Abstract
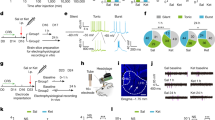
Ketamine produces rapid and robust antidepressant effects in depressed patients within hours of administration, often when traditional antidepressant compounds have failed to alleviate symptoms. We hypothesized that ketamine would translocate Gαs from lipid rafts to non-raft microdomains, similarly to other antidepressants but with a distinct, abbreviated treatment duration. C6 glioma cells were treated with 10 µM ketamine for 15 min, which translocated Gαs from lipid raft domains to non-raft domains. Other NMDA antagonist did not translocate Gαs from lipid raft to non-raft domains. The ketamine-induced Gαs plasma membrane redistribution allows increased functional coupling of Gαs and adenylyl cyclase to increase intracellular cyclic adenosine monophosphate (cAMP). Moreover, increased intracellular cAMP increased phosphorylation of cAMP response element-binding protein (CREB), which, in turn, increased BDNF expression. The ketamine-induced increase in intracellular cAMP persisted after knocking out the NMDA receptor indicating an NMDA receptor-independent effect. Furthermore, 10 µM of the ketamine metabolite (2R,6R)-hydroxynorketamine (HNK) also induced Gαs redistribution and increased cAMP. These results reveal a novel antidepressant mechanism mediated by acute ketamine treatment that may contribute to ketamine’s powerful antidepressant effect. They also suggest that the translocation of Gαs from lipid rafts is a reliable hallmark of antidepressant action that might be exploited for diagnosis or drug development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kessler RC, Bromet EJ. The epidemiology of depression across cultures. Annu Rev Public Health. 2013;34:119–38. https://doi.org/10.1146/annurev-publhealth-031912-114409
Rush AJ, Trivedi MH, Wisniewski SR, Nierenberg AA, Stewart JW, Warden D, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: A STAR*D report. Am J Psychiatry. 2006;163:1905–17. https://doi.org/10.1176/appi.ajp.163.11.1905
Berman, RM, Cappiello, A, Anand, A, Oren, DA, Heninger, GR, Charney, DS, et al. Antidepressant effects of ketamine in depressed patients. Biol Psychiatry. 2000;47:351–4. https://doi.org/10.1016/S0006-3223(99)00230-9
Zarate CA, Singh JB, Carlson PJ, Brutsche NE, Ameli R, Luckenbaugh DA, et al. A randomized trial of an N-methyl-d-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry. 2006;63:856–64. https://doi.org/10.1001/archpsyc.63.8.856
Abdallah CG, Sanacora G, Duman RS, Krystal JH. Ketamine and rapid-acting antidepressants: a window into a new neurobiology for mood disorder therapeutics. Annu Rev Med. 2015;66:509–23. https://doi.org/10.1146/annurev-med-053013-062946
Lener MS, Niciu MJ, Ballard ED, Park M, Park LT, Nugent AC, et al. Glutamate and gamma-aminobutyric acid systems in the pathophysiology of major depression and antidepressant response to ketamine. Biol Psychiatry. 2016;81:1–12. https://doi.org/10.1016/j.biopsych.2016.05.005
Sanacora G, Treccani G, Popoli M. Towards a glutamate hypothesis of depression: an emerging frontier of neuropsychopharmacology for mood disorders. Neuropharmacology. 2012;62:63–77. https://doi.org/10.1016/j.neuropharm.2011.07.036
Sanacora G, Schatzberg AF. Ketamine: promising path or false prophecy in the development of novel therapeutics for mood disorders? Neuropsychopharmacology. 2015;40:259–67. https://doi.org/10.1038/npp.2014.261
Li, N. mTOR-dependent synapse formation. Science. 2010;329:959–65. https://doi.org/10.1126/science.1190287
Autry AE, Adachi M, Nosyreva E, Na ES, Los MF, Cheng P, et al. NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature. 2011;475:91–95. https://doi.org/10.1038/nature10130
Maeng S, Zarate CA, Du J, Schloesser RJ, McCammon J, Chen G, Manji HK. Cellular mechanisms underlying the antidepressant effects of ketamine: role of α-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptors. Biol Psychiatry. 2008;63:349–52. https://doi.org/10.1016/j.biopsych.2007.05.028
Newport DJ,Div M,Carpenter LL,Mcdonald WM,Potash JB, Ketamine and other NMDA antagonists: early clinical trials and possible mechanisms in depression. Am J Psychiatry. 2015;172:950–66. https://doi.org/10.1176/appi.ajp.2015.15040465.
