Abstract
Objectives
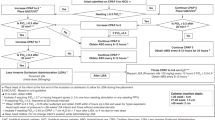
Minimally Invasive Surfactant Treatment (MIST) is a common method for administering surfactant as a treatment for respiratory distress syndrome. However, tracheal catheter placement can be difficult to confirm. We assessed the presence of carbon dioxide (CO2) in tracheal and esophageal gas aspirated using CO2 detector.
Study design
Retrospective arm: 20 infants, MIST catheter placement was assessed with a CO2 detector in two techniques and confirmed with clinical response. Prospective arm—10 infants, aimed to check for CO2 presence in aspirated esophageal gas during routine nasogastric tube insertion.
Results
Retrospective arm: All infants had positive capnography. One infant that had no clinical response to MIST was diagnosed with total anomalous pulmonary venous return. All 10 infants of the prospective arm had a Negative capnography (P < 0.001, Fisher’s exact test).
Conclusions
Readily available CO2 detectors can distinguish between tracheal and esophageal placement of MIST catheters prior to MIST.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
We will provide study data upon a reasonable request.
References
Sweet DG, Carnielli V, Greisen G, Hallman M, Ozek E, Plavka R, et al. European consensus guidelines on the management of respiratory distress syndrome—2016 update. Neonatol [Internet]. 2017;111:107–25.
Abdel-Latif ME, Davis PG, Wheeler KI, De Paoli AG, Dargaville PA. Surfactant therapy via thin catheter in preterm infants with or at risk of respiratory distress syndrome. Cochrane Database Syst Rev. 2021;5:CD011672. https://doi.org/10.1002/14651858.CD011672.pub2.
Klebermass-Schrehof K, Wald M, Schwindt J, Grill A, Prusa A-R, Haiden N, et al. Less invasive surfactant administration in extremely preterm infants: impact on mortality and morbidity. Neonatology. 2013;103:252–8.
Dargaville PA, Aiyappan A, Cornelius A, Williams C, De Paoli AG. Preliminary evaluation of a new technique of minimally invasive surfactant therapy. Arch Dis Child—Fetal Neonatal Ed. 2010;96:F243–8.
Göpel W, Kribs A, Ziegler A, Laux R, Hoehn T, Wieg C, et al. Avoidance of mechanical ventilation by surfactant treatment of spontaneously breathing preterm infants (AMV): an open-label, randomised, controlled trial. Lancet. 2011;378:1627–34.
Kribs A, Härtel C, Kattner E, Vochem M, Küster H, Möller J, et al. Surfactant without intubation in preterm infants with respiratory distress: first multi-center data. Klinische Pädiatrie. 2010;222:13–7.
Aldana-Aguirre JC, Pinto M, Featherstone RM, Kumar M. Less invasive surfactant administration versus intubation for surfactant delivery in preterm infants with respiratory distress syndrome: a systematic review and meta-analysis. Arch Dis Child—Fetal Neonatal Ed. 2016;102:F17–23.
Dargaville PA, Kamlin COF, Orsini F, Wang X, De Paoli AG, Kutman HGK, et al. Effect of minimally invasive surfactant therapy vs sham treatment on death or bronchopulmonary dysplasia in preterm infants with respiratory distress syndrome: The OPTIMIST-A randomized clinical trial. JAMA. 2021;326:2478–87. https://doi.org/10.1001/jama.2021.21892
Roberts CT, Halibullah I, Bhatia R, Green EA, Kamlin COF, Davis PG, et al. Outcomes after introduction of minimally invasive surfactant therapy in two Australian tertiary neonatal units. J Pediatrics. 2021;229:141–6.
Starship.org.nz. gatsby-starship;2021. Available from: https://starship.org.nz/guidelines/practice-recommendation-for-lisa-mist/.
Author information
Authors and Affiliations
Contributions
IN conceptualized and designed the study, wrote the study protocol, and reviewed and critically revised the manuscript. SA-R drafted the initial manuscript, performed some of the clinical tests and performed data collection and analysis. FRM and RAO helped in designing the study, and critically reviewed the manuscript for important intellectual content. DB-S and NA-L performed some of the clinical tests collected data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nitzan, I., Abu Omar, R., Mimouni, F.B. et al. Capnography for catheter location confirmation in minimally invasive surfactant administration. J Perinatol 43, 300–304 (2023). https://doi.org/10.1038/s41372-023-01624-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-023-01624-5