Abstract
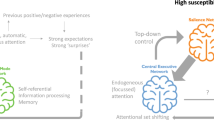
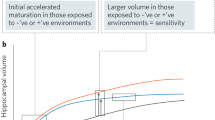
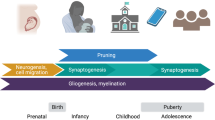
Given the high prevalence and burden of mental disorders, fostering the understanding of protective factors is an urgent issue for translational medicine in psychiatry. The concept of resilience describes individual and environmental protective factors against the backdrop of established adversities linked to mental illness. There is convergent evidence for a crucial role of direct as well as indirect adversity impacting the developing brain, with persisting effects until adulthood. Direct adversity may include childhood maltreatment and family adversity, while indirect social adversity can include factors such as urban living or ethnic minority status. Recently, research has begun to examine protective factors which may be able to buffer against or even reverse these influences. First evidence indicates that supportive social environments as well as trait-like individual protective characteristics might impact on similar neural substrates, thus strengthening the capacity to actively cope with stress exposure in order to counteract the detrimental effects evoked by social adversity. Here, we provide an overview of the current literature investigating the neural mechanisms of resilience with a putative social background, including studies on individual traits and genetic variation linked to resilience. We argue that the regulatory perigenual anterior cingulate cortex and limbic regions, including the amygdala and the ventral striatum, play a key role as crucial convergence sites of protective factors. Further, we discuss possible prevention and early intervention approaches targeting both the individual and the social environment to reduce the risk of psychiatric disorders and foster resilience.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Rutter M. Implications of resilience concepts for scientific understanding. Ann N Y Acad Sci. 2006;1094:1–12.
Rutter M. Resilience as a dynamic concept. Dev Psychopathol. 2012;24:335–44.
Kaufman JCA, Arny L, Jones B, Pittinsky T. Problems defining resiliency: Illustrations from the study of maltreated children. Dev Psychopathol. 1994;6:215–29.
Herrman H, Stewart DE, Diaz-Granados N, Berger EL, Jackson B, Yuen T. What is resilience? Can J Psychiatry. 2011;56:258–65.
Tost H, Champagne FA, Meyer-Lindenberg A. Environmental influence in the brain, human welfare and mental health. Nat Neurosci. 2015;18:1421–31.
Bickart KC, Brickhouse M, Negreira A, Sapolsky D, Barrett LF, Dickerson BC. Atrophy in distinct corticolimbic networks in frontotemporal dementia relates to social impairments measured using the Social Impairment Rating Scale. J Neurol Neurosurg Psychiatry. 2014;85:438–48.
Holz NE, Laucht M, Meyer-Lindenberg A. Recent advances in understanding the neurobiology of childhood socioeconomic disadvantage. Curr Opin Psychiatry. 2015;28:365–70.
Heim CM, Entringer S, Buss C. Translating basic research knowledge on the biological embedding of early-life stress into novel approaches for the developmental programming of lifelong health. Psychoneuroendocrinology. 2019;105:123–37.
Diorio D, Viau V, Meaney MJ. The role of the medial prefrontal cortex (cingulate gyrus) in the regulation of hypothalamic-pituitary-adrenal responses to stress. J Neurosci. 1993;13:3839–47.
Boehringer A, Tost H, Haddad L, Lederbogen F, Wust S, Schwarz E, et al. Neural correlates of the cortisol awakening response in humans. Neuropsychopharmacology. 2015;40:2278–85.
Lederbogen F, Kirsch P, Haddad L, Streit F, Tost H, Schuch P, et al. City living and urban upbringing affect neural social stress processing in humans. Nature. 2011;474:498–501.
Akdeniz C, Tost H, Streit F, Haddad L, Wüst S, Schäfer A, et al. Neuroimaging evidence for a role of neural social stress processing in ethnic minority–associated environmental risk. JAMA Psychiatry. 2014;71:672–80.
Selten JP, van der Ven E, Rutten BP, Cantor-Graae E. The social defeat hypothesis of schizophrenia: an update. Schizophr Bull. 2013;39:1180–6.
