Abstract
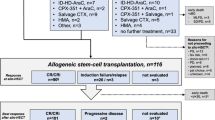
CPX-351 is a liposomal formulation of cytarabine/daunorubicin with a 5:1 fixed molar ratio. We investigated the safety and efficacy of escalating doses of CPX-351 in patients with acute myeloid leukemia (AML) at high risk of induction mortality with standard chemotherapy determined through assessment of leukemia and patient-related risk factors for intensive chemotherapy in an open-label, phase II trial. Patients were randomized to receive 50 or 75 units/m2 on days 1, 3, and 5. Once safety was established, a 100 units/m2 arm was opened. Fifty-six patients were enrolled, 16, 24, and 16 in the 50, 75, and 100 units/m2 arms, respectively. The composite complete remission rate (complete remission + complete remission with incomplete blood count recovery) was lowest with 50 units/m2 (19%) compared with 75 units/m2 (38%) and 100 units/m2 (44%) (P = 0.35). The 50 units/m2 arm had a median OS of 4.3 months, compared with 8.6 and 6.2 months for the 75 and 100 units/m2 respectively (P = 0.04). Nonhematologic grade 3/4 treatment-emergent adverse events included febrile neutropenia (34%), pneumonia (23%), and sepsis (16%). CPX-351 at 75 units/m2 has favorable safety and efficacy for AML patients at high risk of induction mortality with some tolerating the standard dose of 100 units/m2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Appelbaum FR, Gundacker H, Head DR, Slovak ML, Willman CL, Godwin JE, et al. Age and acute myeloid leukemia. Blood. 2006;107:3481–5.
Østgård LSG, Medeiros BC, Sengeløv H, Nørgaard M, Andersen MK, Dufva IH, et al. Epidemiology and clinical significance of secondary and therapy-related acute myeloid leukemia: a national population-based cohort study. J Clin Oncol. 2015;33:3641–9.
Leith CP, Kopecky KJ, Godwin J, McConnell T, Slovak ML, Chen IM, et al. Acute myeloid leukemia in the elderly: assessment of multidrug resistance (MDR1) and cytogenetics distinguishes biologic subgroups with remarkably distinct responses to standard chemotherapy. A Southwest Oncology Group study. Blood. 1997;89:3323–9.
Grimwade D, Hills RK, Moorman AV, Walker H, Chatters S, Goldstone AH, et al. Refinement of cytogenetic classification in acute myeloid leukemia: determination of prognostic significance of rare recurring chromosomal abnormalities among 5876 younger adult patients treated in the United Kingdom Medical Research Council trials. Blood. 2010;116:354–65.
Kantarjian H, O’Brien S, Cortes J, Giles F, Faderl S, Jabbour E, et al. Results of intensive chemotherapy in 998 patients age 65 years or older with acute myeloid leukemia or high-risk myelodysplastic syndrome: predictive prognostic models for outcome. Cancer. 2006;106:1090–8.
Othus M, Kantarjian H, Petersdorf S, Ravandi F, Godwin J, Cortes J, et al. Declining rates of treatment-related mortality in patients with newly diagnosed AML given ‘intense’ induction regimens: a report from SWOG and MD Anderson. Leukemia. 2014;28:289–92.
Lim WS, Tardi PG, Dos Santos N, Xie X, Fan M, Liboiron BD, et al. Leukemia-selective uptake and cytotoxicity of CPX-351, a synergistic fixed-ratio cytarabine:daunorubicin formulation, in bone marrow xenografts. Leuk Res. 2010;34:1214–23.
Kim HP, Gerhard B, Harasym TO, Mayer LD, Hogge DE. Liposomal encapsulation of a synergistic molar ratio of cytarabine and daunorubicin enhances selective toxicity for acute myeloid leukemia progenitors as compared to analogous normal hematopoietic cells. Exp Hematol. 2011;39:741–50.
Tardi P, Johnstone S, Harasym N, Xie S, Harasym T, Zisman N, et al. In vivo maintenance of synergistic cytarabine:daunorubicin ratios greatly enhances therapeutic efficacy. Leuk Res. 2009;33:129–39.
