiTOP and BeCOME study are just two examples, they epitomize the field’s recent efforts to advance clinical taxonomy towards a more reliable and valid approach, both on a phenomenological and biological level.
Depressive-like behaviour and the Research Domain Criteria
Animal models of depressive disorders are mostly based on the induction of biological or behavioural changes seen in depressed patients using a psychosocial or biophysical stressor. Traditionally, rodent models have been judged based on face, construct, and predictive validity [17, 54]. Behavioural measures in laboratory mice include, but are not limited to, anhedonia, apathy, anxiety, despair and hopelessness, irritability, social aversion, cognitive impairment and disturbance of feeding, sleep, and psychomotor activity. These behaviours, either one alone or in an undefined combination, are commonly considered depressive-like phenotypes [7, 17, 55, 56]. The rationale behind this is that non-human animals including mice are commonly considered unable to experience depression in its phenomenological and biological entirety, yet they display behaviours similar to depressive symptomatology in humans. These behaviours are induced by causative factors (e.g., environmental, genetic), can be quantified by standardized behavioural tests and should respond to established antidepressants. Standard assessments include the forced swim test, tail suspension test and sucrose preference test. The latter is deemed the equivalent of anhedonia and is thus considered at the core of the depressive-like spectrum in laboratory mice [7, 15,16,17, 20, 25, 57, 58]. However, in contrast to the clinical setting, there is no consensus or universal standard regarding the composition and duration of the observable behavioural phenomena to be termed depressive-like in laboratory mice. Rather, any behaviour that is part of the depressive symptom spectrum is called depressive-like in mouse models, mostly independent from the induction scheme, its severity, composition, or duration [7, 16, 17, 55, 59,60,61,62].
The hypothesis that depressive behaviours are somewhat comparable between species, here humans and mice, rests on the shared phylogenetic origin and the subsequent interspecies conservation of biological and neuroanatomical structures necessary for complex emotions, cognition and behaviours [57]. The latter must be considered the conditio sine quam non for psychiatric disorders. In line with this assumption, the National Institute of Mental Health has developed the RDoC. It is a comprehensive framework for psychobiological research comprised out of five domains, which represent categories of interspecies homologies on a phenomenological, construct and network level. These domains are positive and negative valence, arousal and regulation, social processes, and cognition [42, 57, 63, 64]. Recently, the sensorimotor systems domain was added [65, 66]. Overall, the RDoC matrix is an elaborate and multidimensional tool, which enables an empirically substantiated default matrix of comparison and two-way translation between humans and laboratory rodents [42, 57, 67]. The RDoC enterprise is a work in progress and has been termed a “calculated gamble” by Lilienfeld [44]. It has been criticized for being reductionistic and inadequate in important clinical disease features [44, 45, 67, 68]. Furthermore, since the RDoC is neither a clinical classification nor established beyond reasonable doubt, it still needs to prove its scientific and societal value [42, 67, 69,70,71].
Combining the symptom-based approach of DSM/ICD with the evolutionary conception of the RDoC yields great potential in the field of translational neuropsychiatry. Merging the two concepts may result in a revolutionary, integrative, and accurate classification which could balance the inherent advantages and drawbacks of the different approaches [67, 72, 73]. This is critically important since precise and integrative rodent models of depression are still lacking [15, 16] and comparability and reproducibility, especially of behavioural paradigms carried out in laboratory rodents, is in great need of improvement [21,22,23,24].
To refine mouse models of depression with a clinical research aim, we suggest revisiting the use of the established yet undefined term ‘depressive-like’ considering the integrative and combinatory heuristic approach. Simultaneously, we advocate an ongoing dialogue between clinicians and neuroscientists to advance back-forward-translation, scientific discovery, and eventually improve therapies [7]. Here, we argue that a combined DSM/ICD/RDoC-definition of ‘depressive-like’ behaviour in the form of a murine DLS could help produce replicable and comparable animal studies, improving overall validity and clinical impact.
Arguments for a standardized depression-like syndrome in laboratory mice
To advance towards a combination of the phenomenological DSM/ICD and the neurobiological RDoC in translational depression modelling, we argue that a novel approach needs to merge core depression symptoms, interspecies commonalities and clinical as well as biological heterogeneity, framed by evolutionary conserved, species-selective considerations.
Based on the syndromic nature of depression entrenched in the DSM/ICD, the behavioural and biological interspecies homologies and evolutionary assumptions of the RDoC [42, 57, 63, 73], and the cogent evidence from human and animal studies unveiling the behavioural and biological mechanisms of chronic stress and depression, it can be reasoned that certain features of depression neurobiology and symptomatology are conserved and thus shared between related species including humans and mice, while others are not [6, 16, 17, 74,75,76,77,78]. For example, the DSM/ICD criterion of inappropriate guilt entails two presuppositions: the concept of inappropriateness and of guilt. Both are uniquely human and thus species-specific concepts. Guilt behaviour in dogs could not be proven [79,80,81,82] and no tests of guilt have been advocated or used in rodents in the context of depressive-like behaviour. The same line of argument stands for suicidal ideation and suicide attempts [83]. For most of the other DSM-5 depression criteria, Czéh et al. have provided a detailed and up-to-date matrix linking them to observable physiological and behavioural domains and available tests in mice [83].
In clinical practice, diagnosis is based on self-reported symptoms and the psychopathological evaluation by a health care professional, which is then matched with diagnostic criteria of the DSM/ICD [84]. However, it is common practice to base response and remission calculations in clinical depression studies considerably or exclusively on clinical self-rating instruments like the Patient Health Questionnaire (PHQ-9) or the Beck-Depression-Inventory (BDI-II) [85,86,87]. This approach focuses on real-world feasibility by condensing depression complexity into a short questionnaire, which solely reflects an internal perspective. While this practice has its strengths and weaknesses, it sets a standard approach for measuring symptom burden in clinical practice and research. Nevertheless, the PHQ-9 has shown good validity and reliability as well as adaptability to diagnose depression [88]. Based on this clinical approach and in line with the notion of between-study comparability, interspecies homology, and the idea of a transspecies translational matrix for DSM/ICD depression criteria we argue that a similar standardized approach can be devised for laboratory mice. This approach should be empirically validated and progressively improved for in vivo depression models.
Mammals share parts of their evolutionary and phylogenetic path [44, 57], as well as the environmental and social stress during development [89,90,91]. Furthermore, both mice and humans exhibit so-called sickness behaviour—an evolutionary conserved biobehavioural response to infection or another immune trauma—with symptoms overlapping depressive symptomatology [92]. Relating the current standards to the paradigms and behavioural tests used to induce and measure depressive-like behaviour in mice [16, 17] it can be reasoned that related species are predestined to develop similar but not necessarily identical symptoms indicative of a shared emotional and cognitive continuum. Growing evidence supports the advanced abilities of non-human mammals like mice [93, 94] including, but not limited to the notion of self-awareness, consciousness [95,96,97,98,99], cognition [100, 101], personality-like traits [102, 103], and complex social capabilities [93, 104], along with the ability to suffer [105] and experience emotions [101, 104, 106, 107]. Aside from ongoing debate concerning theory of mind in non-human mammals and the uniqueness of language [108, 109], we argue that certain non-human mammals including laboratory mice meet the minimal requirements of an ethologically ascertainable depressive-like state, namely typical neurobiological and socioemotional features. This agrees with the notion that the core experience of depression is that of a sustained negative affective and emotional state (i.e., sadness and anhedonia), which is accompanied by a subsequent and non-random socioemotional symptomatology [110]. This perspective shares the ‘if-then’ logic of the core and side symptom concept of the DSM/ICD [28, 29, 31, 32, 36]. Ethological studies have proposed a faunal equivalent of posttraumatic stress disorder and clinical depression in Asian elephants [111,112,113,114], chimpanzees [115, 116] and macaques [117]. These studies indicate that non-human mammals can display non-random and timewise stable symptom patterns indicative of a disorder-like neuropsychiatric phenotype. Ultimately, this similarity between human and certain non-human mammals suggests the notion of applying clinical, human-specific criteria of a stress-related syndrome like depression onto mice to be valid, however to a currently still uncharted, undefined, and non-standardized extent. Based on the presented evidence and the need for novel models in neuropsychiatric research [15], we ask the scientific community to consider mice as being able to display a socioemotional syndromic state related to clinical depression, virtually a species-selective faunal depression-like syndrome. We thus suggest to collectively work towards an evidence-based standard definition of this state to enhance interspecies and inter-model homology, construct, and face validity along with comparability and generalizability of findings. We term this notional faunal equivalent a DLS.