Haddad L, Schafer A, Streit F, Lederbogen F, Grimm O, Wust S, et al. Brain structure correlates of urban upbringing, an environmental risk factor for schizophrenia. Schizophr Bull. 2015;41:115–22.
Gianaros PJ, Horenstein JA, Cohen S, Matthews KA, Brown SM, Flory JD, et al. Perigenual anterior cingulate morphology covaries with perceived social standing. Soc Cogn Affect Neurosci. 2007;2:161–73.
van Harmelen AL, Hauber K, Gunther Moor B, Spinhoven P, Boon AE, Crone EA, et al. Childhood emotional maltreatment severity is associated with dorsal medial prefrontal cortex responsivity to social exclusion in young adults. PLoS One. 2014;9:e85107.
Rudolph KD, Miernicki ME, Troop-Gordon W, Davis MM, Telzer EH. Adding insult to injury: neural sensitivity to social exclusion is associated with internalizing symptoms in chronically peer-victimized girls. Soc Cogn Affect Neurosci. 2016;11:829–42.
Will GJ, van Lier PA, Crone EA, BG. Chronic childhood peer rejection is associated with heightened neural responses to social exclusion during adolescence. J Abnorm Child Psychol. 2016;44:43–55.
Ochsner KN, Ray RD, Cooper JC, Robertson ER, Chopra S, Gabrieli JD, et al. For better or for worse: neural systems supporting the cognitive down- and up-regulation of negative emotion. Neuroimage. 2004;23:483–99.
Luby J, Belden A, Botteron K, Marrus N, Harms MP, Babb C, et al. The effects of poverty on childhood brain development: the mediating effect of caregiving and stressful life events. JAMA Pediatr. 2013;167:1135–42.
Holz NE, Boecker R, Hohm E, Zohsel K, Buchmann AF, Blomeyer D, et al. The long-term impact of early life poverty on orbitofrontal cortex volume in adulthood: results from a prospective study over 25 years. Neuropsychopharmacology. 2015;40:996–1004.
Lawson GM, Duda JT, Avants BB, Wu J, Farah MJ. Associations between children’s socioeconomic status and prefrontal cortical thickness. Dev Sci. 2013;16:641–52.
Kim P, Evans GW, Angstadt M, Ho SS, Sripada CS, Swain JE, et al. Effects of childhood poverty and chronic stress on emotion regulatory brain function in adulthood. Proc Natl Acad Sci USA. 2013;110:18442–7.
Dannlowski U, Stuhrmann A, Beutelmann V, Zwanzger P, Lenzen T, Grotegerd D, et al. Limbic scars: long-term consequences of childhood maltreatment revealed by functional and structural magnetic resonance imaging. Biol Psychiatry. 2012;71:286–93.
Grant MM, Cannistraci C, Hollon SD, Gore J, Shelton R. Childhood trauma history differentiates amygdala response to sad faces within MDD. J Psychiatr Res. 2011;45:886–95.
Tottenham N, Hare TA, Millner A, Gilhooly T, Zevin JD, Casey BJ. Elevated amygdala response to faces following early deprivation. Dev Sci. 2011;14:190–204.
Maheu FS, Dozier M, Guyer AE, Mandell D, Peloso E, Poeth K, et al. A preliminary study of medial temporal lobe function in youths with a history of caregiver deprivation and emotional neglect. Cogn Affect Behav Neurosci. 2010;10:34–49.
Taylor SE, Eisenberger NI, Saxbe D, Lehman BJ, Lieberman MD. Neural responses to emotional stimuli are associated with childhood family stress. Biol Psychiatry. 2006;60:296–301.
Holz NE, Boecker-Schlier R, Buchmann AF, Blomeyer D, Jennen-Steinmetz C, Baumeister S, et al. Ventral striatum and amygdala activity as convergence sites for early adversity and conduct disorder. Soc Cogn Affect Neurosci. 2017;12:261–72.
Gard AM, Waller R, Shaw DS, Forbes EE, Hariri AR, Hyde LW. The long reach of early adversity: parenting, stress, and neural pathways to antisocial behavior in adulthood. Biol Psychiatry Cogn Neurosci Neuroimaging. 2017;2:582–90.
