Abstract
Major depressive disorder (MDD), bipolar disorder (BD), and schizophrenia spectrum disorder (SSD, schizophrenia, and schizoaffective disorder) overlap in symptomatology, risk factors, genetics, and other biological measures. Based on previous findings, it remains unclear what transdiagnostic regional gray matter volume (GMV) alterations exist across these disorders, and with which factors they are associated. GMV (3-T magnetic resonance imaging) was compared between healthy controls (HC; n = 110), DSM-IV-TR diagnosed MDD (n = 110), BD (n = 110), and SSD patients (n = 110), matched for age and sex. We applied a conjunction analysis to identify shared GMV alterations across the disorders. To identify potential origins of identified GMV clusters, we associated them with early and current risk and protective factors, psychopathology, and neuropsychology, applying multiple regression models. Common to all diagnoses (vs. HC), we identified GMV reductions in the left hippocampus. This cluster was associated with the neuropsychology factor working memory/executive functioning, stressful life events, and with global assessment of functioning. Differential effects between groups were present in the left and right frontal operculae and left insula, with volume variances across groups highly overlapping. Our study is the first with a large, matched, transdiagnostic sample to yield shared GMV alterations in the left hippocampus across major mental disorders. The hippocampus is a major network hub, orchestrating a range of mental functions. Our findings underscore the need for a novel stratification of mental disorders, other than categorical diagnoses.
Similar content being viewed by others
Introduction
Given that mental disorders such as major depressive disorder (MDD), bipolar disorder (BD), and schizophrenia spectrum disorder (SSD) exhibit high overlap in many domains (e.g., psychopathology, genetics, neuropsychology, regional brain volume alterations), the research question has emerged whether they are characterized by more communalities rather than differences [1,2,3,4,5,6]. In the domain of brain morphometry, previous work has attempted, but failed, to find a unique neural correlate (i.e., biomarker) specific to one such disorder [2].
Gray matter volume (GMV) changes have been described in single case vs. control studies: In MDD, GMV reductions were reported in the subgenual cingulate cortex, hippocampus, amygdala, thalamus, fusiform gyrus and putamen [7,8,9]. In BD, reductions were found in the anterior cingulate cortex (ACC), superior temporal gyrus, and calcarine cortices [10]. In schizophrenia (SZ; and schizoaffective disorder (SZA), referred to as SSD), reductions were reported in the frontal opercula, insula, superior temporal, mid cingulate, and calcarine cortices, cerebellum, hippocampus, amygdala, and basal ganglia [10, 11]. However, within these diagnostic categories, studies have produced heterogeneous results, which have been attributed to differences in course of illness, medication, age of onset, risk factors, genetics, psychopathology, and comorbidities, among others [7, 9, 12,13,14,15].
To disentangle these heterogeneous intra- and interdiagnostic findings, a landmark meta-analysis across six mental disorders pooled N = 193 original neuroimaging studies with N = 15,892 subjects [2]. Here, transdiagnostic GMV reductions in all patients vs. controls were identified in the bilateral insulae, right thalamus, left subgenual ACC, right dorsal and ventral ACC/mPFC, left amygdala, and left hippocampus. This study included a wide range of psychiatric disorders: MDD, BD, SZ, addiction, anxiety, and obsessive-compulsive disorder.
To answer the question which GMV alterations specifically overlap and are distinct in MDD, BD, and SSD –– that is, disorders of the affective and psychotic spectrum, a direct comparison of large, matched patient groups is necessary.
Thus far, only one study directly compared GMV brain structural alterations in MDD, BD, and SZ [16]. The authors found that almost 88% of GMV reductions overlapped in all three diagnostic groups, including regions in the temporal pole, orbitofrontal cortex (OFC), insula, hippocampus, cingulate and angular gyri. However, this study only included adolescents and young adults (aged 13–30), not matched for age and sex [16].
Since there is first evidence for significant GMV overlap across affective and psychotic disorders [2], it raises the question whether there are common, underlying factors that are responsible for these transdiagnostic findings. From previous structural brain imaging research, we know that the following four domains, among others, have been associated with brain structure in patients and healthy controls (HC): (1) early risk and protective factors: childhood maltreatment, urbanicity, familial risk, gestational age, birth weight, and parental bonding [12, 17,18,19,20], (2) current risk and protective factors: life events, resilience, and social support [21,22,23], (3) psychopathology: global functioning, positive, negative, depressive, and manic symptoms [3, 24], and (4) neuropsychological performance [25]. To the best of our knowledge, it has not been investigated whether transdiagnostic morphometric alterations are related to transdiagnostic phenotypical or risk patterns. However, such investigations are crucial as they could shed light on potential transdiagnostic pathomechanisms or indicate starting points for prevention.
In summary, previous studies on GMV alterations across MDD, BD and SSD have several shortcomings: (1) only comparing two diagnostic groups, (2) small sample sizes, (3) not matching groups for age and sex, and (4) not relating transdiagnostic brain alterations to phenotypical or risk factors. To overcome these issues, we directly compared three large age and sex-matched diagnostic groups (MDD, BD, SSD) to HC, drawn from a cohort of n = 1927, to identify shared and distinct GMV alterations. Further, we associated morphometric alterations with phenotypic and risk factors, i.e., early risk and protective factors, current risk and protective factors, psychopathology, and neuropsychological performance.
Methods
Participants
For the current study, from a cohort of n = 1927, we selected an age and sex-matched sample of N = 440 with n = 110 acute, chronic, and remitted participants per group of MDD, BD, SSD, and HC (matched using the MatchIt package [26] in R [27]) (see Table 1 for descriptive statistics). As the SSD group was the smallest (n = 110), MDD and BD patients and HCs were matched 1:1 to the age and sex-distribution of the SSD group. Participants aged 18–65 were drawn from the Marburg-Münster Affective Disorder Cohort Study (MACS). MACS is a longitudinal bi-center cohort study, and part of FOR2107, a consortium investigating the neurobiology of major psychiatric disorders [28].
