Abstract
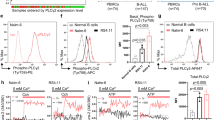
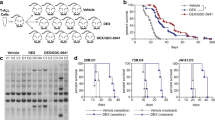
Glucocorticoid (GC) receptor (GR) phosphorylation and signature genes were studied in chronic lymphocytic leukemia (CLL) cells to help place GCs within modern treatment algorithms. In contrast to normal B and T cells, transcription of GC-regulated genes was not rhythmic and the synthetic GC dexamethasone (DEX) could not inhibit toll-like receptor (TLR)-responses in CLL cells. This intrinsic GC-resistance was associated with aberrant GR-phosphorylation on activating Ser211 and inhibitory Ser226 sites. Ibrutinib increased transcription of the GR-signature gene GILZ in circulating CLL cells along with GR(pS211)/GR(pS226) ratios and lytic sensitivity to DEX that were not reversed by the competitive antagonist mifepristone in vitro. However, ibrutinib could not improve GR-responses in circulating CLL cells activated with IL2 and TLR7/8 agonists to mimic conditions in pseudofollicle microenvironments. Addition of the janus kinase inhibitor ruxolitinib to block ibrutinib-insensitive signals increased GILZ transcription in pseudofollicle conditions in vitro and in a clinical trial (NCT02912754), and also increased GR(S211)/GR(S226) ratios and DEX-mediated killing in patient samples in vitro. These observations suggest that intrinsic resistance to endogenous GCs is characteristic of CLL cells and ibrutinib may help increase the therapeutic activity of GCs by non-canonical activation of GR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author (DS) upon request.
References
Spaner DE. Oral high-dose glucocorticoids and Ofatumumab in fludarabine-resistant Chronic Lymphocytic Leukemia. Leukemia. 2012;26:1144–5.
Coutre SE, Furman RR, Flinn IW, Burger JA, Blum K, Sharman J, et al. Extended Treatment with Single-Agent Ibrutinib at the 420 mg Dose Leads to Durable Responses in Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma. Clin Cancer Res. 2017;23:1149–55.
Kino T. GR-regulating Serine/Threonine Kinases: New Physiologic and Pathologic Implications. Trends Endocrinol Metab. 2018;29:260–70.
Robertson S, Rohwer JM, Hapgood JP, Louw A. Impact of glucocorticoid receptor density on ligand-independent dimerization, cooperative ligand-binding and basal priming of transactivation: a cell culture model. PLoS One. 2013;8:e64831.
Hapgood JP, Avenant C, Moliki JM. Glucocorticoid-independent modulation of GR activity: Implications for immunotherapy. Pharmacol Ther. 2016;16:93–113.
Galliher-Beckley AJ, Cidlowski JA. Emerging roles of glucocorticoid receptor phosphorylation in modulating glucocorticoid hormone action in health and disease. IUBMB Life. 2009;61:979–86.
Simic I, Adzic M, Maric N, Savic D, Djordjevic J, Mihaljevic M, et al. A preliminary evaluation of leukocyte phospho-glucocorticoid receptor as a potential biomarker of depressogenic vulnerability in healthy adults. Psychiatry Res. 2013;209:658–64.
Kwon HS, Huang B, Unterman TG, Harris RA. Protein kinase B-alpha inhibits human pyruvate dehydrogenase kinase-4 gene induction by dexamethasone through inactivation of FOXO transcription factors. Diabetes. 2004;53:899–910.
Tung S, Shi Y, Wong K, Zhu F, Gorczynski R, Laister RC, et al. PPARalpha and fatty acid oxidation mediate glucocorticoid resistance in chronic lymphocytic leukemia. Blood. 2013;122:969–80.
Bruscoli S, Biagioli M, Sorcini D, Frammartino T, Cimino M, Sportoletti P, et al. Lack of glucocorticoid-induced leucine zipper (GILZ) deregulates B-cell survival and results in B-cell lymphocytosis in mice. Blood. 2015;126:1790–801.
Shipp LE, Lee JV, Yu CY, Pufall M, Zhang P, Scott DK, et al. Transcriptional regulation of human dual specificity protein phosphatase 1 (DUSP1) gene by glucocorticoids. PLoS One. 2010;5:e13754.
Newton R, Holden NS. Separating transrepression and transactivation: a distressing divorce for the glucocorticoid receptor? Mol Pharmacol. 2007;72:799–809.
Miller GE, Cohen S, Ritchey AK. Chronic psychological stress and the regulation of pro-inflammatory cytokines: a glucocorticoid-resistance model. Health Psychol. 2002;21:531–41.
Oppermann S, Ylanko J, Shi Y, Hariharan S, Oakes CC, Brauer PM, et al. High-content screening identifies kinase inhibitors that overcome venetoclax resistance in activated CLL cells. Blood. 2016;128:934–47.