Mehta MA, Golembo NI, Nosarti C, Colvert E, Mota A, Williams SC, et al. Amygdala, hippocampal and corpus callosum size following severe early institutional deprivation: the English and Romanian Adoptees study pilot. J Child Psychol Psychiatry. 2009;50:943–51.
Tottenham N, Hare TA, Quinn BT, McCarry TW, Nurse M, Gilhooly T, et al. Prolonged institutional rearing is associated with atypically large amygdala volume and difficulties in emotion regulation. Dev Sci. 2010;13:46–61.
Woon FL, Hedges DW. Hippocampal and amygdala volumes in children and adults with childhood maltreatment-related posttraumatic stress disorder: a meta-analysis. Hippocampus. 2008;18:729–36.
Hanson JL, Nacewicz BM, Sutterer MJ, Cayo AA, Schaefer SM, Rudolph KD, et al. Behavioral problems after early life stress: contributions of the hippocampus and amygdala. Biol Psychiatry. 2015;77:314–23.
Noble KG, Houston SM, Kan E, Sowell ER. Neural correlates of socioeconomic status in the developing human brain. Dev Sci. 2012;15:516–27.
Green SA, Goff B, Gee DG, Gabard-Durnam L, Flannery J, Telzer EH, et al. Discrimination of amygdala response predicts future separation anxiety in youth with early deprivation. J Child Psychol Psychiatry. 2016;57:1135–44.
Boecker R, Holz NE, Buchmann AF, Blomeyer D, Plichta MM, Wolf I, et al. Impact of early life adversity on reward processing in young adults: EEG-fMRI results from a prospective study over 25 years. PLoS One. 2014;9:e104185.
Dillon DG, Holmes AJ, Birk JL, Brooks N, Lyons-Ruth K, Pizzagalli DA. Childhood adversity is associated with left basal ganglia dysfunction during reward anticipation in adulthood. Biol Psychiatry. 2009;66:206–13.
Mehta MA, Gore-Langton E, Golembo N, Colvert E, Williams SC, Sonuga-Barke E. Hyporesponsive reward anticipation in the basal ganglia following severe institutional deprivation early in life. J Cogn Neurosci. 2010;22:2316–25.
Holz NE, Boecker-Schlier R, Jennen-Steinmetz C, Hohm E, Buchmann AF, Blomeyer D, et al. Early maternal care may counteract familial liability for psychopathology in the reward circuitry. Soc Cogn Affect Neurosci. 2018;13:1191–201.
Furukawa E, Bado P, Tripp G, Mattos P, Wickens JR, Bramati IE, et al. Abnormal striatal BOLD responses to reward anticipation and reward delivery in ADHD. PLoS One. 2014;9:e89129.
Plichta M, Scheres A. Ventral–striatal responsiveness during reward anticipation in ADHD and its relation to trait impulsivity in the healthy population: a meta-analytic review of the fMRI literature. Neurosci Biobehav Rev. 2013;38:125–34.
Edmiston EE, Wang F, Mazure CM, Guiney J, Sinha R, Mayes LC, et al. Corticostriatal-limbic gray matter morphology in adolescents with self-reported exposure to childhood maltreatment. Arch Pediatr Adolesc Med. 2011;165:1069–77.
Mackey S, Chaarani B, Kan KJ, Spechler PA, Orr C, Banaschewski T, et al. Brain regions related to impulsivity mediate the effects of early adversity on antisocial behavior. Biol Psychiatry. 2017;82:275–82.
Holz NE, Boecker R, Jennen-Steinmetz C, Buchmann AF, Blomeyer D, Baumeister S, et al. Positive coping styles and perigenual ACC volume: two related mechanisms for conferring resilience? Soc Cogn Affect Neurosci. 2016;11:813–20.
Nes LS, Segerstrom SC. Dispositional optimism and coping: a meta-analytic review. Pers Soc Psychol Rev. 2006;10:235–51.