Participants were recruited from in and out-patient departments of the universities of Marburg and Münster, Germany, local psychiatric hospitals (Vitos Marburg, Gießen, Herborn, and Haina, LWL Münster, Germany), and via postings in local newspapers and flyers. During a semi-structured interview, the German version of the structured clinical interview (SCID-I) for the DSM-IV-TR was applied by trained staff [29]. Furthermore, within one week of MRI scanning, psychopathological scales, a large questionnaire battery, a neuropsychological test battery and other rater-based scales were applied (see below). Moreover, clinical variables such as age of onset and number of hospitalizations were assessed during the semi-structured interview and, if available, based on patient records.
Exclusion criteria were a history of neurological or general medical conditions, current or lifetime alcohol dependency, current substance dependency, or current use of benzodiazepines (all assessed during the semi-structured interview performed by trained personnel and via self-report questionnaires), and verbal IQ ≤ 80 (assessed using [30]). Further exclusion criteria for the HC group were current or past mental disorders according to DSM-IV-TR, and lifetime intake of psychotropic medication. The study protocols were approved by the local Ethics Committees of Marburg and Münster, Germany according to the Declaration of Helsinki. All participants gave written informed consent before participation and received financial compensation.
Transdiagnostic phenotypical domains
To associate GMV with phenotype and risk factors, four domains were investigated: (1) early risk and protective factors, (2) current risk and protective factors, (3) psychopathology, (4) neuropsychological performance.
Early risk and protective factors
Childhood maltreatment was assessed using the childhood trauma questionnaire (CTQ) [31], urbanicity was assessed using a score for urban upbringing [32]. Familial risk was assessed via a questionnaire, asking whether a first-degree relative had been diagnosed and/or treated for MDD, BD, or SSD. Gestational age and birth weight were assessed through a questionnaire. Maternal and paternal care as an early protective factor was assessed with the German version of the parental bonding instrument (PBI) [33, 34].
Current risk and protective factors
Stressful life events (during the last six months) and their subjective impact were assessed using the life event questionnaire (LEQ) [35]. Current protective factors such as social support and resilience were assessed using the social support questionnaire (FSozu) [36] and the RS-25 resilience questionnaire, respectively [37].
Psychopathology
Psychopathology was assessed using the Scale for the Assessment of Negative Symptoms (SANS) [38], the Scale for the Assessment of Positive Symptoms (SAPS) [39], the Hamilton Depression Scale (HAM-D) [40], the Hamilton Anxiety Scale (HAMA) [41], and the Young Mania Rating Scale (YMRS) [42]. Further, the Global Assessment of Functioning (GAF) score [43, 44], was applied. All scales were rater-based. Raters were trained in the evaluation of psychopathological symptoms. Interrater reliabilities (ICC) were >0.86 for all scales.
Neuropsychological performance
A comprehensive neuropsychological test battery was applied including the d2 attention test [45], verbal fluency [46], symbol-coding [47], spatial span [48], letter-number span [49], Trail-Making Test A and B (TMT) [50], and the German Verbal Learning and Memory Test (VLMT) [51].
MRI data acquisition and preprocessing
MRI data acquisition and preprocessing was performed according to standardized procedures and using default parameters implemented in the respective toolboxes. At both sites, a 3-T MRI scanner (Marburg: Tim Trio, Siemens, Germany; Münster: Prisma, Siemens, Germany) was used to acquire T1 weighted images using a fast gradient echo MP-RAGE sequence with a slice thickness of 1.0 mm and a field of view of 256 mm. In Marburg, a 12-channel head matrix Rx-coil was used, in Münster, a 20-channel head matrix Rx-coil was used. Parameters differed across sites: Marburg: 176 sagittal slices, time of repetition (TR) = 1.9 s, time of echo (TE) = 2.26 ms, inversion time (TI) = 900 ms, flip angle = 9°; Münster: 192 sagittal slices, TR = 2.13 s, TE = 2.28 ms, TI = 900 ms, flip angle = 8°. MRI data were acquired according to an extensive quality assurance protocol [52].
A senior clinician visually inspected all scans regarding artifacts and anatomical abnormalities before preprocessing. Structural MRI data were preprocessed with the CAT12-Toolbox (Computational Anatomy Toolbox for SPM, build 1720, Structural Brain Mapping group, Jena University Hospital, Germany) (http://dbm.neuro.uni-jena.de/cat/) building on SPM12 (Statistical Parametric Mapping, Institute of Neurology, London, UK) using default parameters. In short, preprocessing included image segmentation into gray matter, white matter, and cerebrospinal fluid [53], spatial registration, and normalization [54]. Data were normalized to Montreal Neurological Institute (MNI) space. A more detailed description of our quality protocol can be found elsewhere [52]. During preprocessing, total intracranial volume (TIV) was calculated. MRI data sets were spatially smoothed with a Gaussian kernel of 8 mm full width half maximum.
Statistical analyses
Factor analysis of neuropsychological tests
Using SPSS 27, (Statistical Package for Social Science, IBM) an explorative principal axis factor analysis with varimax rotation was performed to identify aggregated domains of cognitive functioning (see Supplement). As neuropsychological test variables were differentially scaled, z-transformed values were used for the factor analysis. Suitability of neuropsychological test data for factor analysis was tested using Bartlett’s test of sphericity [55] and the Kaiser–Meyer–Olkin test (KMO) [56]. Factors were extracted according to the Kaiser’s eigenvalue greater than one criterion [57]. Factor loadings were extracted using the regression method as implemented in SPSS.
Brain structural analysis
To identify differences across the four groups (HC, MDD, BD, SSD), smoothed GMVs were applied using a whole brain 1×4 design in SPM (v6906) running under Matlab (R2017a). Post-hoc tests between two respective groups were performed to investigate significant differences. To identify commonly altered areas across the three diagnoses vs. healthy participants, we performed a conjunction analysis. Conjunction analysis allows the identification of overlap in GMV alterations in single diagnosis vs. control comparisons. The conjunction analysis used here was defined as follows: HC > MDD ∩ HC > BD ∩ HC > SSD. As conjunction analysis is known to be conservative, we opted for a region of interest (ROI)-based approach which included previously identified ROIs reported in the meta-analysis [2] in MDD, BD, and SSD patients by Goodkind et al. (2015) [2]. These ROIs were the bilateral insulae, thalamus, ACC, left amygdala, and left hippocampus. Using the Dartel space neuromorphometrics atlas in CAT12, we created one mask that included these anatomical ROIs. We have also included an exploratory whole-brain conjunction analysis in the Supplement.
