Abstract
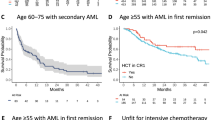
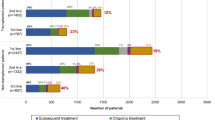
Eligibility criteria for clinical trials are intended to select suitable study subjects but can limit trial participation and generalization of results. While reported for other cancers, non-enrollment rates and evolution of eligibility criteria over time have so far not been studied for randomized controlled trials (RCTs) involving adults with acute myeloid leukemia (AML). Among 3698 studies published between 2010 and 2020, including 447 involving prospective clinical trials, we identified 75 phase three RCTs testing non-transplant therapies for adults with AML. Only 31 studies (41%) provided information on non-enrollment; in these studies, the median non-enrollment rate was 23%, primarily attributed to restrictive eligibility criteria. In 95% of trials, eligibility criteria were reported with the total number per trial increasing over time (P < 0.001), particularly in industry-funded trials. A total of 27 eligibility criteria were used across trials, mostly concerning comorbidities or performance status, with eight of them becoming more common over time. The concordance with recent ASCO – Friends of Cancer Research eligibility criteria recommendations greatly varied, from 35% to 99%. Together, our analyses suggest that the ability to generalize results from non-transplant RCTs may be increasingly limited because of high non-enrollment rates and increasingly restrictive eligibility criteria.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Giantonio BJ. Eligibility in cancer clinical research: the intersection of discovery, generalizability, beneficence, and justice. Clin Cancer Res. 2021;27:2369–71.
Srikanthan A, Vera-Badillo F, Ethier J, Goldstein R, Templeton AJ, Ocana A, et al. Evolution in the eligibility criteria of randomized controlled trials for systemic cancer therapies. Cancer Treat Rev. 2016;43:67–73.
Garcia S, Bisen A, Yan J, Xie X-J, Ramalingam S, Schiller JH, et al. Thoracic oncology clinical trial eligibility criteria and requirements continue to increase in number and complexity. J Thorac Oncol. 2017;12:1489–95.
Unger JM, Vaidya R, Hershman DL, Minasian LM, Fleury ME. Systematic review and meta-analysis of the magnitude of structural, clinical, and physician and patient barriers to cancer clinical trial participation. JNCI J Natl Cancer Inst. 2019;111:245–55.
Unger JM, Hershman DL, Fleury ME, Vaidya R. Association of patient comorbid conditions with cancer clinical trial participation. JAMA Oncol. 2019;5:326.
Harvey RD, Bruinooge SS, Chen L, Garrett-Mayer E, Rhodes W, Stepanski E, et al. Impact of broadening trial eligibility criteria for patients with advanced non–small cell lung cancer: real-world analysis of select ASCO- Friends recommendations. Clin Cancer Res. 2021;27:2430–4.
Gross CP, Mallory R, Heiat A, Krumholz HM. Reporting the recruitment process in clinical trials: who are these patients and how did they get there? Ann Intern Med. 2002;137:10–6.
He Z, Tang X, Yang X, Guo Y, George TJ, Charness N, et al. Clinical trial generalizability assessment in the big data era: a review. Clin Transl Sci. 2020;13:675–84.
Humphreys K, Maisel NC, Blodgett JC, Fuh IL, Finney JW. Extent and reporting of patient nonenrollment in influential randomized clinical trials, 2002 to 2010. JAMA Intern Med. 2013;173:1029.
Humphreys K, Maisel NC, Blodgett JC, Finney JW. Representativeness of patients enrolled in influential clinical trials: a comparison of substance dependence with other medical disorders. J Stud Alcohol Drugs. 2013;74:889–93.
Loh Z, Salvaris R, Chong G, Churilov L, Manos K, Barraclough A, et al. Evolution of eligibility criteria for diffuse large B‐cell lymphoma randomised controlled trials over 30 years. Br J Haematol. 2021;193:741–9.
Van Spall HG, Toren A, Kiss A, Fowler RA. Eligibility criteria of randomized controlled trials published in high-impact general medical journals: a systematic sampling review. JAMA. 2007;297:1233–40.