Dolcos S, Hu Y, Iordan AD, Moore M, Dolcos F. Optimism and the brain: trait optimism mediates the protective role of the orbitofrontal cortex gray matter volume against anxiety. Soc Cogn Affect Neurosci. 2016;11:263–71.
Morey RA, Haswell CC, Hooper SR, De Bellis MD. Amygdala, hippocampus, and ventral medial prefrontal cortex volumes differ in maltreated youth with and without chronic posttraumatic stress disorder. Neuropsychopharmacology. 2016;41:791–801.
Sinha R, Lacadie CM, Constable RT, Seo D. Dynamic neural activity during stress signals resilient coping. Proc Natl Acad Sci USA. 2016;113:8837–42.
Eisenberger NI, Master SL, Inagaki TK, Taylor SE, Shirinyan D, Lieberman MD, et al. Attachment figures activate a safety signal-related neural region and reduce pain experience. Proc Natl Acad Sci USA. 2011;108:11721–6.
Younger J, Aron A, Parke S, Chatterjee N, Mackey S. Viewing pictures of a romantic partner reduces experimental pain: involvement of neural reward systems. PLoS One. 2010;5:e13309.
Coan JA, Schaefer HS, Davidson RJ. Lending a hand: social regulation of the neural response to threat. Psychol Sci. 2006;17:1032–9.
Eisenberger NI, Taylor SE, Gable SL, Hilmert CJ, Lieberman MD. Neural pathways link social support to attenuated neuroendocrine stress responses. Neuroimage. 2007;35:1601–12.
Bowes L, Maughan B, Caspi A, Moffitt TE, Arseneault L. Families promote emotional and behavioural resilience to bullying: evidence of an environmental effect. J Child Psychol Psychiatry. 2010;51:809–17.
Garmezy N, Masten AS, Tellegen A. The study of stress and competence in children: a building block for developmental psychopathology. Child Dev. 1984;55:97–111.
Belsky J, Bakermans-Kranenburg MJ, van Ijzendoorn MH. For better and for worse: Differential susceptibility to environmental influences. Curr Dir Psychol Sci. 2007;16:300–4.
Laucht M, Esser G, Baving L, Gerhold M, Hoesch I, Ihle W, et al. Behavioral sequelae of perinatal insults and early family adversity at 8 years of age. J Am Acad Child Adolesc Psychiatry. 2000;39:1229–37.
Morgan JK, Shaw DS, Forbes EE. Maternal depression and warmth during childhood predict age 20 neural response to reward. J Am Acad Child Adolesc Psychiatry. 2014;53:108–17 e101.
Wang Y, Zhang L, Kong X, Hong Y, Cheon B, Liu J. Pathway to neural resilience: Self-esteem buffers against deleterious effects of poverty on the hippocampus. Hum Brain Mapp. 2016;37:3757–66.
Luthar SS, Cicchetti D, Becker B. The construct of resilience: a critical evaluation and guidelines for future work. Child Dev. 2000;71:543–62.
Kiser D, Steemers B, Branchi I, Homberg JR. The reciprocal interaction between serotonin and social behaviour. Neurosci Biobehav Rev. 2012;36:786–98.
Cao H, Harneit A, Walter H, Erk S, Braun U, Moessnang C, et al. The 5-HTTLPR Polymorphism Affects Network-Based Functional Connectivity in the Visual-Limbic System in Healthy Adults. Neuropsychopharmacology. 2018;43:406–14.
Feder A, Nestler EJ, Charney DS. Psychobiology and molecular genetics of resilience. Nat Rev Neurosci. 2009;10:446–57.
Dreher JC, Meyer-Lindenberg A, Kohn P, Berman KF. Age-related changes in midbrain dopaminergic regulation of the human reward system. Proc Natl Acad Sci USA. 2008;105:15106–11.
Charney DS. Psychobiological mechanisms of resilience and vulnerability: implications for successful adaptation to extreme stress. Am J Psychiatry. 2004;161:195–216.
Dunlop BW, Nemeroff CB. The role of dopamine in the pathophysiology of depression. Arch Gen Psychiatry. 2007;64:327–37.
