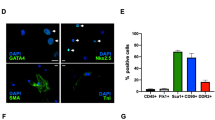
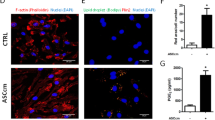
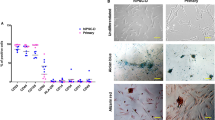
Abstract
Light chain (AL) amyloidosis is a progressive, degenerative disease characterized by the misfolding and amyloid deposition of immunoglobulin light chain (LC). The amyloid deposits lead to organ failure and death. Our laboratory is specifically interested in cardiac involvement of AL amyloidosis. We have previously shown that the fibrillar aggregates of LC proteins can be cytotoxic and arrest the growth of human RFP-AC16 cardiomyocytes in vitro. We showed that adipose-derived mesenchymal stromal cells (AMSC) can rescue the cardiomyocytes from the fibril-induced growth arrest through contact-dependent mechanisms. In this study, we examined the transcriptome changes of human cardiomyocytes and AMSC in the presence of AL amyloid fibrils. The presence of fibrils causes a ‘priming’ immune response in AMSC associated with interferon associated genes. Exposure to AL fibrils induced changes in the pathways associated with immune response and extracellular matrix components in cardiomyocytes. We also observed upregulation of innate immune-associated transcripts (chemokines, cytokines, and complement), suggesting that amyloid fibrils initiate an innate immune response on these cells, possibly due to phenotypic transformation. This study corroborates and expands our previous studies and identifies potential new immunologic mechanisms of action for fibril toxicity on human cardiomyocytes and AMSC rescue effect on cardiomyocytes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Baden EM, Sikkink LA, Ramirez-Alvarado M. Light chain amyloidosis-current findings and future prospects. Curr Protein Pept Sci. 2009;10:500–8.
Blancas-Mejía LM, Ramirez-Alvarado M. Systemic amyloidoses. Annu Rev Biochem. 2013;82:745–74.
Falk RH. Diagnosis and management of the cardiac amyloidoses. Circulation. 2005;112:2047–60.
Merlini G. CyBorD: stellar response rates in AL amyloidosis. Blood. 2012;119:4343–5.
Mikhael JR, Schuster SR, Jimenez-Zepeda VH, Bello N, Spong J, Reeder CB, et al. Cyclophosphamide-bortezomib-dexamethasone (CyBorD) produces rapid and complete hematologic response in patients with AL amyloidosis. Blood. 2012;119:4391–4.
Gertz MA. Immunoglobulin light chain amyloidosis: 2016 update on diagnosis, prognosis, and treatment. Am J Hematol. 2016;91:947–56.
Trinkaus-Randall V, Walsh MT, Steeves S, Monis G, Connors LH, Skinner M. Cellular response of cardiac fibroblasts to amyloidogenic light chains. Am J Pathol. 2005;166:197–208.
Brenner DA, Jain M, Pimentel DR, Wang B, Connors LH, Skinner M, et al. Human amyloidogenic light chains directly impair cardiomyocyte function through an increase in cellular oxidant stress. Circulation Res. 2004;94:1008–10.
Sikkink L, Ramirez-Alvarado M. Cytotoxicity of amyloidogenic immunoglobulin light chains in cell culture. Cell death Dis. 2010;1:e98.
Levinson RT, Olatoye OO, Randles EG, Howell KG, DiCostanzo AC, Ramirez-Alvarado M. Role of mutations in the cellular internalization of amyloidogenic light chains into cardiomyocytes. Sci Rep. 2013;3:1278.
McWilliams-Koeppen HP, Foster JS, Hackenbrack N, Ramirez-Alvarado M, Donohoe D, Williams A, et al. Light chain amyloid fibrils cause metabolic dysfunction in human cardiomyocytes. PLoS ONE. 2015;10:e0137716.
Lin Y, Marin-Argany M, Dick CJ, Redhage KR, Blancas-Mejia LM, Bulur P, et al. Mesenchymal stromal cells protect human cardiomyocytes from amyloid fibril damage. Cytotherapy. 2017;19:1426–37.
Marin-Argany M, Lin Y, Misra P, Williams A, Wall JS, Howell KG, et al. Cell damage in light chain amyloidosis: fibril internalization, toxicity and cell-mediated seeding. J Biol Chem. 2016;291:19813–25.
