Abstract
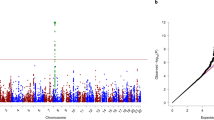
Cannabis use is increasing in the United States, as are its adverse effects. We investigated the genetics of an adverse consequence of cannabis use: cannabis-related aggression (CRA) using a genome-wide association study (GWAS) design. Our GWAS sample included 3269 African Americans (AAs) and 2546 European Americans (EAs). An additional 89 AA subjects from the Grady Trauma Project (GTP) were also examined using a proxy-phenotype replication approach. We identified genome-wide significant risk loci contributing to CRA in AAs at the serotonin receptor 2B receptor gene (HTR2B), and the lead SNP, HTR2B*rs17440378, showed nominal association to aggression in the GTP cohort of cannabis-exposed subjects. A priori evidence linked HTR2B to impulsivity/aggression but not to cannabis response. Human functional data regarding the HTR2B variant further supported our finding. Treating an Htr2b−/− knockout mouse with THC resulted in increased aggressive behavior, whereas wild-type mice following THC administration showed decreased aggression in the resident-intruder paradigm, demonstrating that HTR2B variation moderates the effects of cannabis on aggression. These concordant findings in mice and humans implicate HTR2B as a major locus associated with cannabis-induced aggression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Siever LJ. Neurobiology of aggression and violence. Am J Psychiatry. 2008;165:429–42.
Schoeler T, Theobald D, Pingault JB, Farrington DP, Jennings WG, Piquero AR, et al. Continuity of cannabis use and violent offending over the life course. Psychol Med. 2016;46:1663–77.
Veroude K, Zhang-James Y, Fernandez-Castillo N, Bakker MJ, Cormand B, Faraone SV. Genetics of aggressive behavior: an overview. Am J Med Genet Part B Neuropsychiatr Genet. 2016;171B:3–43.
Waltes R, Chiocchetti AG, Freitag CM. The neurobiological basis of human aggression: a review on genetic and epigenetic mechanisms. Am J Med Genet Part B Neuropsychiatr Genet. 2016;171:650–75.
Brunner HG, Nelen M, Breakefield XO, Ropers HH, van Oost BA. Abnormal behavior associated with a point mutation in the structural gene for monoamine oxidase A. Science. 1993;262:578–80.
Brunner HG, Nelen MR, van Zandvoort P, Abeling NG, van Gennip AH, Wolters EC, et al. X-linked borderline mental retardation with prominent behavioral disturbance: phenotype, genetic localization, and evidence for disturbed monoamine metabolism. Am J Hum Genet. 1993;52:1032–9.
Caspi A, McClay J, Moffitt TE, Mill J, Martin J, Craig IW, et al. Role of genotype in the cycle of violence in maltreated children. Science. 2002;297:851–4.
Weder N, Yang BZ, Douglas-Palumberi H, Massey J, Krystal JH, Gelernter J, et al. MAOA genotype, maltreatment, and aggressive behavior: the changing impact of genotype at varying levels of trauma. Biol Psychiatry. 2009;65:417–24.
Bevilacqua L, Doly S, Kaprio J, Yuan Q, Tikkanen R, Paunio T, et al. A population-specific HTR2B stop codon predisposes to severe impulsivity. Nature. 2010;468:1061–6.
Tikkanen R, Tiihonen J, Rautiainen MR, Paunio T, Bevilacqua L, Panarsky R, et al. Impulsive alcohol-related risk-behavior and emotional dysregulation among individuals with a serotonin 2B receptor stop codon. Transl Psychiatry. 2015;5:e681.
Karberg JCJ, James D. Substance dependence, abuse, and treatment of jail inmates, 2002. In: U.S. Department of Justice, Office of Justice Programs: Statistics BoJ; 2005. Retrieved from: http://www.bjs.gov/index.cfm?ty=pbdetail&iid=1128.
Solowij N, Jones KA, Rozman ME, Davis SM, Ciarrochi J, Heaven PC, et al. Reflection impulsivity in adolescent cannabis users: a comparison with alcohol-using and non-substance-using adolescents. Psychopharmacology. 2012;219:575–86.
Gruber SA, Dahlgren MK, Sagar KA, Gonenc A, Killgore WD. Age of onset of marijuana use impacts inhibitory processing. Neurosci Lett. 2012;511:89–94.
Bhattacharyya S, Atakan Z, Martin-Santos R, Crippa JA, Kambeitz J, Malhi S, et al. Impairment of inhibitory control processing related to acute psychotomimetic effects of cannabis. Eur Neuropsychopharmacol. 2015;25:26–37.
Renard J,Krebs MO,Le Pen G,Jay TM, Long-term consequences of adolescent cannabinoid exposure in adult psychopathology. Front Neurosci. 2014;8:361.
Barthelemy OJ, Richardson MA, Cabral HJ, Frank DA. Prenatal, perinatal, and adolescent exposure to marijuana: relationships with aggressive behavior. Neurotoxicol Teratol. 2016;58:60–77.
Dugre JR, Dellazizzo L, Giguere CE, Potvin S, Dumais A. Persistency of cannabis use predicts violence following acute psychiatric discharge. Front Psychiatry. 2017;8:176.