Hoexter MQ, Fadel G, Felicio AC, Calzavara MB, Batista IR, Reis MA, et al. Higher striatal dopamine transporter density in PTSD: an in vivo SPECT study with [(99m)Tc]TRODAT-1. Psychopharmacol. 2012;224:337–45.
Matosin N, Halldorsdottir T, Binder EB. Understanding the molecular mechanisms underpinning gene by environment interactions in psychiatric disorders: the FKBP5 model. Biol Psychiatry. 2018;83:821–30.
Meyer-Lindenberg A, Buckholtz JW, Kolachana B, Hariri R, Pezawas L, Blasi G, et al. Neural mechanisms of genetic risk for impulsivity and violence in humans. Proc Natl Acad Sci USA. 2006;103:6269–74.
Alia-Klein N, Goldstein RZ, Tomasi D, Woicik PA, Moeller SJ, Williams B, et al. Neural mechanisms of anger regulation as a function of genetic risk for violence. Emotion. 2009;9:385–96.
Lee BT, Ham BJ. Monoamine oxidase A-uVNTR genotype affects limbic brain activity in response to affective facial stimuli. Neuroreport. 2008;19:515–9.
Fan J, Fossella J, Sommer T, Wu Y, Posner MI. Mapping the genetic variation of executive attention onto brain activity. Proc Natl Acad Sci USA. 2003;100:7406–11.
Passamonti L, Fera F, Magariello A, Cerasa A, Gioia MC, Muglia M, et al. Monoamine oxidase-a genetic variations influence brain activity associated with inhibitory control: new insight into the neural correlates of impulsivity. Biol Psychiatry. 2006;59:334–40.
Passamonti L, Cerasa A, Gioia MC, Magariello A, Muglia M, Quattrone A, et al. Genetically dependent modulation of serotonergic inactivation in the human prefrontal cortex. Neuroimage. 2008;40:1264–73.
Byrd AL, Manuck SB. MAOA, childhood maltreatment, and antisocial behavior: meta-analysis of a gene-environment interaction. Biol Psychiatry. 2014;75:9–17.
Caspi A, McClay J, Moffitt TE, Mill J, Martin J, Craig IW, et al. Role of genotype in the cycle of violence in maltreated children. Science. 2002;297:851–4.
Holz N, Boecker R, Buchmann AF, Blomeyer D, Baumeister S, Hohmann S, et al. Evidence for a sex-dependent MAOAx childhood stress interaction in the neural circuitry of aggression. Cereb Cortex. 2016;26:904–14.
Lesch KP, Bengel D, Heils A, Sabol SZ, Greenberg BD, Petri S, et al. Association of anxiety-related traits with a polymorphism in the serotonin transporter gene regulatory region. Science. 1996;274:1527–31.
Munafo MR, Brown SM, Hariri AR. Serotonin transporter (5-HTTLPR) genotype and amygdala activation: a meta-analysis. Biol Psychiatry. 2008;63:852–7.
Pezawas L, Meyer-Lindenberg A, Drabant EM, Verchinski BA, Munoz KE, Kolachana BS, et al. 5-HTTLPR polymorphism impacts human cingulate-amygdala interactions: a genetic susceptibility mechanism for depression. Nat Neurosci. 2005;8:828–34.
Alexander N, Klucken T, Koppe G, Osinsky R, Walter B, Vaitl D, et al. Interaction of the serotonin transporter-linked polymorphic region and environmental adversity: increased amygdala-hypothalamus connectivity as a potential mechanism linking neural and endocrine hyperreactivity. Biol Psychiatry. 2012;72:49–56.
Canli T, Qiu M, Omura K, Congdon E, Haas BW, Amin Z, et al. Neural correlates of epigenesis. Proc Natl Acad Sci USA. 2006;103:16033–8.
van der Meer D, Hoekstra PJ, Zwiers M, Mennes M, Schweren LJ, Franke B, et al. Brain correlates of the interaction between 5-httlpr and psychosocial stress mediating attention deficit hyperactivity disorder severity. Am J Psychiatry. 2015;172:768–75.