Zanos P, Moaddel R, Morris PJ, Georgiou P, Fischell J, Elmer GI, et al. NMDAR inhibition-independent antidepressant actions of ketamine metabolites. Nature. 2016;533:481–6. https://doi.org/10.1038/nature17998
Yang C, Shirayama Y, Zhang J, Ren Q, Yao W, Ma M, et al. R-ketamine: a rapid-onset and sustained antidepressant without psychotomimetic side effects. Transl Psychiatry. 2015;5:e632 https://doi.org/10.1038/tp.2015.136
Nibuya M, Morinobu S, Duman RS. Regulation of BDNF and trkB mRNA in rat brain by chronic electroconvulsive seizure and antidepressant drug treatments. J Neurosci. 1995;15:7539–47.
Malberg JE, Blendy JA. Antidepressant action: to the nucleus and beyond. Trends Pharmacol Sci. 2005;26:631–8. https://doi.org/10.1016/j.tips.2005.10.005
Gass P, Riva MA. CREB neurogenesis and depression. Bioessays. 2007;29:957–61. https://doi.org/10.1002/bies.20658
Dwivedi Y, Pandey GN. Adenylyl cyclase-cyclic AMP signaling in mood disorders: role of the crucial phosphorylating enzyme protein kinase A. Neuropsychiatr Dis Treat. 2008;4(1 A):161–76. https://doi.org/10.2147/NDT.S2380
Fujita M, Richards EM, Niciu MJ, Ionescu DF, Zoghbi SS, Hong J, et al. cAMP signaling in brain is decreased in unmedicated depressed patients and increased by treatment with a selective serotonin reuptake inhibitor HHS public access. Mol Psychiatry. 2017;22:754–9. https://doi.org/10.1038/mp.2016.171
Toki S, Donati RJ, Rasenick MM. Treatment of C6 glioma cells and rats with antidepressant drugs increases the detergent extraction of Gαs from plasma membrane. J Neurochem. 1999;73:1114–20. https://doi.org/10.1046/j.1471-4159.1999.0731114.x
Donati RJ, Dwivedi Y, Roberts RC, Conley RR, Pandey GN, Rasenick MM. Postmortem brain tissue of depressed suicides reveals increased Gαs localization in lipid raft domains where it is less likely to activate adenylyl cyclase. J Neurosci. 2008;28:3042–50. https://doi.org/10.1523/JNEUROSCI.5713-07.2008
Zhang, L, & Rasenick, MM (2010). Chronic treatment with escitalopram but not R-citalopram translocates Gαs from lipid raft domains and potentiates adenylyl cyclase: a 5-hydroxytryptamine transporter-independent action of this antidepressant compound. J Pharmacol Exp Ther. 2010;332: 977–84. https://doi.org/10.1124/jpet.109.162644.sponse
Czysz AH, Schappi JM, Rasenick MM. Lateral diffusion of Gαs in the plasma membrane is decreased after chronic but not acute antidepressant treatment: role of lipid raft and non-raft membrane microdomains. Neuropsychopharmacology. 2014;40:1–8. https://doi.org/10.1038/npp.2014.256
Allen JA, Halverson-Tamboli RA, Rasenick MM. Lipid raft microdomains and neurotransmitter signalling. Nat Rev Neurosci. 2007;8:128–40. https://doi.org/10.1038/nrn2059
Wedegaertner PB, Chu DH, Wilson PT, Levis MJ, Bourne HR. Palmitoylation is required for signaling functions and membrane attachment of Gαq and Gαs. J Biol Chem. 1993;268:25001–8.
Allen JA, Yu JZ, Dave RH, Bhatnagar A, Roth BL, Rasenick MM. Caveolin-1 and lipid microdomains regulate Gαs trafficking and attenuate Gαs /adenylyl cyclase signaling. Mol Pharmacol. 2009;76:1082–93. https://doi.org/10.1124/mol.109.060160
Popp S, Behl B, Joshi JJ, Lanz TA, Spedding M, Schenker E, et al. In search of the mechanisms of ketamine’s antidepressant effects: how robust is the evidence behind the mTor activation hypothesis [version 1; referees: 1 approved, 1 approved with reservations]. F1000Research. 2016;5:634 https://doi.org/10.12688/f1000research.8236.1
Sos P, Klirova M, Novak T, Kohutova B, Horacek J, Palenicek T. Relationship of ketamine’s antidepressant and psychotomimetic effects in unipolar depression. Neuro Endocrinol Lett. 2013;34:287–93.