The covariates age, sex, and total intracranial volume (TIV) were used in all analyses. MRI data were acquired at two sites. As recommended by the MRI quality assurance protocol of the FOR2107 cohort, we used two dummy-coded variables accounting for the change of a body coil and site (Marburg pre body coil: yes/no, Marburg post body coil: yes/no, and Münster as reference category [52]). Following the CAT12 recommendations (http://dbm.neuro.uni-jena.de/cat/), threshold masking with a value of 0.1 was applied for all analyses to exclude non-brain areas. Results were considered significant at p < 0.05 cluster-level family wise error-corrected (FWE) for multiple comparisons after an initial threshold of p < 0.001 uncorrected, with a cluster extend threshold of k > 10. Significant clusters were labelled using the Dartel space Neuromorphometrics atlas (http://www.neuromorphometrics.com/).
Transdiagnostic phenotypical factors associated with GMV
To answer the question which factors were associated with transdiagnostic morphometric findings, we associated the identified conjunction cluster with four different domains. These were (1) early risk and protective factors, (2) current risk and protective factors, (3) psychopathology and (4) neuropsychology (see above). To this end, we extracted eigenvariate values (weighted means) as an approximation of mean value from the conjunction cluster. Using SPSS 27, we ran four separate multiple regression models. We decided against one combined model, as this would have resulted in a largely reduced sample size. Using four models allowed us to minimize sample size loss without having to impute values. In all models, age, sex, TIV, two site/bodycoil variables, and a group variable were used as covariates. Multicollinearity in these variables was absent.
The first model, early risk and protective factors, included childhood maltreatment, urbanicity, parental bonding, gestational age, birth weight, and familial risk. The second model, current risk and protective factors, included current life events, resilience, and social support. The third model, psychopathology, was comprised of global functioning, positive, negative, depressive, and manic symptomatology. The fourth model, neuropsychological performance, included three factors identified in the factor analysis described in the Supplement.
Results
Our analyses were subdivided into four steps: (1) investigation of global effects of GMV (F-Test), (2) investigation of post-hoc two-group comparisons of GMV (t-Tests), (3) conjunction analysis of HC > MDD ∩ HC > BD ∩ HC > SSD, and (4) association of the conjunction cluster with phenotypical domains. Additional analyses controlling for the effect of medication and comorbidity showed that results remained stable. These analyses can be found in the Supplement.
To rule out that group differences were driven by HCs, we re-ran all analyses including n = 220 more HCs, resulting in a total sample of N = 660. These analyses revealed a similar pattern to the original analyses and can be found in the Supplement.
Global effects of GMV (F-Test)
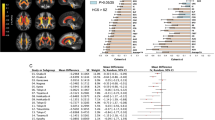
For the four groups (HC, MDD, BD, SSD), three significant clusters emerged in the F-Test. The first cluster comprised parts of the left fusiform gyrus, hippocampus, parahippocampal, inferior medial temporal, and lingual gyri (k = 1897, x/y/z = −37.5/−33/−22.5), F = 13.85, p = 0.0003 FWE cluster-level; k = 69, p = 0.0004 FWE peak-level). The second cluster comprised the right central operculum, superior temporal gyrus, temporal transverse gyrus, planum polare, and the precentral gyrus (k = 2268, x/y/z = 48/−12/7.5), F = 10.08, p = 0.00009 FWE cluster-level). The third cluster comprised parts of the left central and frontal operculum, anterior insula, planum polare, and temporal pole (k = 869, x/y/z = −46.5/4.5/−1.5), F = 9.21, p = 0.0192 FWE cluster-level) (cf. Fig. 1). To exclude the potential influence of duration and severity of illness on the identified clusters, we investigated the association between each cluster with number of hospitalizations, duration of hospitalization, duration in manic/depressive/psychotic state, and age of onset. There was no significant association between any of these factors and any cluster (all ps > 0.05), highlighting that the identified clusters were independent of duration and severity of illness.
Investigation of post-hoc two-group comparisons of GMV (t-Tests)
Next, we investigated which two-group comparisons were driving the clusters of the F-Test. Here, several significant areas (FWE cluster-level corrected) emerged for specific patient–HC comparisons, and within patient group comparisons. Table 2 lists all clusters, and Figs. 2 and 3 visualize these significant clusters in patient–HC comparisons and within patient comparisons, respectively.
GMV differences were present for MDD > SSD (orange) and BD > SSD (green); for details see Table 2.
Common areas of GMV alterations across MDD, BD, and SZ
ROI analyses
To investigate overlapping GMV reductions in all patients compared to HC, we ran a conjunction analysis (HC > MDD ∩ HC > BD ∩ HC > SSD) using the ROIs from the meta-analysis by Goodkind et al., (2015). This analysis revealed GMV reductions in the patient groups in the left hippocampus (k = 316, x/y/z = −30/−33/−10.5), T = 4.25, p = 0.042 FWE cluster-level; k = 316, p = 0.01 FWE peak-level) (see Fig. 4).
Whole-brain analyses
Results of the exploratory whole-brain conjunction analysis can be found in Supplement 3, revealing a similar pattern.
Association of conjunction cluster with phenotypical domains
Lastly, we associated the conjunction cluster in the left hippocampus with four phenotypic and risk domains using multiple regressions.
Early risk and protective factors
The first model included early risk and protective factors previously associated with GMV in mental disorders. The model significantly predicted hippocampal volume, F(12, 190) = 12.89, p < 0.001, R2 = 0.78, (adj. R2 = 0.57). Only the covariates age (β = −0.141, p = 0.004), TIV (β = 0.68, p < 0.001) and the group covariate (β = −0.225, p < 0.001) were significant predictors. Early risk and protective factors (childhood maltreatment, urbanicity, birth weight, gestation age, familial risk, parental bonding, site) were no significant predictors for hippocampal GMV (all ps > 0.05).
Current risk and protective factors
The second model included current risk and protective factors associated with GMV in mental disorders. The model significantly predicted hippocampal volume, F(9, 413) = 60.66, p < 0.001, R2 = 0.57, (adj. R2 = 0.56). LEQ score was a significant predictor (β = −0.071, p = 0.035), along with the covariates TIV (β = 0.673, p < 0.001), age (β = −0.172, p < 0.001), and group (β = −0.201, p < 0.001). Resilience, social support, sex, and the site variables were not significant (all ps > 0.05).