Schmack K, Schlagenhauf F, Sterzer P, Wrase J, Beck A, Dembler T, et al. Catechol-O-methyltransferase val158met genotype influences neural processing of reward anticipation. Neuroimage. 2008;42:1631–8.
Dreher J-C, Kohn P, Kolachana B, Weinberger DR, Berman KF. Variation in dopamine genes influences responsivity of the human reward system. Proc Natl Acad Sci. 2009;106:617–22.
Yacubian J, Sommer T, Schroeder K, Glascher J, Kalisch R, Leuenberger B, et al. Gene-gene interaction associated with neural reward sensitivity. Proc Natl Acad Sci USA. 2007;104:8125–30.
Camara E, Kramer UM, Cunillera T, Marco-Pallares J, Cucurell D, Nager W, et al. The effects of COMT (Val108/158Met) and DRD4 (SNP -521) dopamine genotypes on brain activations related to valence and magnitude of rewards. Cereb Cortex. 2010;20:1985–96.
Forbes EE, Brown SM, Kimak M, Ferrell RE, Manuck SB, Hariri AR. Genetic variation in components of dopamine neurotransmission impacts ventral striatal reactivity associated with impulsivity. Mol Psychiatry. 2009;14:60–70.
Boecker-Schlier R, Holz NE, Buchmann AF, Blomeyer D, Plichta MM, Jennen-Steinmetz C, et al. Interaction between COMT Val(158)Met polymorphism and childhood adversity affects reward processing in adulthood. Neuroimage. 2016;132:556–70.
Van Tol HH, Wu CM, Guan HC, Ohara K, Bunzow JR, Civelli O, et al. Multiple dopamine D4 receptor variants in the human population. Nature. 1992;358:149–52.
McClernon FJ, Hutchison KE, Rose JE, Kozink RV. DRD4 VNTR polymorphism is associated with transient fMRI-BOLD responses to smoking cues. Psychopharmacol. 2007;194:433–41.
Filbey FM, Ray L, Smolen A, Claus ED, Audette A, Hutchison KE. Differential neural response to alcohol priming and alcohol taste cues is associated with DRD4 VNTR and OPRM1 genotypes. Alcohol Clin Exp Res. 2008;32:1113–23.
Nikolova YS, Ferrell RE, Manuck SB, Hariri AR. Multilocus genetic profile for dopamine signaling predicts ventral striatum reactivity. Neuropsychopharmacology. 2011;36:1940–7.
Wittmann BC, Tan GC, Lisman JE, Dolan RJ, Düzel E. DAT genotype modulates striatal processing and long-term memory for items associated with reward and punishment. Neuropsychologia. 2013;51:2184–93.
Aarts E, Roelofs A, Franke B, Rijpkema M, Fernandez G, Helmich RC, et al. Striatal dopamine mediates the interface between motivational and cognitive control in humans: evidence from genetic imaging. Neuropsychopharmacology. 2010;35:1943–51.
Hahn T, Heinzel S, Dresler T, Plichta MM, Renner TJ, Markulin F, et al. Association between reward-related activation in the ventral striatum and trait reward sensitivity is moderated by dopamine transporter genotype. Hum Brain Mapp. 2011;32:1557–65.
Paloyelis Y, Mehta MA, Faraone SV, Asherson P, Kuntsi J. Striatal sensitivity during reward processing in attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2012;51:722–32.
Hoogman M, Onnink M, Cools R, Aarts E, Kan C, Arias Vasquez A, et al. The dopamine transporter haplotype and reward-related striatal responses in adult ADHD. Eur Neuropsychopharmacol. 2013;23:469–78.
Binder EB. The role of FKBP5, a co-chaperone of the glucocorticoid receptor in the pathogenesis and therapy of affective and anxiety disorders. Psychoneuroendocrinology. 2009;34(Suppl 1):S186–S195.
Denny WB, Valentine DL, Reynolds PD, Smith DF, Scammell JG. Squirrel monkey immunophilin FKBP51 is a potent inhibitor of glucocorticoid receptor binding. Endocrinology. 2000;141:4107–13.