Lancet JE, Uy GL, Cortes JE, Newell LF, Lin TL, Ritchie EK, et al. CPX-351 (cytarabine and daunorubicin) liposome for injection versus conventional cytarabine plus daunorubicin in older patients with newly diagnosed secondary acute myeloid leukemia. J Clin Oncol. 2018;36:2684–92.
Döhner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Büchner T, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2017;129:424–47.
Feldman EJ, Lancet JE, Kolitz JE, Ritchie EK, Roboz GJ, List AF, et al. First-in-man study of CPX-351: a liposomal carrier containing cytarabine and daunorubicin in a fixed 5:1 molar ratio for the treatment of relapsed and refractory acute myeloid leukemia. J Clin Oncol. 2011;29:979–85.
Xu J, Jorgensen JL, Wang SA. How do we use multicolor flow cytometry to detect minimal residual disease in acute myeloid leukemia? Clin Lab Med. 2017;37:787–802.
Luthra R, Patel KP, Reddy NG, Haghshenas V, Routbort MJ, Harmon MA, et al. Next-generation sequencing-based multigene mutational screening for acute myeloid leukemia using MiSeq: applicability for diagnostics and disease monitoring. Haematologica. 2014;99:465–73.
Thall PF, Simon RM, Estey EH. Bayesian sequential monitoring designs for single-arm clinical trials with multiple outcomes. Stat Med. 1995;14:357–79.
Ball B, Komrokji RS, Ades L, Sekeres MA, DeZern AE, Pleyer L, et al. Evaluation of induction chemotherapies after hypomethylating agent failure in myelodysplastic syndromes and acute myeloid leukemia. Blood Adv. 2018;2:2063–71.
Jabbour E, Garcia-Manero G, Batty N, Shan J, O’Brien S, Cortes J, et al. Outcome of patients with myelodysplastic syndrome after failure of decitabine therapy. Cancer. 2010;116:3830–4.
Cortes JE, Heidel FH, Hellmann A, Fiedler W, Smith BD, Robak T, et al. Randomized comparison of low dose cytarabine with or without glasdegib in patients with newly diagnosed acute myeloid leukemia or high-risk myelodysplastic syndrome. Leukemia. 2019;33:379–89.
Döhner H, Lübbert M, Fiedler W, Fouillard L, Haaland A, Brandwein JM, et al. Randomized, phase 2 trial of low-dose cytarabine with or without volasertib in AML patients not suitable for induction therapy. Blood 2014;124:1426–33.
Sekeres MA, Lancet JE, Wood BL, Grove LE, Sandalic L, Sievers EL, et al. Randomized, phase IIb study of low-dose cytarabine and lintuzumab <em>versus</em> low-dose cytarabine and placebo in older adults with untreated acute myeloid leukemia. Haematologica. 2013;98:119–28.
DeAngelo DJ, Sekeres MA, Ottmann OG, Sanz MA, Naoe T, Taube T, et al. Phase III randomized trial of volasertib combined with low-dose cytarabine (LDAC) versus placebo plus LDAC in patients aged ≥65 years with previously untreated, acute myeloid leukemia (AML) ineligible for intensive remission induction therapy. Clin Lymphoma, Myeloma Leuk. 2015;15:S194.
Heidel F, Cortes J, Rucker FG, Aulitzky W, Letvak L, Kindler T, et al. Results of a multicenter phase II trial for older patients with c-Kit-positive acute myeloid leukemia (AML) and high-risk myelodysplastic syndrome (HR-MDS) using low-dose Ara-C and Imatinib. Cancer. 2007;109:907–14.
Walter RB, Othus M, Orlowski KF, McDaniel EN, Scott BL, Becker PS, et al. Unsatisfactory efficacy in randomized study of reduced-dose CPX-351 for medically less fit adults with newly diagnosed acute myeloid leukemia or other high-grade myeloid neoplasm. Haematologica. 2018;103:e106–e9.
Walter RB, Othus M, Borthakur G, Ravandi F, Cortes JE, Pierce SA, et al. Prediction of Early Death After Induction Therapy for Newly Diagnosed Acute Myeloid Leukemia With Pretreatment Risk Scores: A Novel Paradigm for Treatment Assignment. J Clin Oncol. 2011;29:4417–24.