Buxbaum JN. Animal models of human amyloidoses: are transgenic mice worth the time and trouble? FEBS Lett 2009;583:2663–73.
Davidson MM, Nesti C, Palenzuela L, Walker WF, Hernandez E, Protas L, et al. Novel cell lines derived from adult human ventricular cardiomyocytes. J Mol Cell Cardiol. 2005;39:133–47.
Poshusta TL, Katoh N, Gertz MA, Dispenzieri A, Ramirez-Alvarado M. Thermal stability threshold for amyloid formation in light chain amyloidosis. Int J Mol Sci. 2013;14:22604–17.
Wall J, Schell M, Murphy C, Hrncic R, Stevens FJ, Solomon A. Thermodynamic instability of human λ6 light chains: correlation with fibrillogenicity. Biochemistry 1999;38:14101–8.
Wall JS, Gupta V, Wilkerson M, Schell M, Loris R, Adams P, et al. Structural basis of light chain amyloidogenicity: comparison of the thermodynamic properties, fibrillogenic potential and tertiary structural features of four Vλ6 proteins. J Mol Recognit. 2004;17:323–31.
Bernardo ME, Locatelli F, Fibbe WE. Mesenchymal stromal cells. Ann New Y Acad Sci. 2009;1176:101–17.
Blancas-Mejia LM, Misra P, Dick CJ, Marin-Argany M, Redhage KR, Cooper SA, et al. Assays for light chain amyloidosis formation and cytotoxicity. Protein Misfolding Dis Springer. 2019;1873:p. 123–53.
Dudakovic A, Camilleri E, Riester SM, Lewallen EA, Kvasha S, Chen X, et al. High‐resolution molecular validation of self‐renewal and spontaneous differentiation in clinical‐grade adipose‐tissue derived human mesenchymal stem cells. J Cell Biochem. 2014;115:1816–28.
Szklarczyk D, Morris JH, Cook H, Kuhn M, Wyder S, Simonovic M, et al. The STRING database in 2017: quality-controlled protein–protein association networks, made broadly accessible. Nucleic Acids Res. 2017;45:D362–8.
Metsalu T, Vilo J. ClustVis: a web tool for visualizing clustering of multivariate data using principal component analysis and heatmap. Nucleic acids Res. 2015;43(W1):W566–W70.
Lin Y, Hogan WJ. Clinical application of mesenchymal stem cells in the treatment and prevention of graft-versus-host disease. Adv Hematol. 2011;2011:17.
Hafer‐Macko CE, Dyck PJ, Koski CL. Complement activation in acquired and hereditary amyloid neuropathy. J Peripheral Nerv Syst. 2000;5:131–9.
Rodolico C, Mazzeo A, Toscano A, Pastura C, Maimone D, Musumeci O, et al. Amyloid myopathy presenting with rhabdomyolysis: evidence of complement activation. Neuromuscul Disord. 2006;16:514–7.
Bodin K, Ellmerich S, Kahan MC, Tennent GA, Loesch A, Gilbertson JA, et al. Antibodies to human serum amyloid P component eliminate visceral amyloid deposits. Nature. 2010;468:93.
Matsuoka Y, Picciano M, Malester B, LaFrancois J, Zehr C, Daeschner JM, et al. Inflammatory responses to amyloidosis in a transgenic mouse model of Alzheimer’s disease. Am J Pathol. 2001;158:1345–54.
Shi Q, Chowdhury S, Ma R, Le KX, Hong S, Caldarone BJ, et al. Complement C3 deficiency protects against neurodegeneration in aged plaque-rich APP/PS1 mice. Sci Transl Med. 2017;9:eaaf6295.
Stoltzner SE, Grenfell TJ, Mori C, Wisniewski KE, Wisniewski TM, Selkoe DJ, et al. Temporal accrual of complement proteins in amyloid plaques in Down’s syndrome with Alzheimer’s disease. Am J Pathol. 2000;156:489–99.
Eikelenboom P, Hack C, Kamphorst W, Rozemuller J. Distribution pattern and functional state of complement proteins and alpha 1-antichymotrypsin in cerebral beta/A4 deposits in Alzheimer’s disease. Res Immunol. 1992;143:617–20.
Halle A, Hornung V, Petzold GC, Stewart CR, Monks BG, Reinheckel T, et al. The NALP3 inflammasome is involved in the innate immune response to amyloid-β. Nat Immunol. 2008;9:857.
