Abstract
Objective
To assess the association between antenatal corticosteroids exposure and postnatal growth in infants born at 23–29 weeks’ gestation.
Study design
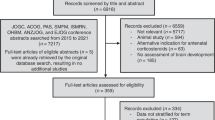
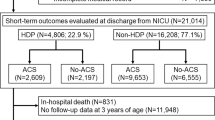
This retrospective study used data from the Pediatrix Clinical Data Warehouse. Maternal-infant dyads from 2018 to 2020 were included. Inverse propensity weighting (IPW) was applied to balance pre-treatment confounders. Primary outcomes included postnatal weight, length, and head circumference growth trajectory percentiles.
Result
The unadjusted cohort consisted of 11,912 dyads. After IPW adjustment, there were 23,231 dyads. Exposed infants showed higher postnatal trajectory percentiles for weight (by 3.4%), length (by 1.8%), and head circumference (by 2.5%) when compared to non-exposed infants. The positive association between antenatal corticosteroids and postnatal growth was only observed in infants not exposed to preeclampsia/eclampsia/HELLP syndrome or without fetal growth restriction.
Conclusion
Antenatal corticosteroids exposure is associated with better postnatal growth. The study is limited by its retrospective nature.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
Data can be requested by contacting Dr. Reese Clark or by completing the online request form at https://mednax2ndhipaaaccount-gajdh.formstack.com/forms/research_application_complete.
References
Roberts D, Brown J, Medley N, Dalziel SR. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database Syst Rev. 2017;3:CD004454.
Saito K, Nishimura E, Ota E, Namba F, Swa T, Ramson J, et al. Antenatal corticosteroids in specific groups at risk of preterm birth: a systematic review. BMJ Open. 2023;13:e065070.
Committee on obstetric practice. https://www.acog.org/-/media/project/acog/acogorg/clinical/files/committee-opinion/articles/2017/08/antenatal-corticosteroid-therapy-for-fetal-maturation.pdf (accessed 30 May 2023). (2017).
Use of antenatal corticosteroids at 22 weeks of gestation. https://www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2021/09/use-of-antenatal-corticosteroids-at-22-weeks-of-gestation (accessed 30 May 2023).
Ikegami M, Jobe AH, Newnham J, Polk DH, Willet KE, Sly P. Repetitive prenatal glucocorticoids improve lung function and decrease growth in preterm lambs. Am J Respir Crit Care Med. 1997;156:178–84.
Jobe AH, Wada N, Berry LM, Ikegami M, Ervin MG. Single and repetitive maternal glucocorticoid exposures reduce fetal growth in sheep. Am J Obstet Gynecol. 1998;178:880–5.
Thorp JA, Jones PG, Knox E, Clark RH. Does antenatal corticosteroid therapy affect birth weight and head circumference? Obstet Gynecol. 2002;99:101–8.
Murphy KE, Willan AR, Hannah ME, Ohlsson A, Kelly EN, Matthews SG, et al. Effect of antenatal corticosteroids on fetal growth and gestational age at birth. Obstet Gynecol. 2012;119:917–23.
Rizzo G, Mappa I, Bitsadze V, Khizroeva J, Makatsariya A, D’Antonio F. Administration of antenatal corticosteroid is associated with reduced fetal growth velocity: a longitudinal study. J Matern Fetal Neonatal Med. 2022;35:2775–80.
Laptook AR, Weydig H, Brion LP, Wyckoff MH, Arnautovic TI, Younge N, et al. Antenatal steroids, prophylactic indomethacin, and the risk of spontaneous intestinal perforation. J Pediatr. 2023;259:113457.
Dickerman BA, García-Albéniz X, Logan RW, Denaxas S, Hernán MA. Avoidable flaws in observational analyses: an application to statins and cancer. Nat Med. 2019;25:1601–6.
Hernán MA, Wang W, Leaf DE. Target trial emulation: a framework for causal inference from observational data. JAMA. 2022;328:2446–7.
Hernández-Díaz S, Huybrechts KF, Chiu Y-H, Yland JJ, Bateman BT, Hernán MA. Emulating a target trial of interventions initiated during pregnancy with healthcare databases: the example of COVID-19 vaccination. Epidemiology. 2023;34:238–46.
Spitzer AR, Ellsbury DL, Handler D, Clark RH. The Pediatrix BabySteps Data Warehouse and the Pediatrix QualitySteps improvement project system-tools for “meaningful use” in continuous quality improvement. Clin Perinatol. 2010;37:49–70.
Chou F-S, Yeh H-W, Clark RH. A comparative study of postnatal anthropometric growth in very preterm infants and intrauterine growth. Nat Commun. 2023;14:5626.
twang: Toolkit for Weighting and Analysis of Nonequivalent Groups. Comprehensive R Archive Network (CRAN). https://cran.r-project.org/web/packages/twang/index.html (accessed 31 May 2023).
Griffin BA, et al. Toolkit for Weighting and Analysis of Nonequivalent Groups (TWANG) Website. Santa Monica, CA: RAND Corporation, 2014. http://www.rand.org/statistics/twang.
Survey: Analysis of Complex Survey Samples. Comprehensive R Archive Network (CRAN). https://cran.r-project.org/web/packages/survey/index.html (accessed 7 February 2023).
Chou, F-S, Yeh, H-W, Clark RH Postnatal Growth Charts for Preterm Infants. https://nicugrowth.app (accessed 25 June 2023).
Thorp JA, Jones PG, Peabody JL, Knox E, Clark RH. Effect of antenatal and postnatal corticosteroid therapy on weight gain and head circumference growth in the nursery. Obstet Gynecol. 2002;99:109–15.
Chou F-S, Yeh H-W, Chen C-Y, Lee GT, Parrish MR, Omede M, et al. Exposure to placental insufficiency alters postnatal growth trajectory in extremely low birth weight infants. J Dev Orig Health Dis. 2020;11:384–91.
Chou F-S, Pandey V, Yeh H-W. Postnatal growth in extremely low birth weight newborns: nature or nurture? J Perinatol 2021;41:648–9.
Economides DL, Nicolaides KH, Linton EA, Perry LA, Chard T. Plasma cortisol and adrenocorticotropin in appropriate and small for gestational age fetuses. Fetal Ther. 1988;3:158–64.
Goland RS, Jozak S, Warren WB, Conwell IM, Stark RI, Tropper PJ. Elevated levels of umbilical cord plasma corticotropin-releasing hormone in growth-retarded fetuses. J Clin Endocrinol Metab. 1993;77:1174–9.
Ng PC. The fetal and neonatal hypothalamic-pituitary-adrenal axis. Arch Dis Child Fetal Neonatal Ed. 2000;82:F250–4.
Author information
Authors and Affiliations
Contributions
FC conceptualized the study, performed data analysis, and drafted the initial and revised manuscript. RHC performed initial data extraction and clean-up, and critically reviewed the manuscript. HY supervised data analysis and critically reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was determined by the Institutional Review Board of Kaiser Permanente Southern California to be non-human subject research due to the dataset’s de-identified nature, and the informed consent requirement was accordingly exempted.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chou, FS., Clark, R.H. & Yeh, HW. The association between antenatal corticosteroids exposure and postnatal growth in infants born between 23 and 29 weeks of gestation. J Perinatol 44, 561–567 (2024). https://doi.org/10.1038/s41372-024-01871-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-024-01871-0