Currently, one hypothesis of a clinical criteria-based depressive syndrome equivalent in mice has been proposed. It rests on the link between chronic stress protocols and behavioural patterns in mice related to depression. Dzirasa and Covington 2012 developed a hypothesis, which they named a mouse affective syndrome (MAS) [118]. In line with the recommendations of Nestler and Hyman 2010 [119], the authors state that chronic stress regimens lead to an ethologically delimitable syndrome in mice, which represents the core features of depression. They propose using clinical criteria to assess murine behaviours framed by three larger biobehavioural domains: (i) reward related, (ii) homoeostatic factors, and (iii) biomarkers. The clinical, symptom-based approach is merged with a biological framework to classify a depressive-like state in mice. Although the MAS has not yet gained mass attention, we believe the underlying hypotheses and approach to be promising.
Blueprint of a murine depression-like syndrome
In the following, we draft the blueprint of a murine DLS. Analogous to the DSM/ICD criteria, the DLS aims to define a stable, multimodal, and primarily phenotype-based read-out rather than the depiction of a phenotypes’ inducibility. Nonetheless, aside the read-outs of interest, the induction method applied in a particular model should be provided to enable the evaluation of aetiological validity [16]. The latter is particularly important, since often used techniques to induce stress and model depression-like chronic social defeat have been originally designed for male mice [120, 121]. Just recently, protocols feasible for females have been devised [122, 123]. Analogously, certain behavioural assessments like the urine sniffing test have only been established for male mice [124]. These sex-related constraints and idiosyncrasies need to be taken into consideration in a translational framework like the DLS, meaning that both male and female mice should be included in an experimental set up and that any measurement methods including behavioural assessments must account for this. This need is substantiated by the fact that depression affects females about two times more often than males [125], as well as by a recent study by Kang and colleagues, which has demonstrated a significant difference in the genetic architecture between depressed men and women [126].
The DLS read-outs are based on the DSM-5 and ICD-11 and define the minimum phenotypes necessary, which must be present over a defined period. These symptoms are assessed and quantified using established behavioural tests and assigned to neuroevolutionary conserved domains or subconstructs of the RDoC framework. However, due to the uniqueness of human language [109], “operationalization” is exclusively based on external ratings [83]. Moreover, symptoms need to be severe enough to cause significant suffering or sociofunctional impairment to qualify as a DLS. In line with the DSM-5 and ICD-11 approach, symptom severity and its impact on sociofunctional domains is used as a syndrome severity proxy [28, 32]. Analogous to the long-established approach of the GAF [34, 35], DLS severity considers the entire sociopsychological state and functional impairment of mice. The definition also entails core biological features of depression [17, 19, 56]. Taken together, the DLS reflects two of the core model criteria, which are face validity (DSM/ICD-based syndrome) and construct validity (major biological aberrations) [17, 54, 56, 127].
The symptoms of a DLS should ideally be uniform and, as far as possible, species independent to foster reciprocal human to mouse comparability and two-way translation. However, the biological and behavioural inter-individual and -species heterogeneity renders this approach fruitless [118, 119, 128,129,130,131,132]. Therefore, DLS read-outs and time criteria must be tailored to an individual species, here mice, based on current evidence. This way, the DLS definition can be formulated ad interim, which can then serve as a starting point for its empirical substantiation and improvement.
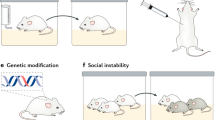
Below we outline DLS criteria specifically for laboratory mice due to their broad use in translational research. Criteria are outlined from general to specific. For illustrative purposes, we have created a translational matrix to highlight the different aspects of the translative process underlying the DLS hypothesis and the relation between mice and humans (Fig. 1).
Starting from the DSM/ICD-based depression definition, the translational matrix illustrates the process of reverse translation from clinical and thus human criteria to mice. While some of the depression symptoms (e.g., feelings of worthlessness) cannot be found in mice due to the evolutionary distance and respective species barrier, many others in addition to core biological features, sociofunctional impairment, and a minimum duration can be operationalized and quantified in mice (e.g., reduced appetite or significant weight loss). These read-outs, both in humans and mice, can be shelved into RDoC domains (Created with BioRender.com).
Minimum duration
In the DSM-5 and ICD-11, diagnosis requires 14 consecutive days with symptom presence almost all the time [28, 33]. In a real-world setting, depending on the extend of professional care provided, a major depressive episode on average lasts between 8.1 and 9.5 months [133]. Thus, it can be reasoned that an episode, on average, lasts 8-10 months. To translate this into laboratory mice, a between-species age-translation matrix is necessary. There have been a variety of attempts to relate the age of non-human mammals to the one of humans [134], for example using the eye lens weight [135], growth plate closure time [136], or molar ageing [137]. Based on the elaborate but practicable human-mouse-matrix developed by Dutta and Sengupta [134], which similar to the human-rat-matrix by Sengupta [138] is mainly based on developmental stages, 10 human months would translate into 52 h (2.16 days) in young adult mice (aged 10–64 weeks) and 176.5 h (7.35 days) in older, presenescent mice (aged 65–72 weeks). In direct contrast, translating the DSM/ICD’s 2-week criterion to mice is pointless, since this would amount to 2.4 h (0.1 days) and defy aetiological plausibility. After all, a day in the life of a mouse has the same total duration as a day in the life of a human. Still, recent studies demonstrate cellular and developmental processes of mice to be faster than those of humans [139,140,141]. Taken together, these findings highlight the complexity of age-translation between species [24] as well as the importance of age selection in mouse models [142]. To account for these developmental, cellular, and species differences, we would argue, that within the above-deducted range between 52 and 176.5 h, a longer rather than a shorter phenotype threshold is preferable. This way, the sociobiological state evoked is likely to impact the long-term individual condition of the respective mouse in terms of its reproductive success, social bonds, and physical fitness. We argue that using 52 ho of phenotype detection and presence as a minimum DLS duration definition is insufficient to significantly and reliably cause chronic, sociofunctional impairment in adult mice. But, this long-lasting sociofunctional impairment is a pivotal component of validity in the context of the DLS framework, since these ramifications essentially are the species analogue of the long-lasting socioeconomic and physical disadvantages faced by depressed patients [3]. Therefore, we recommend 7 days of phenotype presence as the DLS minimum duration. This increases the overall face validity of the DLS framework, in particular with regard to the time component of depression. However, since this analogical conclusion is primarily deduced from the human condition, it warrants step-by-step empirical adjustment so that the species differences depict the actual naturalistic duration of “murine depression” and the DLS
Due to the limitations of conventional behavioural tests, foremost the lack of repeatability of certain assessments, and the unavailability of self-rating, DLS duration needs to be captured by non-invasive methods like video tracking. This way, it is possible to ascertain which symptoms are continuously present. For this purpose, we recommend the use of continuous home-cage monitoring, as this reliably captures many depressive-like symptoms over time [143]. The feasibility of this approach using social boxes has been demonstrated many times, most recently by a work of Lopez and colleagues [144]. Still, since regular animal handling as well as other procedures and behavioural tests need to be performed during DLS assessment, continuous tracking appears difficult to some extent. More so, since handling and testing can influence home-cage activity and introduce significant bias. For the DLS framework, we thus recommend multiple short home-cage tracking periods spaced a few days apart of each other. Using this approach, the time component can be approximated via multiple measurement points of the same variables of interest, while bias can be minimized. Depending on the psychosocial or biophysical stressor used, serial measurements of social or functional impairment could serve as a proxy to ensure the evoked sociofunctional impairment phenotype is stable over the 7 days.
From a feasibility standpoint, proxies of DLS duration should primarily be based on more general symptoms like sociofunctional impairment, and only secondary on singular symptoms, like anhedonia. The rationale is that sociofunctional impairment can be measured non-invasively, whereas symptoms like anhedonia or impaired concentration and indecisiveness require hands-on assessments like the sucrose preference test or a novel object exploration task [17, 83, 145]. Thus, social or functional impairments could serve as proxies of syndrome duration, since they are relatively easy to measure, hardly bias the phenotype by investigator manipulation or enrichment, and can be combined with other depressive-like behaviours such as anhedonia [17].
Symptom duration cannot be translated directly into one distinct RDoC matrix domain, yet it can be co-reported with other factors. For example, increased duration of social avoidance can be integrated into the social process domain and changes of innate motor patterns could be assigned to the sensorimotor systems domain.
