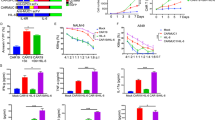
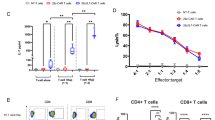
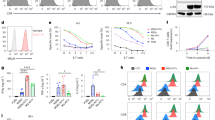
Abstract
CAR-T cell therapy has emerged as a breakthrough therapy for the treatment of relapsed and refractory hematologic malignancies. However, insufficient CAR-T cell expansion and persistence is a leading cause of treatment failure. Exogenous or transgenic cytokines have great potential to enhance CAR-T cell potency but pose the risk of exacerbating toxicities. Here we present a chemical-genetic system for spatiotemporal control of cytokine function gated by the off-patent anti-cancer molecular glue degrader drug lenalidomide and its analogs. When co-delivered with a CAR, a membrane-bound, lenalidomide-degradable IL-7 fusion protein enforced a clinically favorable T cell phenotype, enhanced antigen-dependent proliferative capacity, and enhanced in vivo tumor control. Furthermore, cyclical pharmacologic combined control of CAR and cytokine abundance enabled the deployment of highly active, IL-7-augmented CAR-T cells in a dual model of antitumor potency and T cell hyperproliferation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
For original data, please contact mvmaus@mgh.harvard.edu.
References
Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377:2531–44.
Maude SL, Laetsch TW, Buechner J, Rives S, Boyer M, Bittencourt H, et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N Engl J Med. 2018;378:439–48.
Shah NN, Fry TJ. Mechanisms of resistance to CAR T cell therapy. Nat Rev Clin Oncol. 2019;16:372–85.
Majzner RG, Mackall CL. Clinical lessons learned from the first leg of the CAR T cell journey. Nat Med. 2019;25:1341–55.
Guedan S, Calderon H, Posey AD, Maus MV. Engineering and design of chimeric antigen receptors. Mol Ther Methods Clin Dev. 2019;12:145–56.
Alizadeh D, Wong RA, Yang X, Wang D, Pecoraro JR, Kuo C-F, et al. IL15 enhances CAR-T cell antitumor activity by reducing mTORC1 activity and preserving their stem cell memory phenotype. Cancer Immunol Res. 2019;7:759–72.
Agliardi G, Liuzzi AR, Hotblack A, De Feo D, Núñez N, Stowe CL, et al. Intratumoral IL-12 delivery empowers CAR-T cell immunotherapy in a pre-clinical model of glioblastoma. Nat Commun. 2021;12:444.
Bell M, Gottschalk S. Engineered cytokine signaling to improve CAR T cell effector function. Front Immunol. 2021;12:684642.
Zhang L, Morgan RA, D.Beane J, Zheng Z, Dudley ME, Kassim SH, et al. Tumor infiltrating lymphocytes genetically engineered with an inducible gene encoding interleukin-12 for the immunotherapy of metastatic melanoma. Clin Cancer Res J Am Assoc Cancer Res. 2015;21:2278–88.
Chmielewski M, Abken H. CAR T cells releasing IL-18 convert to T-bethigh FoxO1low effectors that exhibit augmented activity against advanced solid tumors. Cell Rep. 2017;21:3205–19.
Shum T, Omer B, Tashiro H, Kruse RL, Wagner DL, Parikh K, et al. Constitutive signaling from an engineered IL-7 receptor promotes durable tumor elimination by tumor redirected T-cells. Cancer Discov. 2017;7:1238–47.
Aspuria P-J, Vivona S, Bauer M, Semana M, Ratti N, McCauley S, et al. An orthogonal IL-2 and IL-2Rβ system drives persistence and activation of CAR T cells and clearance of bulky lymphoma. Sci Transl Med. 2021;13:eabg7565.
Zhang Q, Hresko ME, Picton LK, Su L, Hollander MJ, Nunez-Cruz S, et al. A human orthogonal IL-2 and IL-2Rβ system enhances CAR T cell expansion and antitumor activity in a murine model of leukemia. Sci Transl Med. 2021;13:eabg6986.
