Abstract
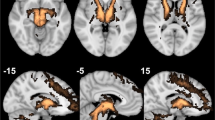
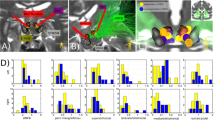
Deep brain stimulation (DBS) to the superolateral branch of the medial forebrain bundle (MFB) has emerged as a quite efficacious therapy for treatment resistant depression (TRD), leading to rapid antidepressant effects. In this study, we complete our assessment of our first 10 enrolled patients throughout one year post-implantation, showing sustained antidepressant effect up to 5 years. The primary outcome measure was a 50% reduction in Montgomery-Åsberg Depression Rating Scale (MADRS) score, which was interpreted as a response. Deterministic fiber tracking was used to individually map the target area. An insertional effect was seen during the 4-week sham stimulation phase (29% mean MADRS reduction, p = 0.02). However, after 2 weeks of initiating stimulation, five patients met response criteria (47% mean MADRS reduction, p < 0.001). One patient withdrew from study participation at 6 weeks. Twelve weeks after initiating stimulation, six of nine remaining patients had a >50% decrease in MADRS scores relative to baseline (52% mean MADRS reduction, p = 0.001); these same six patients continued to meet response criteria at 52 weeks (63% overall mean MADRS reduction, p < 0.001). Four of five patients who achieved the 5-year time point analysis continued to be responders (81% mean MADRS reduction, p < 0.001). Evaluation of modulated fiber tracts reveals significant common prefrontal/orbitofrontal connectivity to the target region in all responders. Key points learned from this study that we can incorporate in future protocols to better elucidate the effect of this therapy are a longer blinded sham stimulation phase and use of scheduled discontinuation concomitant with functional imaging.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Deuschl G, Elble R. Essential tremor-neurodegenerative or nondegenerative disease towards a working definition of ET. Mov Disord. 2009;24:2033–41.
Louis ED. Essential tremor. Lancet Neurol. 2005;4:100–10.
Louis ED. Clinical practice. Essential tremor. N Engl J Med. 2001;345:887–91.
Lozano AM, Mayberg HS, Giacobbe P, Hamani C, Craddock RC, Kennedy SH. Subcallosal cingulate gyrus deep brain stimulation for treatment-resistant depression. Biol Psychiatry. 2008;64:461–467.
Holtzheimer PE, Kelley ME, Gross RE, Filkowski MM, Garlow SJ, Barrocas A, et al. Subcallosal cingulate deep brain stimulation for treatment-resistant unipolar and bipolar depression. Arch Gen Psychiatry. 2012;69:150–158.
Mayberg HS, Lozano AM, Voon V, McNeely HE, Seminowicz D, Hamani C, et al. Deep brain stimulation for treatment-resistant depression. Neuron. 2005;45:651–60.
Holtzheimer PE, Husain MM, Lisanby SH, Taylor SF, Whitworth LA, McClintock S, et al. Subcallosal cingulate deep brain stimulation for treatment-resistant depression: a multisite, randomised, sham-controlled trial. Lancet Psychiatry. 2017;4:839–49.
Maloney DA Jr, Dougherty DD, Rezai AR, Carpenter LL, Friehs GM, Eskander EN, et al. Deep brain stimulation of the ventral capsule/ventral striatum for treatment-resistant depression. Biol Psychiatry. 2009;65:267–75.
Dougherty DD, Rezai AR, Carpenter LL, Howland RH, Bhati MT, O’Rearson JP, et al. A randomized sham-controlled trial of deep brain stimulation of the ventral capsule/ventral striatum for chronic treatment-resistant depression. Biol Psychiatry. 2015;78:240–8.
Bergfeld IO, Mantione M, Hoogendoorn MLC, Ruhe HG, Notten P, van Laarhoven J, et al. Deep brain stimulation of the ventral anterior limb of the internal capsule for treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2016;73:456–64.
Bewernick BH, Hurlemann R, Matusch A, Kayser S, Grubert C, Hadrysiewicz B, et al. Nucleus accumbens deep brain stimulation decreases ratings of depression and anxiety in treatment-resistant depression. Biol Psychiatry. 2010;67:110–6.