Khurana A, Mwangi R, Nowakowski GS, Habermann TM, Ansell SM, LaPlant BR, et al. Impact of organ function–based clinical trial eligibility criteria in patients with diffuse large b-cell lymphoma: who gets left behind? J Clin Oncol. 2021;39:1641–9.
Winters AC, Gutman JA, Purev E, Nakic M, Tobin J, Chase S, et al. Real-world experience of venetoclax with azacitidine for untreated patients with acute myeloid leukemia. Blood Adv. 2019;3:2911–9.
Kim ES, Bruinooge SS, Roberts S, Ison G, Lin NU, Gore L, et al. Broadening eligibility criteria to make clinical trials more representative: american society of clinical oncology and friends of cancer research joint research statement. J Clin Oncol. 2017;35:3737–44.
Kim ES, Uldrick TS, Schenkel C, Bruinooge SS, Harvey RD, Magnuson A, et al. Continuing to broaden eligibility criteria to make clinical trials more representative and inclusive: asco–friends of cancer research joint research statement. Clin Cancer Res. 2021;27:2394–9.
Booth CM, Le Maître A, Ding K, Farn K, Fralick M, Phillips C, et al. Presentation of nonfinal results of randomized controlled trials at major oncology meetings. J Clin Oncol. 2009;27:3938–44.
Del Paggio JC, Berry JS, Hopman WM, Eisenhauer EA, Prasad V, Gyawali B, et al. Evolution of the randomized clinical trial in the era of precision oncology. JAMA Oncol. 2021;7:728.
Buckley SA, Percival M-E, Othus M, Halpern AB, Huebner EM, Becker PS, et al. A comparison of patients with acute myeloid leukemia and high-risk myelodysplastic syndrome treated on versus off study. Leuk Lymphoma. 2019;60:1023–9.
Percival M-EM, Othus M, Mirahsani S, Gardner KM, Shaw C, Halpern AB et al. Survival of patients with newly diagnosed high-grade myeloid neoplasms who do not meet standard trial eligibility. Haematologica. 2021;106:2114–20.
Statler A, Radivoyevitch T, Siebenaller C, Gerds AT, Kalaycio M, Kodish E, et al. The relationship between eligibility criteria and adverse events in randomized controlled trials of hematologic malignancies. Leukemia. 2017;31:1808–15.
Statler A, Othus M, Erba HP, Chauncey TR, Radich JP, Coutre S, et al. Comparable outcomes of patients eligible vs ineligible for SWOG leukemia studies. Blood. 2018;131:2782–8.
Lichtman SM, Harvey RD, Damiette Smit M-A, Rahman A, Thompson MA, Roach N, et al. Modernizing clinical trial eligibility criteria: recommendations of the american society of clinical oncology–friends of cancer research organ dysfunction, prior or concurrent malignancy, and comorbidities working group. J Clin Oncol. 2017;35:3753–9.
Liu R, Rizzo S, Whipple S, Pal N, Pineda AL, Lu M, et al. Evaluating eligibility criteria of oncology trials using real-world data and AI. Nature. 2021;592:629–33.
Acknowledgements
The study was originally conceived and designed with Professor Elihu H. Estey who passed away before it could be completed. We are thankful for his mentorship on this work and so many others.
Author information
Authors and Affiliations
Contributions
Conception and Design: CO, RBW. Collection and Assembly of Data: CO, GJ. Data Analysis and Interpretation: CO, MO, MHB, FRA, RBW. Manuscript Writing: All authors. Final Approval of the Manuscript: All authors.
Corresponding author
Ethics declarations
Competing interests
Employment or Leadership Position: none; Consultant or Advisory Role: MHB, Erytech©; Stock Ownership: none; Honoraria: CO, Novartis, MHB, Abbvie, Incyte, and Jazz Pharmaceuticals; Research Funding: none; Expert Testimony: none; Patents: none; Other Remuneration: none.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Orvain, C., Othus, M., Johal, G. et al. Evolution of eligibility criteria for non-transplant randomized controlled trials in adults with acute myeloid leukemia. Leukemia 36, 2002–2008 (2022). https://doi.org/10.1038/s41375-022-01624-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-022-01624-y