Neelapu SS, Tummala S, Kebriaei P, Wierda W, Gutierrez C, Locke FL, et al. Chimeric antigen receptor T-cell therapy — assessment and management of toxicities. Nat Rev Clin Oncol. 2018;15:47–62.
Lee DW, Santomasso BD, Locke FL, Ghobadi A, Turtle CJ, Brudno JN, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transplant. 2019;25:625–38.
Mackall CL, Fry TJ, Gress RE. Harnessing the biology of IL-7 for therapeutic application. Nat Rev Immunol 2011;11:330–42.
Schluns KS, Kieper WC, Jameson SC, Lefrançois L. Interleukin-7 mediates the homeostasis of naïve and memory CD8 T cells in vivo. Nat Immunol. 2000;1:426–32.
Adachi K, Kano Y, Nagai T, Okuyama N, Sakoda Y, Tamada K. IL-7 and CCL19 expression in CAR-T cells improves immune cell infiltration and CAR-T cell survival in the tumor. Nat Biotechnol. 2018;36:346–51.
Zhou J, Jin L, Wang F, Zhang Y, Liu B, Zhao T. Chimeric antigen receptor T (CAR-T) cells expanded with IL-7/IL-15 mediate superior antitumor effects. Protein Cell. 2019;10:764–9.
Thomas KR, Allenspach EJ, Camp ND, Wray-Dutra MN, Khim S, Zielinska-Kwiatkowska A, et al. Activated interleukin-7 receptor signaling drives B-cell acute lymphoblastic leukemia in mice. Leukemia. 2022;36:42–57.
Zenatti PP, Ribeiro D, Li W, Zuurbier L, Silva MC, Paganin M, et al. Oncogenic IL7R gain-of-function mutations in childhood T-cell acute lymphoblastic leukemia. Nat Genet. 2011;43:932–9.
Abraham N, Ma MC, Snow JW, Miners MJ, Herndier BG, Goldsmith MA. Haploinsufficiency identifies STAT5 as a modifier of IL-7-induced lymphomas. Oncogene. 2005;24:5252–7.
Bailey, SR, Vatsa, S, Larson, RC, Bouffard, AA, Scarfo, I, Kann, MC, et al. Blockade or deletion of IFNg reduces macrophage activation without compromising CAR-T function in hematologic malignancies. Blood Cancer Discov. 2021. https://doi.org/10.1158/2643-3230.BCD-21-0181
Jan M, Scarfò I, Larson RC, Walker A, Schmidts A, Guirguis AA, et al. Reversible ON- and OFF-switch chimeric antigen receptors controlled by lenalidomide. Sci Transl Med. 2021;13:eabb6295.
Scarfò I, Ormhøj M, Frigault MJ, Castano AP, Lorrey S, Bouffard AA, et al. Anti-CD37 chimeric antigen receptor T cells are active against B- and T-cell lymphomas. Blood. 2018;132:1495–506.
Sreekanth, V, Jan, M, Zhao, KT, Lim, D, Davis, JR, McConkey, M, et al. A molecular glue approach to control the half-life of CRISPR-based technologies. 2023. Preprint at https://doi.org/10.1101/2023.03.12.531757.
Liu Z, Chen O, Wall JBJ, Zheng M, Zhou Y, Wang L, et al. Systematic comparison of 2A peptides for cloning multi-genes in a polycistronic vector. Sci Rep. 2017;7:2193.
Schmidts A, Marsh LC, Srivastava AA, Bouffard AA, Boroughs AC, Scarfò I, et al. Cell-based artificial APC resistant to lentiviral transduction for efficient generation of CAR-T cells from various cell sources. J Immunother Cancer. 2020;8:e000990.
Geng C-L, Chen J-Y, Song T-Y, Jung JH, Long M, Song M-F, et al. Lenalidomide bypasses CD28 co-stimulation to reinstate PD-1 immunotherapy by activating Notch signaling. Cell Chem Biol 2022;29:1260–.e8.