Chen SS, Chang BY, Chang S, Tong T, Ham S, Sherry B, et al. BTK inhibition results in impaired CXCR4 chemokine receptor surface expression, signaling and function in chronic lymphocytic leukemia. Leukemia. 2016;30:833–43.
Herishanu Y, Perez-Galan P, Liu D, Biancotto A, Pittaluga S, Vire B, et al. The lymph node microenvironment promotes B-cell receptor signaling, NF-kappaB activation, and tumor proliferation in chronic lymphocytic leukemia. Blood. 2011;117:563–74.
Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Dighiero G, Dohner H, et al. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood. 2008;111:5446–56.
Bjarnason G, Spaner D, Ackloo S, Li J, Zhu P, Nuin P, et al. Diurnal proteomics and biomarker discovery in indolent versus aggressive chronic lymphocytic leukemia patients. J Clin Oncol. 2012;30S:10622–10622.
Tomic J, Lichty B, Spaner DE. Aberrant interferon-signaling is associated with aggressive chronic lymphocytic leukemia. Blood. 2011;117:2668–80.
Hammond CM, White D, Tomic J, Shi Y, Spaner DE. Extracellular calcium sensing promotes human B-cell activation and function. Blood. 2007;110:3985–95.
Oppermann S, Lam AJ, Tung S, Shi Y, McCaw L, Wang G, et al. Janus and PI3-kinases mediate glucocorticoid resistance in activated chronic leukemia cells. Oncotarget. 2016;7:72608–21.
Besedovsky L, Born J, Lange T. Endogenous glucocorticoid receptor signaling drives rhythmic changes in human T-cell subset numbers and the expression of the chemokine receptor CXCR4. FASEB J. 2014;28:67–75.
Almon RR, Yang E, Lai W, Androulakis IP, Ghimbovschi S, Hoffman EP, et al. Relationships between circadian rhythms and modulation of gene expression by glucocorticoids in skeletal muscle. Am J Physiol Regul Integr Comp Physiol. 2008;295:R1031–R1047.
Spaner DE, Lee E, Shi Y, Wen F, Li Y, Tung S, et al. PPAR-alpha is a therapeutic target for chronic lymphocytic leukemia. Leukemia. 2013;27:1090–9.
Stark JL, Avitsur R, Padgett DA, Campbell KA, Beck FM, Sheridan JF. Social stress induces glucocorticoid resistance in macrophages. Am J Physiol Regul Integr Comp Physiol. 2001;280:R1799–R1805.
Haferlach T, Kohlmann A, Wieczorek L, Basso G, Kronnie GT, Bene MC, et al. Clinical utility of microarray-based gene expression profiling in the diagnosis and subclassification of leukemia: report from the International Microarray Innovations in Leukemia Study Group. J Clin Oncol. 2010;28:2529–37.
Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D, et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia. 2004;6:1–6.
Hammond CM, Shi Y, White D, Cervi D, Tomic J, Spaner DE. The B-cell calcium sensor predicts progression of chronic lymphocytic leukemia. Leukemia. 2009;23:426–9.
Verhaar AP, Wildenberg ME, Duijvestein M, Vos AC, Peppelenbosch MP, Lowenberg M, et al. Superantigen-induced steroid resistance depends on activation of phospholipase Cbeta2. J Immunol. 2013;190:6589–95.
Bleasdale JE, Bundy GL, Bunting S, Fitzpatrick FA, Huff RM, Sun FF, et al. Inhibition of phospholipase C dependent processes by U73122. Adv Prostaglandin Thromboxane Leukot Res. 1989;19:590–3.
Woyach JA, Smucker K, Smith LL, Lozanski A, Zhong Y, Ruppert AS, et al. Prolonged lymphocytosis during ibrutinib therapy is associated with distinct molecular characteristics and does not indicate a suboptimal response to therapy. Blood. 2014;123:1810–7.
Advani RH, Buggy JJ, Sharman JP, Smith SM, Boyd TE, Grant B, et al. Bruton Tyrosine Kinase Inhibitor Ibrutinib (PCI-32765) Has Significant Activity in Patients With Relapsed/Refractory B-Cell Malignancies. J Clin Oncol. 2013;31:88–94.
Chapman K, Holmes M, Seckl J. 11beta-hydroxysteroid dehydrogenases: intracellular gate-keepers of tissue glucocorticoid action. Physiol Rev. 2013;93:1139–206.
Nordeen SK, Bona BJ, Moyer ML. Latent agonist activity of the steroid antagonist, RU486, is unmasked in cells treated with activators of protein kinase A. Mol Endocrinol. 1993;7:731–42.