Wochnik GM, Ruegg J, Abel GA, Schmidt U, Holsboer F, Rein T. FK506-binding proteins 51 and 52 differentially regulate dynein interaction and nuclear translocation of the glucocorticoid receptor in mammalian cells. J Biol Chem. 2005;280:4609–16.
Binder EB, Bradley RG, Liu W, Epstein MP, Deveau TC, Mercer KB, et al. Association of FKBP5 polymorphisms and childhood abuse with risk of posttraumatic stress disorder symptoms in adults. JAMA. 2008;299:1291–305.
Zannas AS, Wiechmann T, Gassen NC, Binder EB. Gene-stress-epigenetic regulation of FKBP5: clinical and translational implications. Neuropsychopharmacology. 2016;41:261–74.
White MG, Bogdan R, Fisher PM, Munoz KE, Williamson DE, Hariri AR. FKBP5 and emotional neglect interact to predict individual differences in amygdala reactivity. Genes Brain Behav. 2012;11:869–78.
Holz NE, Buchmann AF, Boecker R, Blomeyer D, Baumeister S, Wolf I, et al. Role of FKBP5 in emotion processing: results on amygdala activity, connectivity and volume. Brain Struct Funct. 2015;220:1355–68.
VanZomeren-Dohm AA, Pitula CE, Koss KJ, Thomas K, Gunnar MR. FKBP5 moderation of depressive symptoms in peer victimized, post-institutionalized children. Psychoneuroendocrinology. 2015;51:426–30.
Davidson RJ, McEwen BS. Social influences on neuroplasticity: stress and interventions to promote well-being. Nat Neurosci. 2012;15:689–95.
Bilek E, Stossel G, Schafer A, Clement L, Ruf M, Robnik L, et al. State-dependent cross-brain information flow in borderline personality disorder. JAMA Psychiatry. 2017;74:949–57.
Trull TJ, Ebner-Priemer U. Ambulatory assessment. Annu Rev Clin Psychol. 2013;9:151–76.
Forbes EE, Hariri AR, Martin SL, Silk JS, Moyles DL, Fisher PM, et al. Altered striatal activation predicting real-world positive affect in adolescent major depressive disorder. Am J Psychiatry. 2009;166:64–73.
Nagai Y. Modulation of autonomic activity in neurological conditions: Epilepsy and Tourette Syndrome. Front Neurosci. 2015;9:278.
Strehl U, Aggensteiner P, Wachtlin D, Brandeis D, Albrecht B, Arana M, et al. Neurofeedback of slow cortical potentials in children with attention-deficit/hyperactivity disorder: a multicenter randomized trial controlling for unspecific effects. Front Hum Neurosci. 2017;11:135.
Konicar L, Veit R, Eisenbarth H, Barth B, Tonin P, Strehl U, et al. Brain self-regulation in criminal psychopaths. Sci Rep. 2015;5:9426.
Herwig U, Lutz J, Scherpiet S, Scheerer H, Kohlberg J, Opialla S, et al. Training emotion regulation through real-time fMRI neurofeedback of amygdala activity. Neuroimage. 2019;184:687–96.
Li Z, Zhang CY, Huang J, Wang Y, Yan C, Li K, et al. Improving motivation through real-time fMRI-based self-regulation of the nucleus accumbens. Neuropsychology. 2018;32:764–76.
Alegria AA, Wulff M, Brinson H, Barker GJ, Norman LJ, Brandeis D, et al. Real-time fMRI neurofeedback in adolescents with attention deficit hyperactivity disorder. Hum Brain Mapp. 2017;38:3190–209.
Scheinost D, Stoica T, Saksa J, Papademetris X, Constable RT, Pittenger C, et al. Orbitofrontal cortex neurofeedback produces lasting changes in contamination anxiety and resting-state connectivity. Transl Psychiatry. 2013;3:e250.
Aggensteiner PM, Brandeis D, Millenet S, Hohmann S, Ruckes C, Beuth S, et al. Slow cortical potentials neurofeedback in children with ADHD: comorbidity, self-regulation and clinical outcomes 6 months after treatment in a multicenter randomized controlled trial. Eur Child Adolesc Psychiatry. 2019;28:1087–95.