Sociofunctional impairment
In laboratory rodents, social impairment can be assessed by a plethora of social interaction and avoidance paradigms [83]. These paradigms make use of video tracking methods to quantify parameters like time spent in the interaction or avoidance zones and total movement [120, 146]. Meanwhile, functional impairment in mice is a more complex affair. For face validity, we advocate to base murine functional impairment on the clinical approach, namely the inability or negligence to perform important tasks in different areas of daily life [28, 33]. Thus, we propose a reduction in goal-directed behaviour (apathy) to assess murine functional impairment, since it reflects self-neglect and the inability to fulfil a sociobiological role. Examples of apathic mouse behaviours are impaired nest building, deterioration of coat state, reduced self-grooming and maternal care as well as diminished social interest [17, 147, 148]. Although classical social interaction and avoidance paradigms still play an important role in preclinical behavioural research, they have several limitations including the dependence on the human observer and a heavy susceptibility to confounders [149]. For that reason, novel approaches are emerging, which enable continuous assessment of complex rodent social behaviours in a naturalistic setting. Nilsson et al., for instance, have developed an open-source analysis package for freely moving rodents called Simple Behavioural Analysis (SimBA) [149]. It uses pose-estimation and supervised machine learning to provide predictive classifiers for rodent social behaviour. Another related approach is the Social Box paradigm, which was recently successfully used by Forkosh et al. [150]. It captures behaviours of freely moving mice in a group and a semi-naturalistic setting over a period of days to depict individual differences using mathematical modelling.
Taken together, using traditional or video-based assessment of sociofunctional behaviours, murine impairment can be effectively operationalized and quantified. However, the specific psychosocial or biophysical stressor regimen used to evoke chronic stress-related phenotypes may influence or limit suitable assessment methods. These quantitative and qualitative results can, for the most part, be assigned to the social processes domain of the RDoC matrix.
Biological features
To guarantee a minimum of construct validity, we propose to include pertinent and empirically substantiated biological factors of depression in the DLS definition and framework. This way, the problems associated with phenotype-based syndrome definitions like in the DSM-5 or ICD-11 are attenuated. The addition of biological read-outs respects the complex biobehavioural nature of depression. However, due the biological heterogeneity of depression [130, 151], and the plethora of aberrations associated with the disease [75], not one but several factors should be considered to determine the biological underpinnings of a DLS. Many studies have identified a complex network of biological factors, which jointly feed into the final common biological pathway of chronic stress and depression, that is a reduction in neuroplasticity in neurogenic regions [152, 153]. In particular, chronic stress and depression cause impaired neuroplasticity in the hippocampus and prefrontal cortex [74, 75, 152], which can be quantified by different methods, depending on the plasticity dimension (e.g., molecular, network or functional neuroplasticity) [152]. In murine models of depression, cellular and molecular neuroplasticity is often quantified by use of bromodeoxyuridine (BrdU) immunohistochemistry [154]. However, other biological parameters can also be used to substantiate a DLS, for example cortisol levels, neuroinflammatory markers like C-reactive protein, or adrenal gland weight. We recommend using robust and common disease features and to refrain from rare phenomena.
Impaired neuroplasticity, along with elevated cortisol levels or increased adrenal gland weight, could be included into the appropriate RDoC subconstruct (e.g., cells, molecules, or physiology) of the domain, in which the measured DLS symptom cluster belongs.
Depressive-like symptoms
As set out previously, disease and syndrome definition in clinical psychiatry and the DSM/ICD are, though experience-driven, socially, and historically skewed, and thus arbitrary to some extent. To improve this partially evidence-based status quo, we propose a data-driven approach for minimum and core symptom determination. However, it must be noted, that aside from methodological rigour, a DLS neither can nor should fully comply with the clinical classification and symptoms of depression, but rather match the most common and core criteria. Analogous to the DSM/ICD symptom criteria that is the simultaneous presence of one core plus multiple additional, but depression-typical symptoms, we argue, that the DLS needs to resemble this clinical one-plus-phenotype and syndrome. In line with the neuroevolutionary arguments presented above, it is reasonable to assume a significant overlap between humans and mice. Nonetheless, one must account for the evolutionary and biopsychosocial differences between the two species, which separated about 75 million years ago [93]. This ambivalence in the context of a partially shared ancestry and somewhat related biopsychosocial capacities is important because the underlying hypothesis of a murine DLS is to capture the faunal equivalent of clinical depression: a murine depressive-like state reflective of an evolutionary conserved entity in certain higher non-human mammals. This idea of depression as an evolutionary explicable entity intertwined with evolutionary psychiatry and psychopathology has already been brought forward by many scholar [131, 155, 156].
In a clinical setting, anhedonia is reported in about 90% of majorly or severely depressed patients [32, 157] and is well suited to distinguish severe from moderate depression [32]. Additionally, the other core symptom, depressed mood, and sadness, is considered unique to humans [74, 83]. Based on this evidence and the DSM/ICD criteria, we advocate anhedonia as the core DSL symptom. Although it has been discussed, even proposed that animals are capable of emotions [101, 104, 107, 158], the core issue remains unsolved: in non-human mammals, observational and biological measurements are insufficient to determine whether data indicative of a certain emotion actually reflect the equivalent of human sadness [93, 106]. Since we cannot talk with a mouse to assess its inner state, the verification of sadness is impossible; at least for the time being. Nevertheless, following the line of argument of animal emotion presented above, it might be fair to assume a sadness equivalent or least sadness-related emotional experience in mice. In contrast, anhedonia is widely accepted in the scientific community in regards to murine depression models and can be readily assessed, for example by the sucrose preference or cookie test [17], intracranial self-stimulation techniques [159], conditional place preference, or urine sniffing [83]. Admittedly, using anhedonia as the DLS core symptom inevitably leads to a definition revolving around anhedonia-present depression. However, Buckner et al. 2008 were able to demonstrate that although anhedonia-present depression differs from sadness-present depression in terms of symptom cluster, there is an overlap in symptoms, particularly regarding reactivity of mood, social impairment, and social withdrawal [157]. Patients with a strong depressive phenotype (i.e., severe clinical depression) display a high concurrence of the DSM-5 core depression symptoms: 100% of patients report sadness, and 90% anhedonia [32]. Based on this, we would argue that a distinction between anhedonia- and sadness-present depression is mostly artificial and not representative of the real-world situation. Thus, centring the DLS definition around anhedonia is reasonable.
On these grounds, we propose to define the murine DLS symptom cluster centred around anhedonia, meaning that anhedonia plus additional, depression-typical symptoms need to be present to constitute the DLS. Concerning these additional, depression-typical symptoms, Czéh et al. have developed a detailed matrix that matches most DSM-5 depression criteria to distinct physiological and behavioural phenomena as well as available quantification methods in rodents [83]. This comprehensive matrix can serve as a basis for the assessment of DLS symptoms. But the question remains: how many additional symptoms are necessary for mice? Due to the species barrier and the fewer depressive symptoms present in mice, we advocate a total of three additional symptoms. To assess the presence of each depressive symptom, we suggest using two different tests, if possible. For instance, to confirm the presence of anhedonia, mice should display biologically plausible and robust differences to a respective control group in the sucrose preference and urine sniffing test. Using this approach, symptom presence is concurrently validated by two measurements of the same symptom, which increases validity and power and enables binarization. Ultimately, this allows to confirm the presence or absence of a symptom. The number of symptoms necessary to constitute a valid and robust depressive-like phenotype and DLS remains to be determined. Prospectively, this can be achieved using a fact-driven, prospective clustering approach, that consolidates the association between different symptom clusters, the other DLS criteria including anhedonia and other, already validated, murine depression models.
Due to the versatile quality of depressive symptoms (cognitive, behavioural, emotional, and somatic), the respective symptom can be easily translated to the RDoC matrix. Impaired concentration and indecisiveness, which can be measured using a Y-maze or a novel object exploration test [83], belong in the RDoC cognition domain. Meanwhile, a disturbed sleep pattern is part of the arousal and regulation domain. Similarly, most DSM-5 symptoms can be categorized into a specific RDoC domain.
Syndrome severity
As outlined above, the DSM-5 estimates depression severity on symptom quantity and intensity, and social as well as functional impairment [29, 31, 33]. In a clinical setting, these criteria are assessed by instruments such as the Hamilton Depression Rating Scale (HAM-D), BDI-II, or GAF score [32, 34, 35]. Similarly, DLS severity estimates should be based on overall sociofunctional impairment and symptom degree [28, 32]. A recent study by Elmer and Stadtfeld 2020 on the depression-isolation hypothesis found that depressed individuals spend less time in social interactions and with known friends, and more time with depressed others and in pair-wise interactions. In addition, the study found that depressive symptoms were negatively correlated with the time spent interacting socially [160]. These findings support the notion that a reduction or non-beneficial change of social behaviours can serve as the primary DLS severity proxy. In addition, symptom degree could be added to approximate the DSM-5 outline. However, in contrast to the DSM-5 and ICD-11 severity assessment, which lacks quantitative standardization, we would argue that to achieve a reliable and biologically valid severity estimate, the approach should be strictly data-driven and based on indexing sociofunctional impairment and co-occurring symptoms. Fortunately, the available tests enable quantification and subsequent binarization of symptoms [83]. Whether the clinical severity categories ‘mild, moderate, and severe’ are detectable and useful in mice remains to be empirically determined [33].