Psychopathology
The third model included positive, negative, depressive, anxious, and manic symptomatology assessed with the sum scores of several psychopathological scales. The total model significantly predicted hippocampal volume, F(12, 427) = 46.11, p < 0.001, R2 = 0.56, (adj. R2 = 0.55). GAF was a significant predictor for hippocampal volume (β = 0.142, p = 0.017). None of the other psychopathology variables, i.e., positive, negative, depressive, anxiety, and manic symptoms (SANS, SAPS, HAMD, HAMA, YMRS), and neither the site variables, nor sex were significant predictors of hippocampal volume (all ps > 0.05). The covariates group (β = −0.111, p = 0.027), TIV (β = 0.665, p < 0.001), and age (β = −0.162, p < 0.001) significantly predicted hippocampal GMV.
Neuropsychological performance
Using exploratory factor analysis, we identified three factors of neuropsychological performance: F1: working memory/executive functioning, F2: verbal fluency, and F3: verbal episodic memory. A detailed description can be found in Supplement 1.
The model including the neuropsychological domain significantly predicted hippocampus volume F(9, 404) = 56.58, p < 0.001, R2 = 0.56, (adj. R2 = 0.55). The neuropsychological performance factor working memory/executive functioning was a significant predictor (β = 0.10, p = 0.009), together with the covariates: group (β = −0.20, p < 0.001), TIV (β = 0.67, p < 0.001), and age (β = −0.117, p = 0.001). The variables for site, sex, verbal fluency, and verbal episodic memory were no significant predictors (all ps > 0.05).
In sum, the transdiagnostic left hippocampal GMV alteration was significantly negatively associated with stressful life events and positively with global functioning and working memory/executive functioning, independent of the presence of a psychiatric disorder.
Discussion
The central goal of this study was the identification and characterization of transdiagnostic GMV alterations common to MDD, BD, and SSD. We identified transdiagnostically reduced left hippocampal GMV, which was associated with stressful life events, global functioning, and executive functioning. We discuss these finding and further group comparisons below.
Group differences
Group differences between HC, MDD, BD, and SSD were detected in three main clusters: insula, superior temporal gyrus (STG), and hippocampus. Post-hoc group comparisons revealed significant GMV differences in the insula and STG only in SSD compared to HC. Further, SSD patients showed reduced GMV in the fusiform gyrus compared to HC, MDD, and BD.
Our findings align with previous research and expand them: The relevance of insula function in psychosis, especially with regard to aberrant information processing and integration, has been discussed extensively [2, 10, 58,59,60,61,62,63]. Reviewing findings of the STG in schizophrenia, Lu et al. (2009) report its involvement particularly in formal thought disorder and hallucinations [64].
GMV in the fusiform gyrus in SSD patients have also been documented and associated with reduced ability for facial recognition [65]. Our study further demonstrated that fusiform gyrus volume was reduced in SSD patients compared to MDD and BD patients, as well as HC, indicating a diagnosis specific effect. Recent studies have further associated fusiform gyrus volume with the paranoid-hallucinatory syndrome and the positive symptoms [3, 66].
Diagnoses-common GMV alterations
Hippocampal volume reductions have been documented in case control studies of MDD, BD, and SZ [7, 14, 67], and in meta-analyses [2, 68, 69], although with inconsistencies within single diagnoses. Hippocampal reductions in patients have been explained through heightened stress, leading to dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis and increased production of glucocorticoids, which have neurotoxic effects [70]. To explore this volume loss, we investigated associations with potential phenotypic and risk and protective factors. We found smaller left hippocampal volume to be associated with higher number of stressful life events, poorer neuropsychological executive and global (social) functioning (GAF), but notably not to other potential factors which have previously been associated with hippocampal volume, e.g., childhood maltreatment or psychopathology [71, 72]. Associations between hippocampal volume and childhood maltreatment have been heterogeneous, with some studies reporting a significant negative association between these factors [71, 73], whereas others did not [15]. A meta-analysis highlighted the importance of age, sex, and diagnosis, as they moderate this association [74].
The association between stressful life events and reduced hippocampal volume has been reported in a longitudinal study [75], and in the context of 9/11 [76]. We demonstrated that the number and impact of stressful life events was associated with reduced hippocampal volume in both patients and healthy subjects. We further demonstrated that GAF score and the neuropsychological performance factor working memory/executive functioning were associated with hippocampal volume. These findings align with results from case control studies, particularly in SZ, and less consistently in affective disorders [77,78,79,80,81].
While previous studies had linked verbal memory to hippocampal volume, our findings show associations with working memory/executive function, which has also been documented in several previous studies (e.g [80, 82,83,84]). However, our results go beyond these findings, as we demonstrated that these effects are not related to the traditional, descriptive diagnoses, but apply to all patients and healthy controls on a dimensional level. Reduced left hippocampal volume in all patients mirrors findings from molecular genetics [1, 85] and imaging studies [2, 16], highlighting that psychiatric disorders share large biological overlap across disorders.
Intriguingly, electroconvulsive therapy (ECT) has been identified as a potential intervention to increase hippocampal volume, global functioning, and quality of life in patients [86,87,88]. We believe these results emphasize the validity of hippocampal GMV as a transdiagnostic correlate of psychopathology that could be a target for intervention and prevention.
We provided a large, matched sample, making our findings more robust and generalizable compared to previous work [2, 16]. First, matched samples are adjusted for effects of confounding variables between groups [26]. Second, we provided data from patients aged 18–65, thus allowing for more generalizable results. Findings from another study (with patients aged 13–30) [16] might be skewed towards early-onset patients, who might represent a distinct subgroup, as median age of onset for MDD and BD is later in life (30–33 years) [89,90,91].