Gelernter J, Kranzler HR, Sherva R, Koesterer R, Almasy L, Zhao H, et al. Genome-wide association study of opioid dependence: multiple associations mapped to calcium and potassium pathways. Biol Psychiatry. 2014;76:66–74.
Gelernter J, Sherva R, Koesterer R, Almasy L, Zhao H, Kranzler HR, et al. Genome-wide association study of cocaine dependence and related traits: FAM53B identified as a risk gene. Mol Psychiatry. 2014;19:717–23.
Gelernter J, Kranzler HR, Sherva R, Almasy L, Herman AI, Koesterer R, et al. Genome-wide association study of nicotine dependence in American populations: identification of novel risk loci in both African-Americans and European-Americans. Biol Psychiatry. 2015;77:493–503.
Gelernter J, Kranzler HR, Sherva R, Almasy L, Koesterer R, Smith AH, et al. Genome-wide association study of alcohol dependence:significant findings in African- and European-Americans including novel risk loci. Mol Psychiatry. 2014;19:41–9.
Gillespie CF, Bradley B, Mercer K, Smith AK, Conneely K, Gapen M, et al. Trauma exposure and stress-related disorders in inner city primary care patients. Gen Hosp Psychiatry. 2009;31:505–14.
First MB, Spitzer RL, Gibbon M, JBW W. Structured clinical interview for DSM-IV axis I disorders (SCID-I research version 2.0). New York: Biometrics Research; 1998.
Straus MA, Douglas EM. A short form of the Revised Conflict Tactics Scales, and typologies for severity and mutuality. Violence Vict. 2004;19:507–20.
Price AL, Patterson NJ, Plenge RM, Weinblatt ME, Shadick NA, Reich D. Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet. 2006;38:904–9.
Patterson N, Price AL, Reich D. Population structure and eigenanalysis. PLoS Genet. 2006;2:e190.
Sherva R, Wang Q, Kranzler H, Zhao H, Koesterer R, Herman A, et al. Genome-wide association study of cannabis dependence severity, novel risk variants, and shared genetic risks. JAMA Psychiatry. 2016;73:472–80.
Zeger SL, Liang KY. Longitudinal data analysis for discrete and continuous outcomes. Biometrics. 1986;42:121–30.
Willer CJ, Li Y, Abecasis GR. METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics. 2010;26:2190–1.
Costa PT Jr., McCrae RR. Stability and change in personality assessment: the revised NEO Personality Inventory in the year 2000. J Pers Assess. 1997;68:86–94.
Long LE, Chesworth R, Huang XF, McGregor IS, Arnold JC, Karl T. Transmembrane domain Nrg1 mutant mice show altered susceptibility to the neurobehavioural actions of repeated THC exposure in adolescence. Int J Neuropsychopharmacol. 2013;16:163–75.
Koolhaas JM, Coppens CM, de Boer SF, Buwalda B, Meerlo P, Timmermans PJ. The resident-intruder paradigm: a standardized test for aggression, violence and social stress. J Vis Exp. 2013;77:e4367.
Powers A, Almli L, Smith A, Lori A, Leveille J, Ressler KJ, et al. A genome-wide association study of emotion dysregulation: evidence for interleukin 2 receptor alpha. J Psychiatr Res. 2016;83:195–202.
Muller CP, Homberg JR. The role of serotonin in drug use and addiction. Behav Brain Res. 2015;277:146–92.
Smith PH, Homish GG, Leonard KE, Collins RL. Marijuana withdrawal and aggression among a representative sample of U.S. marijuana users. Drug Alcohol Depend. 2013;132:63–8.
Hikosaka O, Kim HF, Yasuda M, Yamamoto S. Basal ganglia circuits for reward value-guided behavior. Annu Rev Neurosci. 2014;37:289–306.
Bersani FS, Minichino A, Fojanesi M, Gallo M, Maglio G, Valeriani G, et al. Cingulate cortex in schizophrenia: its relation with negative symptoms and psychotic onset. A review study. Eur Rev Med Pharmacol Sci. 2014;18:3354–67.
Fiddick L. There is more than the amygdala: potential threat assessment in the cingulate cortex. Neurosci Biobehav Rev. 2011;35:1007–18.
Pitychoutis PM, Belmer A, Moutkine I, Adrien J, Maroteaux L. Mice lacking the serotonin Htr2B receptor gene present an antipsychotic-sensitive schizophrenic-like phenotype. Neuropsychopharmacology. 2015;40:2764–73.
Diaz SL, Narboux-Neme N, Boutourlinsky K, Doly S, Maroteaux L. Mice lacking the serotonin 5-HT2B receptor as an animal model of resistance to selective serotonin reuptake inhibitors antidepressants. Eur Neuropsychopharmacol. 2016;26:265–79.
Doly S, Bertran-Gonzalez J, Callebert J, Bruneau A, Banas SM, Belmer A, et al. Role of serotonin via 5-HT2B receptors in the reinforcing effects of MDMA in mice. PLoS ONE. 2009;4:e7952.