Statistical algorithm for depression-like syndrome assessment
Having outlined the DLS blueprint and framework, we finally propose an algorithm to assess the presence of a DLS and its severity. This algorithm uses statistical instruments to guarantee a standardized and data-driven approach.
Due to the issues arising from the difference between statistical and biological significance or relevance, and the associated risk of false discovery [161, 162], we argue that basing DLS criteria presence on statistical significance alone is insufficient. We suggest the combined use of effect size and statistical significance. Given the principal validity of the biobehavioural DLS framework, this dual heuristic approach would enable the biological significance and plausibility of a difference between a group of interest and healthy controls to be found with high probability [163, 164]. This hybrid concept has been termed a minimum effect size plus p value (MESP) by Goodman et al. [163]. For the DLS, we recommend a statistical significance level of alpha ≤ 0.05 as well as a moderate effect size (for example: Cohens d ≥ 0,5 or Cohens f2 ≥ 0.252, depending on the respective statistical measure) as the minimum combined threshold. These DLS-MESP criteria should be obtained from comparison between the group of interest and a healthy control group using sample-appropriate statistical testing and, if necessary, post-hoc correction. We suggest employing two appropriate read-outs for each DLS criterion. Tests and read-outs should meet the DLS-MESP criteria. If only one of the two selected read-outs per symptom domain meets the DLS-MESP criterion, it might be reasonable to report the respective finding and base binarization (DLS symptom present: yes/no) on overall biological plausibility by considering other data for the same DLS criterium and animal. In select cases, however, it might also be feasible to augment a third measurement for the symptom and DLS criterium in question to substantiate binarization in the case of inconsistency between the initially selected two read-outs. Alternatively, investigators might consider assessing another DLS criterium in the same animal to achieve the minimum of four present DLS criteria. While this approach might be justifiable in some situations, a post-hoc augmentation of any read-out should be performed with extreme caution and while ensuring strict blinding of any experimenter involved, since the risk of selection and confirmation bias is high. Therefore, in most cases, we would advise authors to report an incomplete DLS and disclose missing or inconsistent findings as well as their statistical approach and reasoning in detail.
To assess the presence of a DLS, the most basic condition, that is the duration represented by sociofunctional impairment needs to have a duration of 7 days or more. The gathered data should be compared between groups using two parameters, which need to reflect a reduction in social interaction, or a more complex behavioural read-out like the z-score. In other behavioural sciences, the z-score has been successfully employed [165, 166]. For all DLS read-outs including the duration criterion, we strongly recommend using the z-score, since it provides a standardized score for the read-outs of interest, normalized for group mean and standard deviation (SD). This allows comparison between studies [165], which is the ultimate aim of the DLS framework. The z-test states how many SDs (σ) a single observation (X) deviates from the mean of a control group (μ). Hence, the following formula applies for each individual read-out:
The singular z-test values, which represent a single read-out each, then need to be corrected for directionality, so that an increased score will reflect the increase of interest i.e., sociofunctional impairment. To calculate the final z-score for social interaction impairment, the individual z-scores of the read-outs reflecting social impairment need to be added up and divided by the number of tests, as below [165]:
By using two single test read-outs or a z-score of multiple social interaction impairment proxies, statistical instruments like a repeated measures analysis of variance (ANOVA) enable the calculation of differences over the duration or per point in time. Therefore, if DLS-MESP criteria are met, the minimum duration criterion is fulfilled. Consequently, since the duration and sociofunctional impairment criteria are connected, this approach also allows one to state if the animal experienced significant sociofunctional impairment. The degree of impairment can be assessed based on the SD by comparison of the cumulative z-scores between the group of interest and the control groups. We advocate the three clinically analogous levels of severity: mild, moderate, and severe. Animals in the group of interest within one SD are mildly impaired, within the second SD are moderately impaired, and within the third SD are severely impaired. However, whether the human severity estimate is feasible for mice remains to be determined.
To determine the presence of the required biological features, we recommend using two depression-typical read-outs. Both tests per read-out need to comply with DLS-MESP criteria. However, using more than two features is likely to increase the validity of the measurements and thus DLS presence. Here, also the z-score approach could be useful to combine multiple biological read-outs and then compare the group of interest with the controls in respect to the DLS-MESP criteria.
Finally, to determine the presence of the minimum requirement of four symptoms, namely anhedonia plus three other depression-typical symptoms, we advocate using two or more behavioural tests per symptom. Both tests per symptom should meet the DLS-MESP criteria. As for the other DLS criteria, symptom assessments can either be assessed individually or be summed up into a z-score, which then can be evaluated according to the DLS-MESP criteria. Although we deem both approaches practically feasible and tenable, we suggest the use of a z-score since it enables reliable between-study comparison later on.
In summary, we have outlined how the different biobehavioural levels can be assessed, analyzed and binarized based on the DLS-MESP criteria. This will enable researchers to clearly state the presence or absence of a DLS in mice (Fig. 2).
Condensed summary of the assessment process revolving around the DLS criteria. Based on the biobehavioural assessment, the algorithm allows to state if a DLS is in fact present or absent. Moreover, it enables severity estimation based on the comparison of the group of interest with controls using the standard deviation difference between the two groups’ z-scores (Created with BioRender.com).
Conclusion and perspectives
In summary, we herein have drafted a blueprint of a novel reference syndrome for murine depression models in translational psychiatry using defined read-out parameters based on readily available tests and methods. The DLS is a minimum threshold approach combining symptom duration and composition as well as sociofunctional impairment with core biological features. Although we have focused on a DLS approach for mice, it could be easily adjusted for laboratory rats. We believe that as a scientific community entrusted with advancing the understanding of psychiatric diseases, it is our shared obligation to consider every well-reasoned, auspicious, and ethically acceptable proposal and evaluate its strengths and weaknesses over time. A very popular example of such a proposal, admittedly on a more systemic and global level, are the 2010 published RDoC. Somewhat similar, the DLS is a work in progress, a preliminary defined murine psychosocial and biological state related to clinical depression that warrants constant testing and validation by data collection, interdisciplinary synopsis, and step-by-step optimization. The long-term objective of the DLS is to develop a between-model validation standard to improve the widespread use of imprecise phenotyping language and methodology in preclinical models of depression. We believe that an evidence-based DLS definition can provide a microframework for depression research, thus fostering the translation of clinical and preclinical findings. In the future, this DLS microframework can be further improved by the so-called ‘evo-mecho’ approach, which was recently developed by Taborsky et al. 2021. It is a comprehensive research programme connecting evolutionary modelling to empirical research, which will advance our understanding of the stress response evolution based on species and context [167]. The respective findings could help refine the DLS hypothesis and criteria of a non-random, maladaptive biobehavioural stress response pattern that is, at its very core, conserved between certain higher mammals.
The proposed syndromic DLS approach is an auspicious and labour-intensive venture and, for now, relies primarily on old fashioned behavioural assessments, which base complex phenotypes on rather simple but well-established tests like sucrose preference or social avoidance. For that reason, we propose the use of combined and normalized measure like the z-score, since this enables inter-study comparability and complex assessments, while maintaining the use of established methods and knowledge. We believe, that a DLS blueprint based on these tests might increase feasibility and acceptance, and thus aid the transformation towards consensus-based phenotyping. This is pivotal to link existing evidence based on the established tests to a DLS-based assessment and framework. Advanced phenotyping tools like the Social Box Paradigm, SimBA package, IntelliCage [168] or PsyCo [169] along with advanced facial expression tracking are gaining ground [170]. In the future, these methods could help overcome most constraints of conventional testing, foster complex assessments and read-outs, and ultimately feedback into DLS criteria. Given the purely translational hypothesis and empiric foundation of the DLS blueprint, it should prove a valuable addition to the methodological toolbox at the intersection of clinical and animal research in neuropsychiatry. Moreover, it could improve between-species comparability, aid translatability by advancing phenotypic profiling and increase overall quality of the current murine depression models. In addition, a consensus like the DLS introduces the syndrome component to animal modelling, a clinical axiom and core component of human depression widely neglected in preclinical and translational neuropsychiatry. The DLS is a longitudinal, biobehavioural definition designed to mirror and capture a syndromic state. It is not a risk or endophenotype model. In addition to the ever-necessary control group of an experimental set up, the DLS could also serve as a standardized validation option and replication platform for animal models with a clinical objective, while increasing construct and face validity.