Limitations
Some limitations should be noted. First, we did not detect a significant whole-brain difference in the HC > BD contrast but detected a significant hippocampus finding in the conjunction analysis using ROI analysis. Meta- and mega-analyses have demonstrated that lithium medication increases volume in the hippocampus in BD patients [69, 92]. This factor seems to have impeded the detection of a whole-brain effect in BD vs. HC. In their meta-analysis, Hajek and colleagues (2012) [69] described this effect and were able to demonstrate that lithium merely masked the effect of actual hippocampal volume loss in BD patients. Second, while psychiatric medication is known to potentially affect GMV, the inclusion of all psychiatric medication as a covariate has inherent limitations in transdiagnostic samples. Cumulative lifetime intake of psychiatric medication, in particular antipsychotics, has been associated with GMV. However, it is difficult to reliably obtain this information, and we only had information on current medication available. Correcting for medication remains a challenge in transdiagnostic imaging studies: While it might be possible to only include medication naïve patients, this would in turn result in restricted representativeness. In the present study, we covered the full spectrum of patients including a broad age range, duration of illness, severity, and comorbidities. Most importantly, psychopharmacological medication class (antipsychotics, antidepressants, etc.) is highly intercorrelated with DSM diagnoses and illness severity and can therefore not entirely be eliminated in statistical models [93, 94]; inclusion of medication as a covariate would have drastically reduced variance to detect differences in group GMV, and we therefore decided not to include it in the main analyses, in line with previous work [2, 16]. We have, however, included the results of the analysis investigating the impact of medication (using Sackeim scores and chlorpromazine equivalents (CPZ)) in Supplement 2. These analyses reveal a very similar pattern as the main analyses. Third, there are further early risk factors from the epidemiological literature, such as paternal age, or cannabis use which were not included in this study. These factors have mainly been associated with SSD, but not affective disorders, so we did not include them [95]. Fourth, due to the cross-sectional design of this study, no definite inferences can be drawn about causality or directionality.
Conclusion
Smaller hippocampal GMV may constitute a shared neural pathomechanism in affective and psychotic disorders. We provide multimodal data to associate this finding with psychopathology, neuropsychology, and early and current risk and protective factors. Together with findings from other areas of research, such as genetics, immunology, early and late environmental risks, it emerges that there is an overlap across traditional phenomenological diagnoses, which should be addressed in research using novel approaches that might be better able to acknowledge shared features.
References
Anttila V, Bulik-Sullivan B, Finucane HK, Walters RK, Bras J, Duncan L, et al. Analysis of shared heritability in common disorders of the brain. Science. 2018;360.
Goodkind M, Eickhoff SB, Oathes DJ, Jiang Y, Chang A, Jones-Hagata LB, et al. Identification of a common neurobiological substrate for mental Illness. JAMA Psychiatry. 2015;72:305–15.
Stein F, Meller T, Brosch K, Schmitt S, Ringwald K, Pfarr JK, et al. Psychopathological syndromes across affective and psychotic disorders correlate with gray matter volumes. Schizophr Bull. 2021;47:1740–50.
Patel Y. Writing committee for the attention-deficit/hyperactivity disorder, autism spectrum disorder, bipolar disorder, major depressive disorder, obsessive-compulsive disorder, et al. virtual histology of cortical thickness and shared neurobiology in 6 psychiatric disorders. JAMA psychiatry. 2020;78:47–63.
Lalousis PA, Wood SJ, Schmaal L, Chisholm K, Griffiths SL, Reniers RLEP, et al. Heterogeneity and classification of recent onset psychosis and depression: a multimodal machine learning approach. Schizophr Bull. 2021;47:1130–40.
Romer AL, Knodt AR, Houts R, Brigidi BD, Moffitt TE, Caspi A, et al. Structural alterations within cerebellar circuitry are associated with general liability for common mental disorders. Mol Psychiatry. 2018;23:1084–90.
Gray JP, Müller VI, Eickhoff SB, Fox PT. Multimodal abnormalities of brain structure and function in major depressive disorder: a meta-analysis of neuroimaging studies. Am J Psychiatry. 2020;177:422–34.
Arnone D, Job D, Selvaraj S, Abe O, Amico F, Cheng Y, et al. Computational meta-analysis of statistical parametric maps in major depression. Hum Brain Mapp. 2016;37:1393–404.
Schmaal L, Veltman DJ, Van Erp TGM, Smann PG, Frodl T, Jahanshad N, et al. Subcortical brain alterations in major depressive disorder: findings from the ENIGMA Major Depressive Disorder working group. Mol Psychiatry. 2016;21:806–12.
Maggioni E, Crespo-Facorro B, Nenadic I, Benedetti F, Gaser C, Sauer H, et al. Common and distinct structural features of schizophrenia and bipolar disorder: The European Network on Psychosis, Affective disorders and Cognitive Trajectory (ENPACT) study. PLoS One. 2017;12:e0188000.
Van Erp TGM, Hibar DP, Rasmussen JM, Glahn DC, Pearlson GD, Andreassen OA, et al. Subcortical brain volume abnormalities in 2028 individuals with schizophrenia and 2540 healthy controls via the ENIGMA consortium. Mol Psychiatry. 2016;21:547–53.
Teicher MH, Samson JA. Childhood maltreatment and psychopathology: a case for ecophenotypic variants as clinically and neurobiologically distinct subtypes. Am J Psychiatry. 2013;170:1114–33.
Tozzi L, Garczarek L, Janowitz D, Stein DJ, Wittfeld K, Dobrowolny H, et al. Interactive impact of childhood maltreatment, depression, and age on cortical brain structure: Mega-analytic findings from a large multi-site cohort. Psychol Med. 2020;50:1020–31.
Hibar DP, Westlye LT, Doan NT, Jahanshad N, Cheung JW, Ching CRK, et al. Cortical abnormalities in bipolar disorder: an MRI analysis of 6503 individuals from the ENIGMA Bipolar Disorder Working Group. Mol Psychiatry. 2018;23:932–42.
Frodl T, Janowitz D, Schmaal L, Tozzi L, Dobrowolny H, Stein DJ, et al. Childhood adversity impacts on brain subcortical structures relevant to depression. J Psychiatr Res. 2017;86:58–65.
Chang M, Womer FY, Edmiston EK, Bai C, Zhou Q, Jiang X, et al. Neurobiological commonalities and distinctions among three major psychiatric diagnostic categories: a structural MRI study. Schizophr Bull. 2018;44:65–74.
Brosch K, Stein F, Meller T, Schmitt S, Yuksel D, Ringwald KG, et al. DLPFC volume is a neural correlate of resilience in healthy high-risk individuals with both childhood maltreatment and familial risk for depression. Psychol Med. 2021;1–7.