Larson RC, Castano A, Bouffard AA, Kann MC, Schmidts A, Gallagher KM, et al. Abstract 556: Novel anti-TACI single and dual-targeting CAR T cells overcome BCMA antigen loss in multiple myeloma. Cancer Res. 2022;82:556.
Li S, Pal R, Monaghan SA, Schafer P, Ouyang H, Mapara M, et al. IMiD immunomodulatory compounds block C/EBPβ translation through eIF4E down-regulation resulting in inhibition of MM. Blood. 2011;117:5157–65.
Zhu YX, Shi C-X, Bruins LA, Wang X, Riggs DL, Porter B, et al. Identification of lenalidomide resistance pathways in myeloma and targeted resensitization using cereblon replacement, inhibition of STAT3 or targeting of IRF4. Blood. Cancer J. 2019;9:1–12.
Liu K, Zhang D, Chojnacki J, Du Y, Fu H, Grant S, et al. Design and biological characterization of hybrid compounds of curcumin and thalidomide for multiple myeloma. Org Biomol Chem 2013;11:4757–63.
Holstein SA, Tong H, Hohl RJ. Differential activities of thalidomide and isoprenoid biosynthetic pathway inhibitors in multiple myeloma cells. Leuk Res 2010;34:344–51.
Mestermann K, Giavridis T, Weber J, Rydzek J, Frenz S, Nerreter T, et al. The tyrosine kinase inhibitor dasatinib acts as a pharmacologic on/off switch for CAR T cells. Sci Transl Med. 2019;11:eaau5907.
Weber EW, Parker KR, Sotillo E, Lynn RC, Anbunathan H, Lattin J, et al. Transient rest restores functionality in exhausted CAR-T cells through epigenetic remodeling. Science. 2021;372:eaba1786.
Kim MY, Jayasinghe R, Devenport JM, Ritchey JK, Rettig MP, O’Neal J, et al. A long-acting interleukin-7, rhIL-7-hyFc, enhances CAR T cell expansion, persistence, and anti-tumor activity. Nat Commun. 2022;13:3296.
Li L, Li Q, Yan Z-X, Sheng L-S, Fu D, Xu P, et al. Transgenic expression of IL-7 regulates CAR-T cell metabolism and enhances in vivo persistence against tumor cells. Sci Rep. 2022;12:12506.
Barata JT, Durum SK, Seddon B. Flip the coin: IL-7 and IL-7R in health and disease. Nat Immunol 2019;20:1584–93.
Hucks G, Rheingold SR. The journey to CAR T cell therapy: the pediatric and young adult experience with relapsed or refractory B-ALL. Blood Cancer J. 2019;9:10.
Nie Y, Lu W, Chen D, Tu H, Guo Z, Zhou X, et al. Mechanisms underlying CD19-positive ALL relapse after anti-CD19 CAR T cell therapy and associated strategies. Biomark Res. 2020;8:18.
Neelapu SS. CAR-T efficacy: is conditioning the key? Blood. 2019;133:1799–1800.
Hirayama AV, Gauthier J, Hay KA, Voutsinas JM, Wu Q, Gooley T, et al. The response to lymphodepletion impacts PFS in patients with aggressive non-Hodgkin lymphoma treated with CD19 CAR T cells. Blood. 2019;133:1876–87.
Korell F, Schubert M-L, Sauer T, Schmitt A, Derigs P, Weber TF, et al. Infection complications after lymphodepletion and dosing of chimeric antigen receptor T (CAR-T) cell therapy in patients with relapsed/refractory acute lymphoblastic leukemia or B cell non-hodgkin lymphoma. Cancers. 2021;13:1684.
Melenhorst JJ, Chen GM, Wang M, Porter DL, Chen C, Collins MA, et al. Decade-long leukaemia remissions with persistence of CD4+ CAR T cells. Nature. 2022;602:503–9.
Busch DH, Fräßle SP, Sommermeyer D, Buchholz VR, Riddell SR. Role of memory T cell subsets for adoptive immunotherapy. Semin Immunol. 2016;28:28–34.