Arguably, certain research questions might not require the syndrome concept to produce valuable findings. However, many others and especially clinically oriented translational studies might benefit from considering this clinical and human feature of depression. The DLS could fill the gap between RDoC and clinical questions of applicability and increase model scope. We urge the scientific community to consider the potential of a valid, reliable, and reproducible syndrome definition and phenotyping standard for murine depression models.
References
GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018;392:1789–858.
König H, König HH, Konnopka A. The excess costs of depression: a systematic review and meta-analysis. Epidemiol Psychiatr Sci. 2019;29:e30.
Kessler RC. The costs of depression. Psychiatr Clin N Am. 2012;35:1–14.
Moreno-Agostino D, Wu Y-T, Daskalopoulou C, Hasan MT, Huisman M, Prina M. Global trends in the prevalence and incidence of depression:a systematic review and meta-analysis. J Affect Disord. 2021;281:235–43.
Licinio J, Wong M-L. Advances in depression research: second special issue, 2020, with highlights on biological mechanisms, clinical features, co-morbidity, genetics, imaging, and treatment. Mol Psychiatry. 2020;25:1356–60.
Kraus C, Kadriu B, Lanzenberger R, Zarate CA Jr, Kasper S. Prognosis and improved outcomes in major depression: a review. Transl Psychiatry. 2019;9:127.
Söderlund J, Lindskog M. Relevance of rodent models of depression in clinical practice: can we overcome the obstacles in translational neuropsychiatry? Int J Neuropsychopharmacol 2018;21:668–76.
Cuijpers P, Stringaris A, Wolpert M. Treatment outcomes for depression: challenges and opportunities. Lancet Psychiatry. 2020;7:925–7.
Maslej MM, Furukawa TA, Cipriani A, Andrews PW, Sanches M, Tomlinson A, et al. Individual differences in response to antidepressants: a meta-analysis of placebo-controlled randomized clinical trials. JAMA Psychiatry. 2021;78:490–7.
Richards D. Prevalence and clinical course of depression: a review. Clin Psychol Rev. 2011;31:1117–25.
Nuggerud-Galeas S, Sáez-Benito Suescun L, Berenguer Torrijo N, Sáez-Benito Suescun A, Aguilar-Latorre A, Magallón Botaya R, et al. Analysis of depressive episodes, their recurrence and pharmacologic treatment in primary care patients: a retrospective descriptive study. PLoS ONE. 2020;15:e0233454.
Mattisson C, Bogren M, Horstmann V, Munk-Jörgensen P, Nettelbladt P. The long-term course of depressive disorders in the Lundby Study. Psychol Med. 2007;37:883–91.
Kennedy N, Abbott R, Paykel ES. Remission and recurrence of depression in the maintenance era: long-term outcome in a Cambridge cohort. Psychol Med. 2003;33:827–38.
Cipriani A, Furukawa TA, Salanti G, Chaimani A, Atkinson LZ, Ogawa Y, et al. Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: a systematic review and network meta-analysis. Lancet 2018;391:1357–66.
Bale TL, Abel T, Akil H, Carlezon WA Jr., Moghaddam B, Nestler EJ, et al. The critical importance of basic animal research for neuropsychiatric disorders. Neuropsychopharmacology 2019;44:1349–53.
Gururajan A, Reif A, Cryan JF, Slattery DA. The future of rodent models in depression research. Nat Rev Neurosci. 2019;20:686–701.
Planchez B, Surget A, Belzung C. Animal models of major depression: drawbacks and challenges. J Neural Transm. 2019;126:1383–408.
Hanrahan C, New JP. Antidepressant medications: the FDA-approval process and the need for updates. Ment Health Clin. 2014;4:11–6.
Belzung C. Innovative drugs to treat depression: did animal models fail to be predictive or did clinical trials fail to detect effects? Neuropsychopharmacology 2014;39:1041–51.
Kalueff AV, Wheaton M, Murphy DL. What’s wrong with my mouse model?: Advances and strategies in animal modeling of anxiety and depression. Behav Brain Res. 2007;179:1–18.
Voelkl B, Altman NS, Forsman A, Forstmeier W, Gurevitch J, Jaric I, et al. Reproducibility of animal research in light of biological variation. Nat Rev Neurosci. 2020;21:384–93.
Macleod M, Mohan S. Reproducibility and rigor in animal-based research. ILAR J. 2019;60:17–23.
Kafkafi N, Agassi J, Chesler EJ, Crabbe JC, Crusio WE, Eilam D, et al. Reproducibility and replicability of rodent phenotyping in preclinical studies. Neurosci Biobehav Rev. 2018;87:218–32.
Bell AM, Hankison SJ, Laskowski KL. The repeatability of behaviour: a meta-analysis. Anim Behav. 2009;77:771–83.
Baker M, Hong S-I, Kang S, Choi D-S. Rodent models for psychiatric disorders: problems and promises. Lab Anim Res. 2020;36:9.
Kenneth S, Kendler MD. The phenomenology of major depression and the representativeness and nature of DSM Criteria. Am J Psychiatry. 2016;173:771–80.
Tzeferakos G, Douzenis A. Sacred psychiatry in ancient Greece. Ann Gen Psychiatry. 2014;13:11.
Stein DJ, Szatmari P, Gaebel W, Berk M, Vieta E, Maj M, et al. Mental, behavioral and neurodevelopmental disorders in the ICD-11: an international perspective on key changes and controversies. BMC Med. 2020;18:21.
Abramson LY, Metalsky GI, Alloy LB. Hopelessness depression: a theory-based subtype of depression. Psychological Rev. 1989;96:358–72.
Horst Dilling WM, Schmidt MH. Internationale Klassifikation psychischer Störungen - ICD-10 Kapitel V (F), Klinisch-diagnostische Leitlinie. Bern/Genf: hogrefe Verlag, WHO; 2015.
Zimmerman M, Morgan TA, Stanton K. The severity of psychiatric disorders. World Psychiatry. 2018;17:258–75.
Tolentino JC, Schmidt SL. DSM-5 criteria and depression severity: implications for clinical practice. Front Psychiatry. 2018;9:450.
APA. Depressive disorders. In: Diagnostic and statistical manual of mental disorders (DSM-5). Washington (DS), London (England): American PSychiatric Publishing (A Divison of the American Psychiatric Association); 2013.
Aas IHM. Guidelines for rating Global Assessment of Functioning (GAF). Ann Gen Psychiatry. 2011;10:2.
Aas IHM, Sonesson O, Torp S. A qualitative study of clinicians experience with rating of the Global Assessment of Functioning (GAF) Scale. Community Ment Health J. 2018;54:107–16.
McGlinchey JB, Zimmerman M, Young D, Chelminski I. Diagnosing major depressive disorder VIII: are some symptoms better than others? J Nerv Ment Dis. 2006;194:785–90.
Reed GM, Sharan P, Rebello TJ, Keeley JW, Elena Medina-Mora M, Gureje O, et al. The ICD-11 developmental field study of reliability of diagnoses of high-burden mental disorders: results among adult patients in mental health settings of 13 countries. World Psychiatry. 2018;17:174–86.
Zou W, Feng R, Yang Y. Changes in the serum levels of inflammatory cytokines in antidepressant drug-naïve patients with major depression. PLoS ONE. 2018;13:e0197267.
Reed GM, Ayuso-Mateos JL. Towards a more clinically useful International World Health Organisation classification of mental disorders. Rev Psiquiatr Salud Ment. 2011;4:113–6.
Frances AJ, Nardo JM. ICD-11 should not repeat the mistakes made by DSM-5. Br J Psychiatry. 2013;203:1–2.
Montgomery S. Are the ICD-10 or DSM-5 diagnostic systems able to define those who will benefit from treatment for depression? CNS Spectr. 2016;21:283–8.
Clark LA, Cuthbert B, Lewis-Fernández R, Narrow WE, Reed GM. Three approaches to understanding and classifying mental disorder: ICD-11, DSM-5, and the National Institute of Mental Health’s Research Domain Criteria (RDoC). Psychol Sci Public Interest. 2017;18:72–145.
Tyrer P. A comparison of DSM and ICD classifications of mental disorder. Adv Psychiatr Treat. 2014;20:280–5.
Lilienfeld SO. The Research Domain Criteria (RDoC): an analysis of methodological and conceptual challenges. Behav Res Ther. 2014;62:129–39.
Frances A. RDoC is necessary, but very oversold. World Psychiatry. 2014;13:47–9.
Radden J. Rethinking disease in psychiatry: disease models and the medical imaginary. J Eval Clin Pract. 2018;24:1087–92.