Lemola S, Oser N, Urfer-Maurer N, Brand S, Holsboer-Trachsler E, Bechtel N, et al. Effects of gestational age on brain volume and cognitive functions in generally healthy very preterm born children during school-age: A voxel-based morphometry study. PLoS One. 2017;12:e0183519.
Frissen A, van Os J, Peeters S, Gronenschild E, Marcelis M. Evidence that reduced gray matter volume in psychotic disorder is associated with exposure to environmental risk factors. Psychiatry Res - Neuroimaging. 2018;271:100–10.
Zhang X, Deng M, Ran G, Tang Q, Xu W, Ma Y, et al. Brain correlates of adult attachment style: a voxel-based morphometry study. Brain Res. 2018;1699:34–43.
Ringwald KG, Meller T, Schmitt S, Andlauer TFM, Stein F, Brosch K, et al. Interaction of developmental factors and ordinary stressful life events on brain structure in adults. NeuroImage Clin. 2021;30:102683.
Kahl M, Wagner G, de la Cruz F, Köhler S, Schultz CC. Resilience and cortical thickness: a MRI study. Eur Arch Psychiatry Clin Neurosci. 2018;0:0.
Che XW, Wei DT, Li WF, Li HJ, Qiao L, Qiu J, et al. The correlation between gray matter volume and perceived social support: a voxel-based morphometry study. Soc Neurosci. 2014;9.
Besteher B, Gaser C, Langbein K, Dietzek M, Sauer H, Nenadić I. Effects of subclinical depression, anxiety and somatization on brain structure in healthy subjects. J Affect Disord. 2017;215:111–7.
Vasic N, Walter H, Höse A, Wolf RC. Gray matter reduction associated with psychopathology and cognitive dysfunction in unipolar depression: A voxel-based morphometry study. J Affect Disord. 2008;109:107–16.
Ho DE, Imai K, King G, Stuart EA. Matching as nonparametric preprocessing for reducing model dependence in parametric causal inference. Polit Anal. 2007;15:199–236.
R Core Team (2020). R: A language and environment for statistical computing. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. 2020.
Kircher T, Wöhr M, Nenadic I, Schwarting R, Schratt G, Alferink J, et al. Neurobiology of the major psychoses: a translational perspective on brain structure and function-the FOR2107 consortium. Eur Arch Psychiatry Clin Neurosci. 2018;1:3.
Wittchen HU, Gruschwitz S, Wunderlich U, Zaudig M. Strukturiertes Klinisches Interview für DSM-IV (SKID-I). Achse I: Psychische Störungen. Göttingen: Hogrefe. 1997.
Lehrl S, Triebig G, Fischer B. Multiple choice vocabulary test MWT as a valid and short test to estimate premorbid intelligence. Acta Neurol Scand. 1995;91:335–45. May 1
Wingenfeld K, Spitzer C, Mensebach C, Grabe HJ, Hill A, Gast U, et al. The german version of the Childhood Trauma Questionnaire (CTQ):Preliminary psychometric properties. PPmP Psychother Psychosom Medizinische Psychol. 2010;60:442–50.
Lederbogen F, Kirsch P, Haddad L, Streit F, Tost H, Schuch P, et al. City living and urban upbringing affect neural social stress processing in humans. Nature 2011;474:498–501.
Lutz R, Heyn C, Kommer D. Fragebogen zur elterlichen Bindung-FEB. In: Lutz R, Mark N, editors. Wie gesund sind Kranke? Zur seelichen Gesundheit kranker. Göttingen: Verlag für angewandte Psychologie; 1995. p. 183–99.
Parker G, Tupling H, Brown LB. A Parental Bonding Instrument. Br J Med Psychol. 1979;52.
Norbeck JS. Modification of life event questionnaires for use with female respondents. Res Nurs Health. 1984;7:61–71.
Fydrich T, Sommer G, Braehler E. F-SOZU Fragebogen zur sozialen Unterstuetzung. Diagnostische Verfahren in der Psychotherapie. 2007.
Wagnild GM, Young HM. Development and psychometric evaluation of the resilience scale. J Nurs Meas. 1993;1:165–78.
Andreasen N. The scale for the assessment of negative symptoms (SANS). Iowa City: University of Iowa; 1983.
Andreasen N. The scale for the assessment of positive symptoms (SAPS). Iowa City: Universityof Iowa; 1984.
Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23:56–62.
Hamilton M. The assessment of anxiey states by rating. Br J Med Psychol. 1959;32:50–5.
Young RC, Biggs JT, Ziegler VE, Meyer DA. A rating scale for mania: reliability, validity and sensitivity. Br J Psychiatry. 1978;133:429–35.
Saß H, Wittchen H-U, Zaudig M, Houben I. Diagnostische Kriterien des Diagnostischen und Statistischen Manuals Psychischer Störungen. DSM-IV-TR. Göttingen: Hogrefe; 2003. 47–49 p.
American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 4th, Text ed. Washington, DC; 2000.
Brickenkamp R. Der Aufmerksamkeits-Belastungstest d2. Göttingen: Hogrefe; 2002.
Aschenbrenner A, Tucha O, Lange K. RWT. Regensburger Wortflüssigkeits-Test. Göttingen: Hogrefe; 2000.
Wechsler D, Coalson DL, Raiford SE. WAIS-IV: Wechsler adult intelligence scale. Pearson San Antonio, TX; 2008.
Wechsler F Wechsler Memory Scale: Administration and Scoring Manual. San Antonio, TX: Harcourt Brace & Co; 1997.
Gold JM, Carpenter C, Randolph C, Goldberg TE, Weinberger DR. Auditory working memory and Wisconsin Card Sorting Test performance in schizophrenia. Arch Gen Psychiatry. 1997;54:159–65.
Reitan RM Trail Making Test. Manual for administration and scoring. Tuscon, USA: Reitan Neuropsychology Laboratory; 1979.
Helmstaedter C, Lendt M, Lux S. Verbaler Lern- und Merkfähigkeitstest. Göttingen: Hogrefe; 2001.
Vogelbacher C, Möbius TWD, Sommer J, Schuster V, Dannlowski U, Kircher T, et al. The Marburg-Münster affective disorders cohort study (MACS): A quality assurance protocol for MR neuroimaging data. Neuroimage 2018;172:450–60.