Doly S, Quentin E, Eddine R, Tolu S, Fernandez SP, Bertran-Gonzalez J, et al. Serotonin 2B receptors in mesoaccumbens dopamine pathway regulate cocaine responses. J Neurosci. 2017;37:10372–88.
Muris P, Meesters C, Blijlevens P. Self-reported reactive and regulative temperament in early adolescence: relations to internalizing and externalizing problem behavior and “Big Three” personality factors. J Adolesc. 2007;30:1035–49.
Trull TJ, Widiger TA. Dimensional models of personality: the five-factor model and the DSM-5. Dialogues Clin Neurosci. 2013;15:135–46.
Need AC, Goldstein DB. Next generation disparities in human genomics: concerns and remedies. Trends Genet. 2009;25:489–94.
Stoops WW, Bennett JA, Lile JA, Sevak RJ, Rush CR. Influence of aripiprazole pretreatment on the reinforcing effects of methamphetamine in humans. Prog Neuropsychopharmacol Biol Psychiatry. 2013;47:111–7.
Chen J, Gao K, Kemp DE. Second-generation antipsychotics in major depressive disorder: update and clinical perspective. Curr Opin Psychiatry. 2011;24:10–7.
Fleischhacker WW, McQuade RD, Marcus RN, Archibald D, Swanink R, Carson WH. A double-blind, randomized comparative study of aripiprazole and olanzapine in patients with schizophrenia. Biol Psychiatry. 2009;65:510–7.
Owen R, Sikich L, Marcus RN, Corey-Lisle P, Manos G, McQuade RD, et al. Aripiprazole in the treatment of irritability in children and adolescents with autistic disorder. Pediatrics. 2009;124:1533–40.
Acknowledgements
We appreciate the work in recruitment and assessment provided at Yale University School of Medicine and the APT Foundation by James Poling, Ph.D.; at McLean Hospital by Roger Weiss, M.D., at the Medical University of South Carolina by Kathleen Brady, M.D., Ph.D., and Raymond Anton, M.D.; and at the University of Pennsylvania by David Oslin, M.D. We are grateful to Ann Marie Lacobelle and Christa Robinson for their excellent technical assistance, to the SSADDA interviewers who devoted substantial time and effort to phenotype the study sample, and to John Farrell and Alexan Mardigan for database management assistance. Assistance with data cleaning was provided by the National Center for Biotechnology Information. This study was supported by National Institutes of Health grants RC2 DA028909, R01 DA12690, R01 DA12849, R01 DA18432, R01 AA11330, R01 AA017535, and the VA Connecticut and Philadelphia VA MIRECCs; the Biological Sciences Training Program through Grant Number 5T32 MH14276 and the NARSAD Young Investigator Grant to JLMO. Funding support for genotyping, which was performed at the Johns Hopkins University Center for Inherited Disease Research, was provided by the NIH GEI (U01HG004438), the National Institute on Alcohol Abuse and Alcoholism, the National Institute on Drug Abuse, and the NIH contract “High-throughput genotyping for studying the genetic contributions to human disease” (HHSN268200782096C). LM and ID’A have been supported by funds from the Centre National de la Recherche Scientifique, the Institut National de la Santé et de la Recherche Médicale, the Université Pierre et Marie Curie, and by grants from the Fondation pour la Recherche Médicale “Equipe FRM DEQ2014039529”, the French Ministry of Research (Agence Nationale pour la Recherche ANR-12-BSV1-0015 and ANR-17-CE16-0008 and the Investissements d’Avenir programme ANR-11-IDEX-0004-02). LM’s team is part of the École des Neurosciences de Paris Ile-de-France network and of the Bio-Psy Labex and as such this work was supported by French state funds managed by the ANR within the Investissements d’Avenir programme under reference ANR-11-IDEX-0004-02.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Although unrelated to the current study, HRK has been a consultant, advisory board member, or CME speaker for Indivior and Lundbeck. He is also a member of the American Society of Clinical Psychopharmacology’s Alcohol Clinical Trials Initiative, which in the last 3 years was supported by Abbvie, Alkermes, Amygdala Neurosciences, Arbor, Ethypharm, Indivior, Lilly, Lundbeck, Otsuka, and Pfizer. The remaining authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Montalvo-Ortiz, J., Zhou, H., D’Andrea, I. et al. Translational studies support a role for serotonin 2B receptor (HTR2B) gene in aggression-related cannabis response. Mol Psychiatry 23, 2277–2286 (2018). https://doi.org/10.1038/s41380-018-0077-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-018-0077-6
This article is cited by
-
SP1 transcriptionally activates HTR2B to aggravate traumatic spinal cord injury by shifting microglial M1/M2 polarization
Journal of Orthopaedic Surgery and Research (2024)
-
Htr2b Promotes M1 Microglia Polarization and Neuroinflammation after Spinal Cord Injury via Inhibition of Neuregulin-1/ErbB Signaling
Molecular Neurobiology (2024)
-
Cannabis consumption and prosociality
Scientific Reports (2022)
-
Association of serotonin system-related genes with homicidal behavior and criminal aggression in a prison population of Pakistani Origin
Scientific Reports (2021)