Blaeschke F, Stenger D, Kaeuferle T, Willier S, Lotfi R, Kaiser AD, et al. Induction of a central memory and stem cell memory phenotype in functionally active CD4+ and CD8+ CAR T cells produced in an automated good manufacturing practice system for the treatment of CD19+ acute lymphoblastic leukemia. Cancer Immunol Immunother CII. 2018;67:1053–66.
Tantalo, Oliver DG, Scheidt AJ, von B, Harrison AJ, Mueller SN, et al. Understanding T cell phenotype for the design of effective chimeric antigen receptor T cell therapies. J Immunother Cancer. 2021;9:e002555.
Krönke J, Fink EC, Hollenbach PW, MacBeth KJ, Hurst SN, Udeshi ND, et al. Lenalidomide induces ubiquitination and degradation of CK1α in del(5q) MDS. Nature. 2015;523:183–8.
Watanabe K, Terakura S, Martens AC, van Meerten T, Uchiyama S, Imai M, et al. Target antigen density governs the efficacy of anti–CD20-CD28-CD3 ζ chimeric antigen receptor–modified effector CD8+ T cells. J Immunol 2015;194:911–20.
Arcangeli S, Rotiroti MC, Bardelli M, Simonelli L, Magnani CF, Biondi A, et al. Balance of anti-CD123 chimeric antigen receptor binding affinity and density for the targeting of acute myeloid leukemia. Mol Ther 2017;25:1933–45.
Walker AJ, Majzner RG, Zhang L, Wanhainen K, Long AH, Nguyen SM, et al. Tumor antigen and receptor densities regulate efficacy of a chimeric antigen receptor targeting anaplastic lymphoma kinase. Mol Ther 2017;25:2189–201.
Gandhi AK, Kang J, Havens CG, Conklin T, Ning Y, Wu L, et al. Immunomodulatory agents lenalidomide and pomalidomide co-stimulate T cells by inducing degradation of T cell repressors Ikaros and Aiolos via modulation of the E3 ubiquitin ligase complex CRL4(CRBN). Br J Haematol. 2014;164:811–21.
Krönke J, Udeshi ND, Narla A, Grauman P, Hurst SN, McConkey M, et al. Lenalidomide causes selective degradation of IKZF1 and IKZF3 in multiple myeloma cells. Science. 2014;343:301–5.
Jan M, Sperling AS, Ebert BL. Cancer therapies based on targeted protein degradation - lessons learned with lenalidomide. Nat Rev Clin Oncol. 2021;18:401–17.
Works M, Soni N, Hauskins C, Sierra C, Baturevych A, Jones JC, et al. Anti-B-cell maturation antigen chimeric antigen receptor T cell function against multiple myeloma is enhanced in the presence of lenalidomide. Mol Cancer Ther. 2019;18:2246–57.
Kuramitsu S, Ohno M, Ohka F, Shiina S, Yamamichi A, Kato A, et al. Lenalidomide enhances the function of chimeric antigen receptor T cells against the epidermal growth factor receptor variant III by enhancing immune synapses. Cancer Gene Ther. 2015;22:487–95.
Carbonneau S, Sharma S, Peng L, Rajan V, Hainzl D, Henault M, et al. An IMiD-inducible degron provides reversible regulation for chimeric antigen receptor expression and activity. Cell Chem Biol 2021;28:802–.e6.
Chakravarti D, Caraballo LD, Weinberg BH, Wong WW. Inducible gene switches with memory in human T cells for cellular immunotherapy. ACS Synth Biol. 2019;8:1744–54.
Nguyen, TM, Deb, A, Kokkonda, P, Sreekanth, V, Tiwari, PK, Shoba, V, et al. Proteolysis targeting chimeras with reduced Off-targets. 2021. Preprint at https://doi.org/10.1101/2021.11.18.468552.
Smole A, Benton A, Poussin MA, Eiva MA, Mezzanotte C, Camisa B, et al. Expression of inducible factors reprograms CAR-T cells for enhanced function and safety. Cancer Cell. 2022;40:1470–87.e7.