Ng HP, Jennings S, Wang J, Molina PE, Nelson S, Wang G. Non-canonical Glucocorticoid Receptor Transactivation of gilz by Alcohol Suppresses Cell Inflammatory Response. Front Immunol. 2017;8:661.
Patel V, Balakrishnan K, Bibikova E, Ayres M, Keating MJ, Wierda W, et al. Comparison of acalabrutinib, a selective BTK inhibitor, with ibrutinib in chronic lymphocytic leukemia cells. Clin Cancer Res. 2016;23:3734–343.
Page T, Urbaniak A, Espirito Santo A, Danks L, Smallie T, Williams L, et al. BTK regulates TLR7/8-induced TNF transcription via nuclear factor-κB recruitment. Biochem Biophys Res Commun. 2018;499:260–6.
Woyach JA, Ruppert AS, Guinn D, Lehman A, Blachly JS, Lozanski A, et al. BTKC481S-mediated resistance to ibrutinib in chronic lymphocytic leukemia. J Clin Oncol. 2017;35:1437–93.
Thompson PA, O’Brien SM, Xiao L, Wang X, Burger JA, Jain N, et al. beta 2-microglobulin normalization within 6 months of ibrutinib-based treatment is associated with superior progression-free survival in patients with chronic lymphocytic leukemia. Cancer. 2016;122:565–73.
Tarafdar A, Michie AM. Protein kinase C in cellular transformation: a valid target for therapy? Biochem Soc Trans. 2014;42:1556–62.
Calissano C, Damle RN, Marsilio S, Yan X-J, Yancopoulos S, Hayes G, et al. Intraclonal complexity in CLL: fractions enriched in recently born/divided and older/quiescent cells. Mol Med. 2011;17:1374–82.
Spaner DE, Wang G, McCaw L, Li Y, Disperati P, Cussen M, et al. Activity of the janus kinase inhibitor Ruxolitinib in chronic lymphocytic leukemia: results of a phase II trial. Haematologica. 2016;101:e192–e195.
Burger JA, Montserrat E. Coming full circle: 70 years of chronic lymphocytic leukemia cell redistribution, from glucocorticoids to inhibitors of B-cell receptor signaling. Blood. 2013;121:1501–9.
Kfir-Erenfeld S, Sionov RV, Spokoini R, Cohen O, Yefenof E. Protein kinase networks regulating glucocorticoid-induced apoptosis of hematopoietic cancer cells: fundamental aspects and practical considerations. Leuk Lymphoma. 2010;51:1968–2005.
Schmidt S, Rainer J, Ploner C, Presul E, Riml S, Kofler R. Glucocorticoid-induced apoptosis and glucocorticoid resistance: molecular mechanisms and clinical relevance. Cell Death Differ. 2004;11S1:S45–S55.
Sato Y, Suzuki H, Sato T, Suda T, Yoda T, Iwakura Y, et al. The role of endogenous glucocorticoids in lymphocyte development in melanocortin receptor 2-deficient mice. Biochem Biophys Res Commun. 2010;403:253–7.
Li Y, Shi Y, McCaw L, Li YJ, Zhu F, Gorczynski R, et al. Microenvironmental interleukin-6 suppresses toll-like receptor signaling in human leukemia cells through miR-17/19A. Blood. 2015;126:766–78.
Matthews LC, Berry AA, Morgan DJ, Poolman TM, Bauer K, Kramer F, et al. Glucocorticoid receptor regulates accurate chromosome segregation and is associated with malignancy. Proc Natl Acad Sci USA. 2015;112:5479–84.
Yu J, Chen L, Cui B, Wu C, Choi MY, Chen Y, et al. Cirmtuzumab inhibits Wnt5a-induced Rac1 activation in chronic lymphocytic leukemia treated with ibrutinib. Leukemia. 2017;31:1333–9.
Real PJ, Tosello V, Palomero T, Castillo M, Hernando E, de Stanchina E, et al. Gamma-secretase inhibitors reverse glucocorticoid resistance in T cell acute lymphoblastic leukemia. Nat Med. 2009;15:50–58.
Acknowledgements
This work was supported by CIHR grants FRN130479 and FRN153291, the Leukemia and Lymphoma Society of Canada (DS), and the National Institutes of Health, National Cancer Institute R01 CA197870 (JAW).
Author contributions
YS and DES conceived the project, designed the experiments, and wrote the manuscript. YS and GW performed the experiments. LM helped write manuscript. CW and GB performed gene array analysis for circadian expression and helped write the manuscript. EM and JAW helped designed and performed experiments and helped write revised manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Shi, Y., Wang, G., Muhowski, E.M. et al. Ibrutinib reprograms the glucocorticoid receptor in chronic lymphocytic leukemia cells. Leukemia 33, 1650–1662 (2019). https://doi.org/10.1038/s41375-019-0381-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-019-0381-4