Masters SL, O’Neill LA. Disease-associated amyloid and misfolded protein aggregates activate the inflammasome. Trends Mol Med. 2011;17:276–82.
Diomede L, Romeo M, Rognoni P, Beeg M, Foray C, Ghibaudi E, et al. Cardiac light chain amyloidosis: the role of metal ions in oxidative stress and mitochondrial damage. Antioxid redox Signal. 2017;27:567–82.
Russo RC, Garcia CC, Teixeira MM, Amaral FA. The CXCL8/IL-8 chemokine family and its receptors in inflammatory diseases. Expert Rev Clin Immunol. 2014;10:593–619.
Kruger P, Saffarzadeh M, Weber AN, Rieber N, Radsak M, von Bernuth H, et al. Neutrophils: between host defence, immune modulation, and tissue injury. PLoS Pathog. 2015;11:e1004651.
Kolaczkowska E, Kubes P. Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol. 2013;13:159.
Keeling J, Teng J, Herrera GA. AL-amyloidosis and light-chain deposition disease light chains induce divergent phenotypic transformations of human mesangial cells. Lab Investig. 2004;84:1322.
Teng J, Russell WJ, Gu X, Cardelli J, Jones ML, Herrera GA. Different types of glomerulopathic light chains interact with mesangial cells using a common receptor but exhibit different intracellular trafficking patterns. Lab Investig. 2004;84:440.
Horwitz EM, Andreef M, Frassoni F. Mesenchymal stromal cells. Curr Opin Hematol. 2006;13:419–25.
Duijvestein M, Wildenberg ME, Welling MM, Hennink S, Molendijk I, van Zuylen VL, et al. Pretreatment with interferon‐γ enhances the therapeutic activity of mesenchymal stromal cells in animal models of colitis. Stem Cells. 2011;29:1549–58.
Pessina A, Bonomi A, Coccè V, Invernici G, Navone S, Cavicchini L, et al. Mesenchymal stromal cells primed with paclitaxel provide a new approach for cancer therapy. PLoS ONE. 2011;6:e28321.
Bridge AJ, Pebernard S, Ducraux A, Nicoulaz A-L, Iggo R. Induction of an interferon response by RNAi vectors in mammalian cells. Nat Genet. 2003;34:263.
Salomon R, Staeheli P, Kochs G, Yen H-L, Franks J, Rehg JE, et al. Mx1 gene protects mice against the highly lethal human H5N1 influenza virus. Cell Cycle. 2007;6:2417–21.
Soscia SJ, Kirby JE, Washicosky KJ, Tucker SM, Ingelsson M, Hyman B, et al. The Alzheimer’s disease-associated amyloid β-protein is an antimicrobial peptide. PloS one. 2010;5:e9505.
Fulop T, Witkowski JM, Bourgade K, Khalil A, Zerif E, Larbi A, et al. Can an infection hypothesis explain the beta amyloid hypothesis of Alzheimer’s disease? Frontiers in aging. Neuroscience. 2018;10:224.
Acknowledgements
We thank the staff of the Medical Genome Facility Expression Core for carrying out the RNAseq analysis and the staff of the Flow Cytometry Core for their assistance. We also thank Michael Bergman, Shawna Cooper, Christopher Parks, and Christopher Paradise for their contributions to this project. TLJ is a graduate student at Mayo Clinic Graduate School of Biomedical Sciences. This work is submitted in partial fulfillment of the requirement for the PhD program. This study was supported in part by NIH R01 GM 128253, the Mayo Foundation, and the generous support of amyloidosis patients and their families.
Author information
Authors and Affiliations
Contributions
TLJ, MRA and YL designed experiments, analyzed results, and wrote the manuscript. TLJ, KM, PM, CJD, LBM, KRR, AW, conducted experiments AvW analyzed data and wrote the manuscript JSW analyzed data and wrote the manuscript
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Jordan, T.L., Maar, K., Redhage, K.R. et al. Light chain amyloidosis induced inflammatory changes in cardiomyocytes and adipose-derived mesenchymal stromal cells. Leukemia 34, 1383–1393 (2020). https://doi.org/10.1038/s41375-019-0640-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-019-0640-4
This article is cited by
-
Inflammation across the spectrum of hypertrophic cardiac phenotypes
Heart Failure Reviews (2023)