Parker KJ, Buckmaster CL, Schatzberg AF, Lyons DM. Prospective investigation of stress inoculation in young monkeys. Arch Gen Psychiatry. 2004;61:933–41.
Schweizer S, Walsh ND, Stretton J, Dunn VJ, Goodyer IM, Dalgleish T. Enhanced emotion regulation capacity and its neural substrates in those exposed to moderate childhood adversity. Soc Cogn Affect Neurosci. 2016;11:272–81.
Arango C, Diaz-Caneja CM, McGorry PD, Rapoport J, Sommer IE, Vorstman JA, et al. Preventive strategies for mental health. Lancet Psychiatry. 2018;5:591–604.
Acknowledgements
The authors gratefully acknowledge grant support from the German Research Foundation (grant numbers DFG HO 5674/2-1, GRK2350/1) and the Olympia Morata Program of the University of Heidelberg to NEH. AML acknowledges grant support by the German Research Foundation (DFG, Research Training Group GRK2350/1 project B02, Collaborative Research Center SFB 1158 project B09, Collaborative Research Center TRR 265 project S02, grant ME 1591/4-1), German Federal Ministry of Education and Research (BMBF, grants 01EF1803A, 01ZX1314G, 01GQ1003B), European Union’s Seventh Framework Programme (FP7, grants 602450, 602805, 115300, HEALTH-F2-2010-241909), Innovative Medicines Initiative Joint Undertaking (IMI, grant 115008) and Ministry of Science, Research and the Arts of the State of Baden-Wuerttemberg, Germany (MWK, grant 42-04HV.MED(16)/16/1). HT acknowledges grant support by the DFG (Research Training Group GRK2350/1 project B02, Collaborative Research Center SFB 1158 project B04, Collaborative Research Center TRR 265 project A04) and BMBF (grants 01EF1803A project WP3, 01GQ1102). The funding source had no role in the writing of the report and in the decision to submit the article for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
AML has received consultant fees from American Association for the Advancement of Science, Atheneum Partners, Blueprint Partnership, Boehringer Ingelheim, Daimler und Benz Stiftung, Elsevier, F. Hoffmann-La Roche, ICARE Schizophrenia, K. G. Jebsen Foundation, L.E.K Consulting, Lundbeck International Foundation (LINF), R. Adamczak, Roche Pharma, Science Foundation, Sumitomo Dainippon Pharma, Synapsis Foundation—Alzheimer Research Switzerland, System Analytics, and has received lectures fees including travel fees from Boehringer Ingelheim, Fama Public Relations, Institut d’investigacions Biomèdiques August Pi i Sunyer (IDIBAPS), Janssen-Cilag, Klinikum Christophsbad, Göppingen, Lilly Deutschland, Luzerner Psychiatrie, LVR Klinikum Düsseldorf, LWL Psychiatrie Verbund Westfalen-Lippe, Otsuka Pharmaceuticals, Reunions i Ciencia S. L., Spanish Society of Psychiatry, Südwestrundfunk Fernsehen, Stern TV, and Vitos Klinikum Kurhessen. The other authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Holz, N.E., Tost, H. & Meyer-Lindenberg, A. Resilience and the brain: a key role for regulatory circuits linked to social stress and support. Mol Psychiatry 25, 379–396 (2020). https://doi.org/10.1038/s41380-019-0551-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-019-0551-9
This article is cited by
-
Midbrain glutamatergic circuit mechanism of resilience to socially transferred allodynia in male mice
Nature Communications (2024)
-
Positive parent–child interactions moderate certain maltreatment effects on psychosocial well-being in 6-year-old children
Pediatric Research (2024)
-
Blunted brain responses to neutral faces in healthy first-degree relatives of patients with schizophrenia: an image-based fMRI meta-analysis
Schizophrenia (2024)
-
How does the macroenvironment influence brain and behaviour—a review of current status and future perspectives
Molecular Psychiatry (2024)
-
Broadening the Scope of Resilience in Chronic Pain: Methods, Social Context, and Development
Current Rheumatology Reports (2024)