Ashburner J, Friston KJ. Unified segmentation. Neuroimage 2005;26:839–51.
Ashburner J. A fast diffeomorphic image registration algorithm. Neuroimage 2007;38:95–113.
Bartlett MS. Properties of sufficiency and statistical tests. Proc R Soc Lond Ser A, Math Phys Sci. 1937;160:268–82.
Kaiser HF. An index of factorial simplicity. Psychometrika 1974;39:31–6.
Kaiser HF. The application of electronic computers to factor analysis. Educ Psychol Meas. 1960;20:141–51.
Sheffield JM, Huang AS, Rogers BP, Blackford JU, Heckers S, Woodward ND. Insula sub-regions across the psychosis spectrum: morphology and clinical correlates. Transl Psychiatry. 2021;11:1–13.
Stoyanov D, Aryutova K, Kandilarova S, Paunova R, Arabadzhiev Z, Todeva-Radneva A, et al. Diagnostic task specific activations in functional mri and aberrant connectivity of insula with middle frontal gyrus can inform the differential diagnosis of psychosis. Diagnostics. 2021;11:95.
Corradi-Dell’Acqua C, Tomelleri L, Bellani M, Rambaldelli G, Cerini R, Pozzi-Mucelli R, et al. Thalamic-insular dysconnectivity in schizophrenia: Evidence from structural equation modeling. Hum Brain Mapp. 2012;33:740–52.
Palaniyappan L, Simmonite M, White TP, Liddle EB, Liddle PF. Neural primacy of the salience processing system in schizophrenia. Neuron. 2013;79:814–28.
Wylie KP, Tregellas JR. The role of the insula in schizophrenia. Schizophr Res. 2010;123:93–104.
Shepherd AM, Matheson SL, Laurens KR, Carr VJ, Green MJ. Systematic meta-analysis of insula volume in schizophrenia. Biol Psychiatry. 2012;72:775–84.
Sun J, Maller JJ, Guo L, Fitzgerald PB. Superior temporal gyrus volume change in schizophrenia: a review on Region of Interest volumetric studies. Brain Res Rev. 2009;61:14–32.
Onitsuka T, Shenton ME, Kasai K, Nestor PG, Toner SK, Kikinis R, et al. Fusiform gyrus volume reduction and facial recognition in chronic schizophrenia. Arch Gen Psychiatry. 2003;60:349–55.
Padmanabhan JL, Tandon N, Haller CS, Mathew IT, Eack SM, Clementz BA, et al. Correlations between brain structure and symptom dimensions of psychosis in schizophrenia, schizoaffective, and psychotic bipolar i disorders. Schizophr Bull. 2015;41:154–62.
Schmaal L, Hibar DP, Sämann PG, Hall GB, Baune BT, Jahanshad N, et al. Cortical abnormalities in adults and adolescents with major depression based on brain scans from 20 cohorts worldwide in the ENIGMA Major Depressive Disorder Working Group. Mol Psychiatry. 2017;22:900–9.
McKinnon MC, Yucel K, Nazarov A, MacQueen GM. A meta-analysis examining clinical predictors of hippocampal volume in patients with major depressive disorder. J Psychiatry Neurosci. 2009;34:41–54.
Hajek T, Kopecek M, Höschl C, Alda M. Smaller hippocampal volumes in patients with bipolar disorder are masked by exposure to lithium: a meta-analysis. J Psychiatry Neurosci. 2012;37:333–43.
Frodl T, O’Keane V. How does the brain deal with cumulative stress? A review with focus on developmental stress, HPA axis function and hippocampal structure in humans. Neurobiol Dis. 2013;52:24–37.
Dannlowski U, Stuhrmann A, Beutelmann V, Zwanzger P, Lenzen T, Grotegerd D, et al. Limbic scars: Long-term consequences of childhood maltreatment revealed by functional and structural magnetic resonance imaging. Biol Psychiatry. 2012;71:286–93.
Besteher B, Squarcina L, Spalthoff R, Bellani M, Gaser C, Brambilla P, et al. Hippocampal volume as a putative marker of resilience or compensation to minor depressive symptoms in a nonclinical sample. Front Psychiatry. 2019;10:1–7.
Opel N, Redlich R, Zwanzger P, Grotegerd D, Arolt V, Heindel W, et al. Hippocampal atrophy in major depression: a function of childhood maltreatment rather than diagnosis. Neuropsychopharmacology 2014;39:2723–31.
Paquola C, Bennett MR, Lagopoulos J. Understanding heterogeneity in grey matter research of adults with childhood maltreatment—A meta-analysis and review. Neurosci Biobehav Rev. 2016;69:299–312.
Papagni SA, Benetti S, Arulanantham S, McCrory E, McGuire P, Mechelli A. Effects of stressful life events on human brain structure: a longitudinal voxel-based morphometry study. Stress. 2011;14:227–32.
Ganzel BL, Kim P, Glover GH, Temple E. Resilience after 9/11: Multimodal neuroimaging evidence for stress-related change in the healthy adult brain. Neuroimage. 2008;40:788–95.
Brambilla P, Perlini C, Rajagopalan P, Saharan P, Rambaldelli G, Bellani M, et al. Schizophrenia severity, social functioning and hippocampal neuroanatomy: Three-dimensional mapping study. Br J Psychiatry. 2013;202:50–5.
Orellana G, Slachevsky A. Executive functioning in schizophrenia. Front in Psychiatry. 2013;4:35.
Knöchel C, Stäblein M, Storchak H, Reinke B, Jurcoane A, Prvulovic D, et al. Multimodal assessments of the hippocampal formation in schizophrenia and bipolar disorder: Evidences from neurobehavioral measures and functional and structural MRI. NeuroImage Clin. 2014;6:134–44.
O’Brien JT, Lloyd A, McKeith I, Gholkar A, Ferrier N. A longitudinal study of hippocampal volume, cortisol levels, and cognition in older depressed subjects. Am J Psychiatry. 2004;161:2081–90.
Shi J, Guo H, Fan F, Fan H, An H, Wang Z, et al. Sex differences of hippocampal structure in bipolar disorder. Psychiatry Res - Neuroimaging. 2018;273:35–41.
Zhang W, Guo L, Liu D. Concurrent interactions between prefrontal cortex and hippocampus during a spatial working memory task. Brain Struct Funct. 2022;227:1735–55.