Schlaepfer TE, Bewernick BH, Kayser S, Madler B, Coenen VA. Rapid effects of deep brain stimulation for treatment-resistant major depression. Biol Psychiatry. 2013;73:1204–12.
Bewernick BH, Kayser S, Gippert SM, Switala C, Coenen VA, Schlaepfer TE. Deep brain stimulation to the medial forebrain bundle for depression- long-term outcomes and a novel data analysis strategy. Brain Stimul. 2017;10:664–71.
Coenen VA, Bewernick BH, Kayser S, Kilian H, Boström J, Greschus S, et al. Superolateral medial forebrain bundle deep brain stimulation in major depression: a gateway trial. Neuropsychopharmacology. 2019;44:1224–32.
Gálvez JF, Keser Z, Mwangi B, Ghouse A, Fenoy AJ, Schulz PE, et al. The medial forebrain bundle as a deep brain stimulation target for treatment resistant depression: a review of published data. Prog Neuropsychopharmacol Biol Psychiatry. 2015;58:59–70.
Nestler EJ, Carlezon WA Jr. The mesolimbic dopamine reward circuit in depression. Biol Psychiatry. 2006;59:1151–9.
Russo SJ, Nestler EJ. The brain reward circuitry in mood disorders. Nat Rev Neurosci. 2013;14:609–25.
Fenoy AJ, Schulz PE, Selvaraj S, Burrows CL, Spiker D, Cao B, et al. Deep brain stimulation of the medial forebrain bundle: distinctive responses in resistant depression. J Affect Disord. 2016;203:143–51.
Fenoy AJ, Schulz PE, Selvaraj S, Burrows CL, Zunta-Soares G, Durkin K, et al. A longitudinal study on deep brain stimulation of the medial forebrain bundle for treatment-resistant depression. Transl Psychiatry. 2018;8:111–8.
First MB, Spitzer RL, Gibbon M, Williams JBW. Structured clinical interview for DSM-IV axis I disorders (SCID I). American Psychiatric Press, Washington DC; 1997.
Hamilton M. Development of a rating scale for primary depressive illness. Br J Soc Clin Psychol. 1967;6:278–96.
Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134:382–9.
Jones SH, Thornicroft G, Coffey M, Dunn G. A brief mental health outcome scale-reliability and validity of the Global Assessment of Functioning (GAF). Br J Psychiatry. 1995;166:654–9.
First MB, Spitzer RL, Gibbon M, Williams JBW, Benjamin LS. Structured clinical interview for DSM-IV Axis II personality disorders (SCID II). American Psychiatric Press, Washington, DC; 1996.
Millon T, Millon C, David R. Millon Clinical Multiaxial Inventory-III (MCMI-III) manual. National Computer Systems, Minneapolis, MN; 1994.
Hamilton M. Hamilton Anxiety Scale. In: Guy W editor. ECDEU Assessment manual for psychopharmacology. RevEd, Rockville, MD; 1976. p. 193–8.
Young RC, Biggs JT, Ziegler VE, Meyer DA. A rating scale for mania: reliability, validity and sensitivity. Br J Psychiatry. 1978;133:429–35.
Busner J, Targum SD. The clinical global impressions scale: applying a research tool in clinical practice. Psychiatry. 2007;4:28–37.
Coenen VA, Panksepp J, Hurwitz TA, Urbach H, Mädler B. Human medial forebrain bundle (MFB) and anterior thalamic radiation (ATR): imaging of two major subcortical pathways and the dynamic balance of opposite affects in understanding depression. J Neuropsychiatry Clin Neurosci. 2012;24:223–36.
Fenoy AJ, Simpson RK Jr. Management of device-related wound com- plications in deep brain stimulation surgery. J Neurosurg. 2012;11:1324–32.
Fenoy AJ, Simpson RK Jr. Risks of common complications in deep brain stimulation surgery: management and avoidance. J Neurosurg. 2014;120:132–9.