Ruggero CJ, Kotov R, Hopwood CJ, First M, Clark LA, Skodol AE, et al. Integrating the Hierarchical Taxonomy of Psychopathology (HiTOP) into clinical practice. J Consult Clin Psychol. 2019;87:1069–84.
Kotov R, Krueger RF, Watson D, Achenbach TM, Althoff RR, Bagby RM, et al. The Hierarchical Taxonomy of Psychopathology (HiTOP): a dimensional alternative to traditional nosologies. J Abnorm Psychol. 2017;126:454–77.
Wakefield JC. DSM-5, psychiatric epidemiology and the false positives problem. Epidemiol Psychiatr Sci. 2015;24:188–96.
Murphy D. “Philosophy of Psychiatry”, The Stanford Encyclopedia of Philosophy (Fall 2020 Edition),: Edward N. Zalta. https://www.plato.stanford.edu/archives/fall2020/entries/psychiatry/.
Krueger RF, Kotov R, Watson D, Forbes MK, Eaton NR, Ruggero CJ, et al. Progress in achieving quantitative classification of psychopathology. World Psychiatry. 2018;17:282–93.
Brückl TM, Spoormaker VI, Sämann PG, Brem AK, Henco L, Czamara D, et al. The biological classification of mental disorders (BeCOME) study: a protocol for an observational deep-phenotyping study for the identification of biological subtypes. BMC Psychiatry. 2020;20:213.
Waszczuk MA, Eaton NR, Krueger RF, Shackman AJ, Waldman ID, Zald DH, et al. Redefining phenotypes to advance psychiatric genetics: implications from hierarchical taxonomy of psychopathology. J Abnorm Psychol. 2020;129:143–61.
Willner P. Validation criteria for animal models of human mental disorders: learned helplessness as a paradigm case. Prog Neuropsychopharmacol Biol Psychiatry. 1986;10:677–90.
Boyko M, Kutz R, Grinshpun J, Zvenigorodsky V, Gruenbaum BF, Gruenbaum SE, et al. The effect of depressive-like behavior and antidepressant therapy on social behavior and hierarchy in rats. Behav Brain Res. 2019;370:111953.
Hao Y, Ge H, Sun M, Gao Y. Selecting an appropriate animal model of depression. Int J Mol Sci. 2019;20:4827.
Anderzhanova E, Kirmeier T, Wotjak CT. Animal models in psychiatric research: The RDoC system as a new framework for endophenotype-oriented translational neuroscience. Neurobiol Stress. 2017;7:47–56.
Monteggia LM, Heimer H, Nestler EJ. Meeting report: can we make animal models of human mental illness? Biol Psychiatry. 2018;84:542–5.
Solberg LC, Ahmadiyeh N, Baum AE, Vitaterna MH, Takahashi JS, Turek FW, et al. Depressive-like behavior and stress reactivity are independent traits in a Wistar Kyoto × Fisher 344 cross. Mol Psychiatry. 2003;8:423–33.
Iwata M, Ota KT, Li X-Y, Sakaue F, Li N, Dutheil S, et al. Psychological stress activates the inflammasome via release of adenosine triphosphate and stimulation of the purinergic type 2x7 receptor. Biol Psychiatry. 2016;80:12–22.
Moraes MMT, Galvão MC, Cabral D, Coelho CP, Queiroz-Hazarbassanov N, Martins MFM, et al. Propentofylline prevents sickness behavior and depressive-like behavior induced by lipopolysaccharide in rats via neuroinflammatory pathway. PLoS ONE. 2017;12:e0169446.
Du X, Yin M, Yuan L, Zhang G, Fan Y, Li Z, et al. Reduction of depression-like behavior in rat model induced by ShRNA targeting norepinephrine transporter in locus coeruleus. Transl Psychiatry. 2020;10:130.
Thomas Insel MD, Cuthbert B, Garvey M, Heinssen R, Pine DS, Quinn K, et al. Research Domain Criteria (RDoC): toward a new classification framework for research on mental disorders. Am J Psychiatry. 2010;167:748–51.
Insel TR. The NIMH Research Domain Criteria (RDoC) Project: precision medicine for psychiatry. Am J Psychiatry. 2014;171:395–7.
Salles J, Lacassagne E, Benvegnu G, Berthoumieu SÇ, Franchitto N, Tauber M. The RDoC approach for translational psychiatry: Could a genetic disorder with psychiatric symptoms help fill the matrix? the example of Prader–Willi syndrome. Transl Psychiatry. 2020;10:274.
Hart KL, Perlis RH, McCoy TH. Mapping of transdiagnostic neuropsychiatric phenotypes across patients in two general hospitals. J Acad Consultation-Liaison Psychiatry. 2021;62:430–9.
Lilienfeld SO, Treadway MT. Clashing diagnostic approaches: DSM-ICD versus RDoC. Annu Rev Clin Psychol. 2016;12:435–63.
Ross CA, Margolis RL. Research domain criteria: strengths, weaknesses, and potential alternatives for future psychiatric research. Complex Psychiatry 2019;5:218–36.
Cuthbert BN, Workgroup NR. The RDoC framework: continuing commentary. World Psychiatry. 2014;13:196–7.
Pernu TK. Elimination, not reduction: lessons from the Research Domain Criteria (RDoC) and multiple realisation. Behav Brain Sci. 2019;42:e22.
Cuthbert BN, Kozak MJ. Constructing constructs for psychopathology: the NIMH research domain criteria. J Abnorm Psychol. 2013;122:928–37.
Fluyau D. Integrating DSM/ICD, research domain criteria, and descriptive psychopathology in teaching and practice of psychiatry. Front Psychiatry. 2018;9:484. https://doi.org/10.3389/fpsyt.2018.00484.
Cuthbert BN. The RDoC framework: facilitating transition from ICD/DSM to dimensional approaches that integrate neuroscience and psychopathology. World Psychiatry. 2014;13:28–35.
Krishnan V, Nestler EJ. Animal models of depression: molecular perspectives. Curr Top Behav Neurosci. 2011;7:121–47.
Krishnan V, Nestler EJ. The molecular neurobiology of depression. Nature 2008;455:894–902.
Ménard C, Hodes GE, Russo SJ. Pathogenesis of depression: insights from human and rodent studies. Neuroscience 2016;321:138–62.
Yirmiya R, Rimmerman N, Reshef R. Depression as a microglial disease. Trends Neurosci. 2015;38:637–58.
Tafet GE, Nemeroff CB. The links between stress and depression: psychoneuroendocrinological, genetic, and environmental interactions. J Neuropsychiatry Clin Neurosci. 2016;28:77–88.
Ostojić L, Tkalčić M, Clayton NS. Are owners’ reports of their dogs’ ‘guilty look’ influenced by the dogs’ action and evidence of the misdeed? Behav Process. 2015;111:97–100.
Hecht J, Miklósi Á, Gácsi M. Behavioral assessment and owner perceptions of behaviors associated with guilt in dogs. Appl Anim Behav Sci. 2012;139:134–42.
Horowitz A. Disambiguating the “guilty look”: salient prompts to a familiar dog behaviour. Behav Process. 2009;81:447–52.
Shen L. The evolution of shame and guilt. PLoS ONE. 2018;13:e0199448.
Czéh B, Fuchs E, Wiborg O, Simon M. Animal models of major depression and their clinical implications. Prog Neuropsychopharmacol Biol Psychiatry. 2016;64:293–310.
Fähndrich E, Stieglitz R-D. Leitfaden zur Erfassung des psychopathologischen Befunds. Göttingen: hogrefe; 2018.
Kendrick T, Moore M, Leydon G, Stuart B, Geraghty AWA, Yao G, et al. Patient-reported outcome measures for monitoring primary care patients with depression (PROMDEP): study protocol for a randomised controlled trial. Trials 2020;21:441.
Salaminios G, Duffy L, Ades A, Araya R, Button KS, Churchill R, et al. A randomised controlled trial assessing the severity and duration of depressive symptoms associated with a clinically significant response to sertraline versus placebo, in people presenting to primary care with depression (PANDA trial): study protocol for a randomised controlled trial. Trials 2017;18:496.
Lewis G, Duffy L, Ades A, Amos R, Araya R, Brabyn S, et al. The clinical effectiveness of sertraline in primary care and the role of depression severity and duration (PANDA): a pragmatic, double-blind, placebo-controlled randomised trial. Lancet Psychiatry. 2019;6:903–14.
Sun Y, Fu Z, Bo Q, Mao Z, Ma X, Wang C. The reliability and validity of PHQ-9 in patients with major depressive disorder in psychiatric hospital. BMC Psychiatry. 2020;20:474.
Nevo E. Evolution under environmental stress at macro- and microscales. Genome Biol Evolution. 2011;3:1039–52.