Erb, SJ, Schappi, JM, & Rasenick, MM (2016). Antidepressants accumulate in lipid rafts independent of monoamine transporters to modulate redistribution of the G protein, Gαs. J Biol Chem. 2016;291:19725–33. https://doi.org/10.1074/jbc.M116.727263
Tewson PH, Martinka S, Shaner NC, Hughes TE, Quinn AM. New DAG and cAMP sensors optimized for live-cell assays in automated laboratories. J Biomol Screen. 2016;21:298–305. https://doi.org/10.1177/1087057115618608
Ulbrich MH, Isacoff EY. Rules of engagement for NMDA receptor subunits. Proc Natl Acad Sci USA. 2008;105:14163–8. https://doi.org/10.1073/pnas.0802075105
Duman RS, Malberg J, Thome J. Neural plasticity to stress and antidepressant treatment. Biol Psychiatry. 1999;46:1181–91. https://doi.org/10.1016/S0006-3223(99)00177-8
Browne GJ, Proud CG. Regulation of peptide-chain elongation in mammalian cells. Eur J Biochem. 2002;5368:5360–8. https://doi.org/10.1046/j.1432-1033.2002.03290.x. October
Coppell AL, Pei Q, Zetterström TSC. Bi-phasic change in BDNF gene expression following antidepressant drug treatment. Neuropharmacology. 2003;44:903–10. https://doi.org/10.1016/S0028-3908(03)00077-7
Suzuki K, Nosyreva E, Hunt KW, Kavalali ET, Monteggia LM. Effects of a ketamine metabolite on synaptic NMDAR function. Nature. 2017;546:E1–E3. https://doi.org/10.1038/nature22084
Xue W, Wang W, Gong T, Zhang H, Tao W, Xue L, et al. PKA-CREB-BDNF signaling regulated long lasting antidepressant activities of Yueju but not ketamine. Sci Rep. 2016;6:26331 https://doi.org/10.1038/srep26331.
Réus GZ, Stringari RB, Ribeiro KF, Ferraro AK, Vitto MF, Cesconetto P, et al. Ketamine plus imipramine treatment induces antidepressant-like behavior and increases CREB and BDNF protein levels and PKA and PKC phosphorylation in rat brain. Behav Brain Res. 2011;221:166–71. https://doi.org/10.1016/j.bbr.2011.02.024
Wang Q, Jie W, Liu JH, Yang JM, Gao TM. An astroglial basis of major depressive disorder? An overview. Glia. 2017;65:1227–50. https://doi.org/10.1002/glia.23143
Quesseveur G, David DJ, Gaillard MC, Pla P, Wu MV, Nguyen HT, et al. BDNF overexpression in mouse hippocampal astrocytes promotes local neurogenesis and elicits anxiolytic-like activities. Transl Psychiatry. 2013;3:e253 https://doi.org/10.1038/tp.2013.30.
Singh, H, Wray, N, Schappi, JM, & Rasenick, MM (2018). Disruption of lipid-raft localized Gαs /tubulin complexes by antidepressants: a unique feature of HDAC6 inhibitors, SSRI and tricyclic compounds. Neuropsychopharmacology. 2018; 1–11. https://doi.org/10.1038/s41386-018-0016-x
Jerabek H, Pabst G, Rappolt M, Stockner T. Membrane-mediated effect on ion channels induced by the anesthetic drug ketamine. J Am Chem Soc. 2010;132:7990–7. https://doi.org/10.1021/ja910843d
Bademosi AT, Steeves J, Karunanithi S, Zalucki OH, Gormal RS, Liu S, et al. Trapping of Syntaxin1a in presynaptic nanoclusters by a clinically relevant general anesthetic. Cell Rep. 2018;22:427–40. https://doi.org/10.1016/j.celrep.2017.12.054
Acknowledgements
This research is supported by VA Merit Award BX00149 (MMR) and NIH R01AT009169 (MMR). NHW and JS were supported by T32 MH067631 and HS by an AHA postdoctoral fellowship. (2R,6R)-HNK was gifted by the NIH.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
MMR has received research support from Eli Lilly and Lundbeck, Inc. and is consultant to Otsuka Pharmaceuticals. He also has ownership in Pax Neuroscience. The remaining authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wray, N.H., Schappi, J.M., Singh, H. et al. NMDAR-independent, cAMP-dependent antidepressant actions of ketamine. Mol Psychiatry 24, 1833–1843 (2019). https://doi.org/10.1038/s41380-018-0083-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-018-0083-8
This article is cited by
-
Hydroxynorketamine, but not ketamine, acts via α7 nicotinic acetylcholine receptor to control presynaptic function and gene expression
Translational Psychiatry (2024)
-
Antidepressants enter cells, organelles, and membranes
Neuropsychopharmacology (2024)
-
Enhanced TARP-γ8-PSD-95 coupling in excitatory neurons contributes to the rapid antidepressant-like action of ketamine in male mice
Nature Communications (2023)
-
A novel peripheral biomarker for depression and antidepressant response
Molecular Psychiatry (2022)
-
Target deconvolution studies of (2R,6R)-hydroxynorketamine: an elusive search
Molecular Psychiatry (2022)