Watson D, Clark LA, Tellegen A. Development and validation of brief measures of positive and negative affect: the PANAS Scales. J Pers Soc Psychol. 1988;54:1063–70.
Woolrich MW, Jbabdi S, Patenaude B, Chappell M, Makni S, Behrens T, et al. Bayesian analysis of neuroimaging data in FSL. Neuroimage. 2009;45:S173–S186. Suppl.
Smith SM. Fast robust automated brain extraction. Hum Brain Mapp. 2002;17:143–55.
Jenkinson M, Beckmann CF, Behrens TE, Wollrich MW, Smith SM. Fsl. Neuroimage. 2012;62:782–90.
Behrens TE, Berg HJ, Jbabdi S, Rushworth MF, Woolrich MW. Probabilistic diffusion tractography with multiple fibre orientations: What can we gain? Neuroimage. 2007;34:144–55.
Nowacki A, Schlaier J, Debove I, Pollo C. Validation of diffusion tensor imaging tractography to visualize the dentatorubothalamic tract for surgical planning. J Neurosurg. 2018;130:99–108.
Butson CR, Cooper SE, Henderson JM, McIntyre CC. Patient-specific analysis of the volume of tissue activated during deep brain stimulation. Neuroimage. 2007;34:661–70.
Perez-Caballero L, Perez-Egea R, Romero-Grimaldo C, Puigdemont D, Molet J, Caso J-R, et al. Early responses to deep brain stimulation in depression are modulated by anti-inflammatory drugs. Mol Psychiatry. 2014;19:607–14.
Chang SY, Shon YM, Agnesi F, Lee KH. Microthalamotomy effect during deep brain stimulation: potential involvement of adenosine and glutamate efflux. In: Proceedings of the conference of IEEE Engineering in Medicine and Biology Society. 2009; p. 3294–97.
Fenoy AJ, Goetz L, Chabardès S, Xia Y. Deep brain stimulation: are astrocytes a key driver behind the scene? CNS Neurosci Ther. 2014;20:191–201.
Riva-Posse P, Choi KS, Holtzheimer PE, McIntyre CC, Gross RE, Chaturvedi A, et al. Defining critical white matter pathways mediating successful subcallosal cingulate deep brain stimulation for treatment-resistant depression. Biol Psychiatry. 2014;76:963–9.
Rao VR, Sellers KK, Wallace DL, Lee MB, Bijanzadeh M, Sani OG, et al. Direct electrical stimulation of the lateral orbitofronral cortex acutely improves mood in individuals with symptoms of depression. Curr Biol. 2018;28:3893–902.
Coenen VA, Schlaepfer TE, Sajonz B, Döbrössy M, Kaller CP, Urbach H, et al. Tractographic description of major subcortical projection pathways passing the anterior limb of the internal capsule. Corticopetal organization of networks relevant for psychiatric disorders. Neuroimage Clin. 2020;25:102165.
Coenen VA, Schlaepfer TE, Maedler B, Panksepp J. Cross-species affective functions of the medial forebrain bundle-implications for the treatment of affective pain and depression in humans. Neurosci Biobehav Rev. 2011;35:1971–81.
Hamid AA, Pettibone JR, Mabrouk OS, Hetrick VL, Schmidt R, Vander Weele CM, et al. Mesolimbic dopamine signals the value of work. Nat Neurosci. 2016;19:117–26.
Ikemoto S. Brain reward circuitry beyond the mesolimbic dopamine system: a neurobiological theory. Neurosci Biobehav Rev. 2010;35:129–50.
Coenen VA, Madler B, Schlaepfer TE. Reply to: Medial Forebrain bundle stimulation—speed access to an old or entry into a new depression neurocircuit? Biol Psychol. 2013;74:e45–e46.
Castro DC, Berridge KC. Advances in the neurobiological bases for food “liking” versus “wanting.”. Physiol Behav. 2014;136:22–30.
Berridge KC, Kringelbach ML. Pleasure systems in the brain. Neuron. 2015;86:646–64.