Flinn M. Evolution of stress response to social threat. In: Oxford handbook of evolutionary psychology. Oxford University Press; 2012. p. 273–96. https://doi.org/10.1093/oxfordhb/9780198568308.013.0020.
Nesse R, Bhatnagar S, Ellis B. Evolutionary origins and functions of the stress response system. In: Concepts, Cognition, Emotion, and Behavior. Handbook of Stress Series 1st Ed. Amsterdam, Co. Elsevier; 2016. Vol. 1 p. 95–101.
Maes M, Berk M, Goehler L, Song C, Anderson G, Gałecki P, et al. Depression and sickness behavior are Janus-faced responses to shared inflammatory pathways. BMC Med. 2012;10:66.
Mogil JS. Mice are people too: Increasing evidence for cognitive, emotional and social capabilities in laboratory rodents. Can Psychol. 2019;60:14–20.
Proctor H. Animal sentience: where are we and where are we heading? Animals 2012;2:628–39.
David DeGrazia: Self-awareness in animals. In: Robert W. Lurz editor. The Philosophy of Animal Minds. Cambridge: Cambridge University Press; 2009. p. 201–17.
Hyatt CW, Hopkins WD. Self-awareness in bonobos and chimpanzees: a comparative perspective. In: Boccia ML, Mitchell RW, Parker ST, editors. Self-awareness in animals and humans: developmental perspectives. Cambridge: Cambridge University Press; 1994. p. 248–53.
Ueno H, Suemitsu S, Murakami S, Kitamura N, Wani K, Takahashi Y, et al. Behavioural changes in mice after getting accustomed to the mirror. Behav Neurol. 2020;2020:4071315.
Bekoff M. Awareness: animal reflections. Nature 2002;419:255.
Birch J, Schnell AK, Clayton NS. Dimensions of animal consciousness. Trends Cogn Sci. 2020;24:789–801.
Stephan M, Volkmann P, Rossner MJ. Assessing behavior and cognition in rodents, nonhuman primates, and humans: where are the limits of translation? Dialogues Clin Neurosci. 2019;21:249–59.
Al Dahhan NZ, De Felice FG, Munoz DP. Potentials and pitfalls of cross-translational models of cognitive impairment. Front Behav Neurosci. 2019;13.
Cabrera D, Nilsson JR, Griffen BD. The development of animal personality across ontogeny: a cross-species review. Anim Behav. 2021;173:137–44.
Sih A, Mathot KJ, Moirón M, Montiglio P-O, Wolf M, Dingemanse NJ. Animal personality and state–behaviour feedbacks: a review and guide for empiricists. Trends Ecol Evolution. 2015;30:50–60.
Bekoff M. Animal Emotions: Exploring Passionate Natures: Current interdisciplinary research provides compelling evidence that many animals experience such emotions as joy, fear, love, despair, and grief—we are not alone. BioScience 2000;50:861–70.
Sollereder B. When humans are not unique: perspectives on suffering and redemption. Expository 2016;127:269–76.
de Vere AJ, Kuczaj SA II. Where are we in the study of animal emotions? WIREs. Cogn Sci. 2016;7:354–62.
Kremer L, Klein Holkenborg SEJ, Reimert I, Bolhuis JE, Webb LE. The nuts and bolts of animal emotion. Neurosci Biobehav Rev. 2020;113:273–86.
Krupenye C, Call J. Theory of mind in animals: current and future directions. WIREs. Cogn Sci. 2019;10:e1503.
Fisher SE, Marcus GF. The eloquent ape: genes, brains and the evolution of language. Nat Rev Genet. 2006;7:9–20.
Kanter JW, Busch AM, Weeks CE, Landes SJ. The nature of clinical depression: symptoms, syndromes, and behavior analysis. Behav Anal. 2008;31:1–21.
Cantor C. Post-traumatic stress disorder: evolutionary perspectives. Aust NZ J Psychiatry. 2009;43:1038–48.
Bradshaw GA, Schore AN, Brown JL, Poole JH, Moss CJ. Elephant breakdown. Nature 2005;433:807.
Shannon G, Slotow R, Durant SM, Sayialel KN, Poole J, Moss C, et al. Effects of social disruption in elephants persist decades after culling. Front Zool. 2013;10:62.
Rizzolo JB, Bradshaw GA. Prevalence and patterns of complex PTSD in Asian elephants (Elephas maximus). In: Wickramaarachchi T, de Zoysa HN, editors. International Conference on Asian Elephants in Culture & Nature, 20th – 21st August 2016, Anura Manatunga, K.A.T. Chamara. Sri Lanka: Centre for Asian Studies, University of Kelaniya; 2016. p. 180.
Bradshaw GA, Capaldo T, Lindner L, Grow G. Building an inner sanctuary: complex PTSD in Chimpanzees. J Trauma Dissociation. 2008;9:9–34.
Ferdowsian HR, Durham DL, Kimwele C, Kranendonk G, Otali E, Akugizibwe T, et al. Signs of mood and anxiety disorders in chimpanzees. PLoS ONE. 2011;6:e19855.
Xu F, Wu Q, Xie L, Gong W, Zhang J, Zheng P, et al. Macaques exhibit a naturally-occurring depression similar to humans. Sci Rep. 2015;5:9220.
Dzirasa K, Covington HE 3rd. Increasing the validity of experimental models for depression. Ann N Y Acad Sci. 2012;1265:36–45.
Nestler EJ, Hyman SE. Animal models of neuropsychiatric disorders. Nat Neurosci. 2010;13:1161–9.
Golden SA, Covington HE 3rd, Berton O, Russo SJ. A standardized protocol for repeated social defeat stress in mice. Nat Protoc. 2011;6:1183–91.
Hollis F, Kabbaj M. Social defeat as an animal model for depression. ILAR J. 2014;55:221–32.
Harris AZ, Atsak P, Bretton ZH, Holt ES, Alam R, Morton MP, et al. A novel method for chronic social defeat stress in female mice. Neuropsychopharmacology 2018;43:1276–83.
van Doeselaar L, Yang H, Bordes J, Brix L, Engelhardt C, Tang F, et al. Chronic social defeat stress in female mice leads to sex-specific behavioral and neuroendocrine effects. Stress 2021;24:168–80.
Malkesman O, Scattoni ML, Paredes D, Tragon T, Pearson B, Shaltiel G, et al. The female urine sniffing test: a novel approach for assessing reward-seeking behavior in rodents. Biol Psychiatry. 2010;67:864–71.
Van de Velde S, Bracke P, Levecque K. Gender differences in depression in 23 European countries. Cross-national variation in the gender gap in depression. Soc Sci Med. 2010;71:305–13.
Kang H-J, Park Y, Yoo K-H, Kim K-T, Kim E-S, Kim J-W, et al. Sex differences in the genetic architecture of depression. Sci Rep. 2020;10:9927.
Willner P. Validity, reliability and utility of the chronic mild stress model of depression: a 10-year review and evaluation. Psychopharmacology 1997;134:319–29.
Goldberg D. The heterogeneity of “major depression”. World Psychiatry. 2011;10:226–8.
Lux V, Kendler KS. Deconstructing major depression: a validation study of the DSM-IV symptomatic criteria. Psychol Med. 2010;40:1679–90.
Milaneschi Y, Lamers F, Berk M, Penninx B. Depression heterogeneity and its biological underpinnings: toward immunometabolic depression. Biol Psychiatry. 2020;88:369–80.
Rantala MJ, Luoto S, Krams I, Karlsson H. Depression subtyping based on evolutionary psychiatry: Proximate mechanisms and ultimate functions. Brain Behav Immun. 2018;69:603–17.
Rush AJ. The varied clinical presentations of major depressive disorder. J Clin Psychiatry. 2007;68:4–10.
Spijker J, De Graaf R, Bijl RV, Beekman ATF, Ormel J, Nolen WA. Duration of major depressive episodes in the general population: results from the Netherlands Mental Health Survey and Incidence Study (NEMESIS). Br J Psychiatry. 2002;181:208–13.
Dutta S, Sengupta P. Men and mice: relating their ages. Life Sci. 2016;152:244–8.
Augusteyn RC. Growth of the eye lens: I. Weight accumulation in multiple species. Mol Vis. 2014;20:410–26.
Kilborn SH, Trudel G, Uhthoff H. Review of growth plate closure compared with age at sexual maturity and lifespan in laboratory animals. Contemp Top Lab Anim Sci. 2002;41:21–6.
Pankakoski E. An improved method for age determination in the muskrat, Ondatra zibethica (L.). Ann Zool Fennici. 1980;17:113–21.
Sengupta P. The laboratory rat: relating its age with human’s. Int J Prev Med. 2013;4:624–30.