Huang J, Zhu Y, Fan F, Chen S, Hong Y, Cui Y, et al. Hippocampus and cognitive domain deficits in treatment-resistant schizophrenia: A comparison with matched treatment-responsive patients and healthy controls. Psychiatry Res - Neuroimaging. 2020;297:111043.
Shunkai L, Su T, Zhong S, Chen G, Zhang Y, Zhao H, et al. Abnormal dynamic functional connectivity of hippocampal subregions associated with working memory impairment in melancholic depression. Psychol Med. 2021.
Lee PH, Anttila V, Won H, Feng YCA, Rosenthal J, Zhu Z, et al. Genomic relationships, novel loci, and pleiotropic mechanisms across eight psychiatric disorders. Cell. 2019;179:1469–82.
Janouschek H, Camilleri JA, Peterson Z, Sharkey RJ, Eickhoff CR, Grözinger M, et al. Meta-analytic evidence for volume increases in the medial temporal lobe after electroconvulsive therapy. Biological Psychiatry. 2021;90:e11–7.
Camilleri JA, Hoffstaedter F, Zavorotny M, Zöllner R, Wolf RC, Thomann P, et al. Electroconvulsive therapy modulates grey matter increase in a hub of an affect processing network. NeuroImage Clin. 2020;25:102114.
Tor PC, Tan XW, Martin D, Loo C. Comparative outcomes in electroconvulsive therapy (ECT): A naturalistic comparison between outcomes in psychosis, mania, depression, psychotic depression and catatonia. Eur Neuropsychopharmacol. 2021;51:43–54.
Koutsouleris N, Meisenzahl EM, Borgwardt S, Riecher-Rössler A, Frodl T, Kambeitz J, et al. Individualized differential diagnosis of schizophrenia and mood disorders using neuroanatomical biomarkers. Brain 2015;138:2059–73.
Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62:593–602.
Solmi M, Radua J, Olivola M, Croce E, Soardo L, Salazar de Pablo G, et al. Age at onset of mental disorders worldwide: large-scale meta-analysis of 192 epidemiological studies. Mol Psychiatry. 2021;27:281–95.
Hallahan B, Newell J, Soares JC, Brambilla P, Strakowski SM, Fleck DE, et al. Structural magnetic resonance imaging in bipolar disorder: An international collaborative mega-analysis of individual adult patient data. Biol Psychiatry. 2011;69:326–35.
Fusar-Poli P, Smieskova R, Kempton MJ, Ho BC, Andreasen NC, Borgwardt S. Progressive brain changes in schizophrenia related to antipsychotic treatment? A meta-analysis of longitudinal MRI studies. Neurosci Biobehav Rev. 2013;37:1680–91.
Ho B-C, Andreasen NC, Ziebell S, Pierson R, Magnotta V. Long-term antipsychotic treatment and brain volumes a longitudinal study of first-episode schizophrenia. Arch Gen Psychiatry. 2011;68:128–37.
Vassos E, Sham P, Kempton M, Trotta A, Stilo SA, Gayer-Anderson C, et al. The Maudsley environmental risk score for psychosis. Psychol Med. 2019;50:2213–20.
Acknowledgements
We are deeply indebted to all study participants and staff. A list of acknowledgments can be found at: for2107.de/acknowledgements.
Funding
This work is part of the German multicenter consortium “Neurobiology of Affective Disorders. A translational perspective on brain structure and function “, funded by the German Research Foundation (Deutsche Forschungsgemeinschaft DFG; Forschungsgruppe/Research Unit FOR2107). Tilo Kircher (speaker FOR2107; DFG grant numbers KI 588/14-1, KI 588/14-2, KI 588/15-1, KI 588/17-1), Udo Dannlowski (co-speaker FOR2107; DA 1151/5-1, DA 1151/5-2, DA 1151/6-1), Axel Krug (KR 3822/5-1, KR 3822/7-2), Igor Nenadic (NE 2254/1-2), Tim Hahn (HA7070/2-2), Andreas Jansen (JA1890/7-1, JA1890/7-2). Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
KB & FS*: writing, analysis, data interpretation, literature search, figures, data collection *contributed equally, SS: data collection, review, JP: data collection, data curation, KR: data collection, review, FTO: data collection, review, TM: data collection, data curation, OS: data curation, data collection, LW: data collection, review, HL: data collection, review, SM: data collection, data curation, AW: data collection, data curation, review, FB: data collection, review, KT: data collection, review, DG: data collection, review, data curation, TH: data collection, funding acquisition, data curation, AJ: funding acquisition, data curation, study design, UD: funding acquisition, data collection, data curation, study design, AK: funding acquisition, data collection, data curation, study design, IN: funding acquisition, data collection, data curation, study design, data interpretation, TK: funding acquisition, data collection, data curation, data interpretation, study design, review. All authors have full access to all data, have contributed to the manuscript, and have approved the final manuscript.
Corresponding author
Ethics declarations
COMPETING INTERESTS
Biomedical financial interests or potential conflicts of interest: Tilo Kircher received unrestricted educational grants from Servier, Janssen, Recordati, Aristo, Otsuka, and neuraxpharm. All other authors declare no competing interests.
Ethical approval
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Brosch, K., Stein, F., Schmitt, S. et al. Reduced hippocampal gray matter volume is a common feature of patients with major depression, bipolar disorder, and schizophrenia spectrum disorders. Mol Psychiatry 27, 4234–4243 (2022). https://doi.org/10.1038/s41380-022-01687-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-022-01687-4
This article is cited by
-
Functional phenotypes in schizophrenia spectrum disorders: defining the constructs and identifying biopsychosocial correlates using data-driven methods
Schizophrenia (2024)
-
N-acetylcysteine during critical neurodevelopmental periods prevents behavioral and neurochemical deficits in the Poly I:C rat model of schizophrenia
Translational Psychiatry (2024)
-
Asymmetry, cytoarchitectonic morphology and genetics associated with Broca’s area in schizophrenia
Nature Mental Health (2024)
-
Factor analysis of lifetime psychopathology and its brain morphometric and genetic correlates in a transdiagnostic sample
Translational Psychiatry (2024)
-
Brain aging patterns in a large and diverse cohort of 49,482 individuals
Nature Medicine (2024)