Döbrössy MD, Furlanetti LL, Coenen VA. Electrical stimulation of the medial forebrain bundle in pre-clinical studies of psychiatric disorders. Neurosci Biobehav Rev. 2015;49:32–42.
Karreman M, Westerink BH, Moghaddam B. Excitatory amino acid receptors in the ventral tegmental area regulate dopamine release in the ventral striatum. J Neurochem. 1996;67:601–7.
Helbing C, Brocka M, Scherf T, Lippert MT, Angenstein F. The role of the mesolimbic dopamine system in the formation of blood-oxygen-level dependent responses in the medial prefrontal/anterior cingulate cortex during high-frequency stimulation of the rat perforant pathway. J Cereb Blood Flow Metab. 2016;36:2177–93.
Settell ML, Testini P, Cho S, Lee JH, Blaha CD, Jo HJ, et al. Functional circuitry effect of ventral tegmental area deep brain stimulation: imaging and neurochemical evidence of mesocortical and mesolimbic pathway modulation. Front Neurosci. 2017;11:104.
Schaltenbrand G, Wahren W. Atlas for stereotaxy of the human brain. 2nd ed. New York: Thieme; 1977.
Acknowledgements
The Center of Excellence on Mood Disorders is funded by the Pat Rutherford Jr. Chair in Psychiatry, John S. Dunn Foundation, and Anne and Don Fizer Foundation Endowment for Depression Research.
Author information
Authors and Affiliations
Contributions
AJF was responsible for preparation and editing of all drafts and figures of this manuscript; PES, MS, SS, CLB, BA, JQ, JCS contributed to editing of final manuscript draft; CRC contributed to final figure production.
Corresponding author
Ethics declarations
Competing interests
AJF serves as a consultant for Medtronic, Inc and receives grant support from the NIH/NINDS (1R01NS113893-01A1). PES is a consultant for Lilly, Acadia, and Biogen, has grant support from the Weston Brain Foundation, the Frontotemporal Disease association, and the Kleberg Foundation, and has NIH support on unrelated projects. SS has received grants/research support from NIH/NIMH (1R21MH119441-01A1) and SAMHSA (6H79FG000470-01M003); research support from Compass Pathways, LivaNova, Janssen; speaking honoraria from the British Medical Journal Publishing Group and copyright with Cambridge University Press. JQ receives research support from the NIH/NIMH (1R21MH117636-01A1), the Faillace Department of Psychiatry and Behavioral Sciences, and LivaNova; has speaker bureau membership with Myriad Neuroscience, Janssen Pharmaceuticals, and Abbvie; is consultant for Eurofarma; is stockholder at Instituto de Neurociencias Dr. Joao Quevedo; and receives copyrights from Artmed Editora, Artmed Panamericana, and Elsevier/Academic Press. JCS receives grant/research support from Bristol-Meyers Squibb, Forest Laboratories, Merck and Elan Pharmaceuticals, and serves as a consultant for Pfizer, Abbot, and Astellas Pharma, Inc. MS, CRC, and CLB reported no biomedical financial interests or potential conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Fenoy, A.J., Schulz, P.E., Sanches, M. et al. Deep brain stimulation of the “medial forebrain bundle”: sustained efficacy of antidepressant effect over years. Mol Psychiatry 27, 2546–2553 (2022). https://doi.org/10.1038/s41380-022-01504-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-022-01504-y
This article is cited by
-
Deep brain stimulation for refractory major depressive disorder: a comprehensive review
Molecular Psychiatry (2024)
-
Opportunities and challenges for the use of deep brain stimulation in the treatment of refractory major depression
Discover Mental Health (2024)
-
Discontinuation of deep brain stimulation to the medial forebrain bundle leads to depression relapse: considerations when reinstating stimulation
Molecular Psychiatry (2023)
-
Are we getting closer to offering deep brain stimulation for treatment-resistant depression in clinical practice?
Molecular Psychiatry (2023)
-
Brain metabolic changes and clinical response to superolateral medial forebrain bundle deep brain stimulation for treatment-resistant depression
Molecular Psychiatry (2022)