DiNardo CD, Pratz KW, Letai A, Jonas BA, Wei AH, Thirman M, et al. Safety and preliminary efficacy of venetoclax with decitabine or azacitidine in elderly patients with previously untreated acute myeloid leukaemia: a non-randomised, open-label, phase 1b study. Lancet Oncol. 2018;19:216–28.
Wei AH, Strickland SA Jr., Hou JZ, Fiedler W, Lin TL, Walter RB, et al. Venetoclax combined with low-dose cytarabine for previously untreated patients with acute myeloid leukemia: results from a phase Ib/II Study. J Clin Oncol. 2019;37:1277–84.
Acknowledgements
GCI received funding through the K12 Paul Calabresi Clinical Scholarship Award (NIH/NCI K12 CA088084).
Funding
The study was supported in part by Celator/Jazz, and by the Cancer Center Support Grant (NCI Grant P30 CA016672).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
GCI received research funding from Celgene and served on an advisory board for Novartis. HMK received research funding from Ariad; Astex; Bristol-Myers Squibb; Cyclacel; Daiichi-Sankyo; Pfizer; Immunogen; Jazz; Novartis and honoraria from Pfizer; Immunogen; Actinium and Takeda. YA received research funding from Jazz and honoraria from Abbott. GB received research funding from AbbVie; Incyte; Janssen; Cyclacel; BioLine Rx; NKarta: and consulting honoraria from BioLine Rx; NKarta; PTC Therapeutics; Oncoceutics, Inc. ND received research funding from Sunesis Pharmaceuticals, Inc.; Karyopharm; Immunogen; Pfizer; Incyte; Bristol-Myers Squibb; Daiichi-Sankyo; Kiromic and consulting honoraria from Sunesis Pharmaceuticals, Inc.; Karyopharm; Pfizer; Incyte; Bristol-Myers Squibb; Novartis; Otsuka America Pharmaceutical, Inc. CDD received research funding from Agios; Novartis; Celgene; Daiichi-Sankyo; Calithera Biosciences and consulting honoraria from AbbVie; Agios; Novartis; Celgene; Daiichi-Sankyo; Jazz; Notable Laboratories and is a Scientific Advisory Board member of Notable Laboratories. PB received research funding from Incyte; Celgene; CTI BioPharma; Blueprint Medicines; Constellation Pharmaceuticals; Kartos Therapeutics; Astellas; Pfizer; NS Pharma; Promedior and consulting honoraria from Incyte; Celgene; CTI BioPharma; Blueprint Medicines; Kartos Therapeutics. NJ received research funding and consulting honoraria from AbbVie; Pharmacyclics; Genentech; Bristol-Myers Squibb; Pfizer; ADC Therapeutics; AstraZeneca; Servier; Cellectis; Verastem; Precision Biosciences; Adaptive. NP received research funding from Stemline; Novartis; Abbvie; Samus; Cellectis; Plexxikon; Daiichi-Sankyo; Affymetrix; and received consulting honoraria from Celgene; Stemline; Incyte; Novartis; MustangBio; Roche Di, LFB. KT received research funding from Onconova; MEI, served on an advisory board for Symbio Pharmaceuticals; GSK; Celgene and received honoraria from Dava Oncology; Kyowa Hakko Kirin. MA received research funding from Daiichi Sankyo, Inc.; Jazz; and consultancy honoraria from Jazz; Celgene; Amgen; AstaZeneca; Dimensions Capital; and equity ownership from Reata; Aptose; Europics; Senti Bio; Oncoceutics; Oncolyze. FR received research funding from Amgen, Bristol-Myers Squibb, Merck, Seattle Genetics, Sunesis Pharmaceuticals; Pfizer; and honoraria for consulting or advisory role for Jazz; Amgen; Seattle Genetics; Sunesis Pharmaceuticals. JEC received research funding (to the institution while at the MD Anderson) and consulting honoraria from Celator/Jazz, Novartis, Daiichi, Astellas, Daiichi, Merus, Immunogen, Biopath Holdings.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Issa, G.C., Kantarjian, H.M., Xiao, L. et al. Phase II trial of CPX-351 in patients with acute myeloid leukemia at high risk for induction mortality. Leukemia 34, 2914–2924 (2020). https://doi.org/10.1038/s41375-020-0916-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-020-0916-8