Allen GM, Frankel NW, Reddy NR, Bhargava HK, Yoshida MA, Stark SR, et al. Synthetic cytokine circuits that drive T cells into immune-excluded tumors. Science. 2022;378:eaba1624.
Sockolosky JT, Trotta E, Parisi G, Picton L, Su LL, Le AC, et al. Selective targeting of engineered T cells using orthogonal IL-2 cytokine-receptor complexes. Science. 2018;359:1037–42.
Cook, PJ, Yang, SJ, Uenishi, GI, Grimm, A, West, SE, Wang, L-J, et al. A chemically inducible IL-2 receptor signaling complex allows for effective in vitro and in vivo selection of engineered CD4+ T cells. Mol. Ther. J. Am. Soc. Gene Ther. 2023:S1525-0016 00255–1. https://doi.org/10.1016/j.ymthe.2023.04.021.
Lin RJ, Nager AR, Park S, Sutton J, Lay C, Melton Z, et al. Design and validation of inducible TurboCARs with tunable induction and combinatorial cytokine signaling. Cancer Immunol Res. 2022;10:1069–83.
Richman SA, Wang L-C, Moon EK, Khire UR, Albelda SM, Milone MC. Ligand-induced degradation of a CAR permits reversible remote control of CAR T cell activity in vitro and in vivo. Mol Ther. 2020;28:1600–13.
Nakao S, Arai Y, Tasaki M, Yamashita M, Murakami R, Kawase T, et al. Intratumoral expression of IL-7 and IL-12 using an oncolytic virus increases systemic sensitivity to immune checkpoint blockade. Sci Transl Med. 2020;12:eaax7992.
Chiocca EA, Yu JS, Lukas RV, Solomon IH, Ligon KL, Nakashima H, et al. Regulatable interleukin-12 gene therapy in patients with recurrent high-grade glioma: Results of a phase 1 trial. Sci Transl Med. 2019;11:eaaw5680.
Acknowledgements
This work was supported by R01CA238268 to MVM and NIH K08CA255932, Leukemia Research Foundation New Investigator Award, Damon Runyon Cancer Research Foundation Innovator Award, American Cancer Society Discovery Boost Grant, and Edward P. Evans Foundation Discovery Research Grant to MJ. Select figures created with BioRender.com.
Author information
Authors and Affiliations
Contributions
MCK, EMS, AJA, ICL, AAB, VMS, HNT, AT, and MJ performed the experiments; MCK and MJ analyzed the results and created the figures; RCL, MBL, and BLE provided critical expertise in immune-oncology and targeted protein degradation; MCK, MVM, and MJ designed the study; MCK, MVM, and MJ wrote the manuscript; and all other authors read, edited, and approved the final version of this manuscript.
Corresponding author
Ethics declarations
Competing interests
MCK, MVM, and MJ are listed as inventors on patents related to this work and held by the Massachusetts General Hospital and Partners Health Care. BLE has received research funding from Celgene, Deerfield, Novartis, and Calico and consulting fees from GRAIL. He is a member of the scientific advisory board and shareholder for Neomorph Inc., TenSixteen Bio, Skyhawk Therapeutics, and Exo Therapeutics. MVM is an inventor on patents related to adoptive cell therapies, held by Massachusetts General Hospital (some licensed to Promab) and University of Pennsylvania (some licensed to Novartis). MVM holds equity in 2SeventyBio, Century Therapeutics, Genocea, Neximmune, Oncternal, and TCR2 and has served as a consultant for multiple companies involved in cell therapies. MJ has received consulting income from RA Ventures. MJ is an inventor on patents related to molecular switch control of genetically engineered immune effector cell therapies held by Mass General Brigham and the Broad Institute.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kann, M.C., Schneider, E.M., Almazan, A.J. et al. Chemical genetic control of cytokine signaling in CAR-T cells using lenalidomide-controlled membrane-bound degradable IL-7. Leukemia 38, 590–600 (2024). https://doi.org/10.1038/s41375-023-02113-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-023-02113-6