Matsuda M, Hayashi H, Garcia-Ojalvo J, Yoshioka-Kobayashi K, Kageyama R, Yamanaka Y, et al. Species-specific segmentation clock periods are due to differential biochemical reaction speeds. Science 2020;369:1450–5.
Diaz-Cuadros M, Wagner DE, Budjan C, Hubaud A, Tarazona OA, Donelly S, et al. In vitro characterization of the human segmentation clock. Nature 2020;580:113–8.
Marshall M. These cellular clocks help explain why elephants are bigger than mice. Nature 2021;592:682–4.
Jackson SJ, Andrews N, Ball D, Bellantuono I, Gray J, Hachoumi L, et al. Does age matter? The impact of rodent age on study outcomes. Lab Anim. 2017;51:160–9.
Voikar V, Gaburro S. Three pillars of automated home-cage phenotyping of mice: novel findings, refinement, and reproducibility based on literature and experience. Front Behav Neurosci. 2020;14:575434. https://doi.org/10.3389/fnbeh.2020.575434.
Lopez JP, Lücken MD, Brivio E, Karamihalev S, Kos A, De Donno C, et al. Ketamine exerts its sustained antidepressant effects via cell-type-specific regulation of Kcnq2. Neuron. 2022;110:2283–98.e9. https://doi.org/10.1016/j.neuron.2022.05.001.
Heisler JM, Morales J, Donegan JJ, Jett JD, Redus L, O’Connor JC. The attentional set shifting task: a measure of cognitive flexibility in mice. J Vis Exp. 2015:51944. https://doi.org/10.3791/51944.
Toth I, Neumann ID. Animal models of social avoidance and social fear. Cell Tissue Res. 2013;354:107–18.
Cathomas F, Hartmann MN, Seifritz E, Pryce CR, Kaiser S. The translational study of apathy-an ecological approach. Front Behav Neurosci. 2015;9:241. https://doi.org/10.3389/fnbeh.2015.00241.
Farooq RK, Tanti A, Ainouche S, Roger S, Belzung C, Camus V. A P2X7 receptor antagonist reverses behavioural alterations, microglial activation and neuroendocrine dysregulation in an unpredictable chronic mild stress (UCMS) model of depression in mice. Psychoneuroendocrinology 2018;97:120–30.
Nilsson SRO, Goodwin NL, Choong JJ, Hwang S, Wright HR, Norvilleet ZC, et al. Golden: Simple Behavioral Analysis (SimBA) – an open source toolkit for computer classification of complex social behaviors in experimental animals. bioRxiv 2020. 2020.04.19.049452. https://doi.org/10.1101/2020.04.19.049452.
Forkosh O, Karamihalev S, Roeh S, Alon U, Anpilov S, Touma C, et al. Identity domains capture individual differences from across the behavioral repertoire. Nat Neurosci. 2019;22:2023–8.
Nguyen T-D, Harder A, Xiong Y, Kowalec K, Hägg S, Cai N, et al. Genetic heterogeneity and subtypes of major depression. 2021. https://www.medrxiv.org/content/10.1101/2021.03.05.21252911v2.
Price RB, Duman R. Neuroplasticity in cognitive and psychological mechanisms of depression: an integrative model. Mol Psychiatry. 2020;25:530–43.
Pittenger C, Duman RS. Stress, depression, and neuroplasticity: a convergence of mechanisms. Neuropsychopharmacology 2008;33:88–109.
Wojtowicz JM, Kee N. BrdU assay for neurogenesis in rodents. Nat Protoc. 2006;1:1399–405.
Miller AH, Raison CL. The role of inflammation in depression: from evolutionary imperative to modern treatment target. Nat Rev Immunol. 2016;16:22–34.
Sloman L. A new comprehensive evolutionary model of depression and anxiety. J Affect Disord. 2008;106:219–28.
Buckner JD, Joiner TE, Pettit JW, Lewinsohn PM, Schmidt NB. Implications of the DSM’s emphasis on sadness and anhedonia in major depressive disorder. Psychiatry Res. 2008;159:25–30.
Zych AD, Gogolla N. Expressions of emotions across species. Curr Opin Neurobiol. 2021;68:57–66.
Wang Q, Timberlake MA 2nd, Prall K, Dwivedi Y. The recent progress in animal models of depression. Prog Neuropsychopharmacol Biol Psychiatry. 2017;77:99–109.
Elmer T, Stadtfeld C. Depressive symptoms are associated with social isolation in face-to-face interaction networks. Sci Rep. 2020;10:1444.
Halsey LG. The reign of the p-value is over: what alternative analyses could we employ to fill the power vacuum? Biol Lett. 2019;15:20190174.
Lovell DP. Biological importance and statistical significance. J Agric Food Chem. 2013;61:8340–8.
Goodman WM, Spruill SE, Komaroff E. A proposed hybrid effect size plus p-value criterion: empirical evidence supporting its use. Am Statistician. 2019;73:168–85.
Grabowski T, Tomczyk A, Wolc A, Cox G. Shayne between biological relevancy and statistical significance - step for assessment harmonization. Am J Biomed Sci Res. 2021;13:514–22.
Guilloux JP, Seney M, Edgar N, Sibille E. Integrated behavioral z-scoring increases the sensitivity and reliability of behavioral phenotyping in mice: relevance to emotionality and sex. J Neurosci Methods. 2011;197:21–31.
Labots M, Laarakker MC, Schetters D, Arndt SS, van Lith HA. An improved procedure for integrated behavioral z-scoring illustrated with modified Hole Board behavior of male inbred laboratory mice. J Neurosci Methods. 2018;293:375–88.
Taborsky B, English S, Fawcett TW, Kuijper B, Leimar O, McNamara JM, et al. Towards an evolutionary theory of stress responses. Trends Ecol Evol. 2021;36:39–48.
Kiryk A, Janusz A, Zglinicki B, Turkes E, Knapska E, Konopka W, et al. IntelliCage as a tool for measuring mouse behavior – 20 years perspective. Behav Brain Res. 2020;388:112620.
Volkmann P, Stephan M, Krackow S, Jensen N, Rossner MJ. PsyCoP - a platform for systematic semi-automated behavioral and cognitive profiling reveals gene and environment dependent impairments of Tcf4 transgenic mice subjected to social defeat. Front Behav Neurosci. 2020;14:618180.
Dolensek N, Gehrlach DA, Klein AS, Gogolla N. Facial expressions of emotion states and their neuronal correlates in mice. Science 2020;368:89–94.
Acknowledgements
IVM receives funding from the Else-Kroener-Fresenius Foundation. IVM and CR receive minor funding from the International Max Planck Research School for Translational Psychiatry. LU received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 766124. JB receives funding from the “Kids2Health” grant of the Federal Ministry of Education and Research (01GL1743C). CR receives a sponsorship from the Gunter Sachs Donation. MVS receives funding by the “OptiMD” grant of the Federal Ministry of Education and Research (01EE1401D), the BioM M4 award “PROCERA” of the Bavarian State Ministry, and by the “Kids2Health” grant of the Federal Ministry of Education and Research (01GL1743C). JMD receives funding from the German Federal Ministry of Education and Research (IMADAPT, FKZ: 01KU1901) and from the Marie Skłodowska-Curie innovative training network PurinesDX. The authors thank Jessica Keverne for her help with language editing of this manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
IVM reviewed the scientific literature, developed the hypotheses, wrote the manuscript, and created the table and graphics. LU contributed to the revision and structuring of the manuscript and helped to improve the graphics. JB aided manuscript revision and structuring and added valuable aspects concerning the statistical aspects. CR contributed valuable methodological aspects and assisted in the revision of the manuscript. MS reviewed the manuscript several times and provided essential scientific advisement, guidance, and support. JMD reviewed and edited the manuscript and provided scientific advice and support.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
von Mücke-Heim, IA., Urbina-Treviño, L., Bordes, J. et al. Introducing a depression-like syndrome for translational neuropsychiatry: a plea for taxonomical validity and improved comparability between humans and mice. Mol Psychiatry 28, 329–340 (2023). https://doi.org/10.1038/s41380-022-01762-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-022-01762-w
This article is cited by
-
Ketamine’s rapid and sustained antidepressant effects are driven by distinct mechanisms
Cellular and Molecular Life Sciences (2024)
-
Automatically annotated motion tracking identifies a distinct social behavioral profile following chronic social defeat stress
Nature Communications (2023)
-
Systematic evaluation of a predator stress model of depression in mice using a hierarchical 3D-motion learning framework
Translational Psychiatry (2023)
-
Linking neuropsychiatric disease to neuronal metabolism
Nature Metabolism (2023)