Abstract
Objective
To evaluate the outcomes of extremely premature infants who received inhaled nitric oxide(iNO) for hypoxic respiratory failure(HRF).
Study design
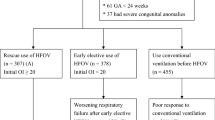
Retrospective analysis of 107 infants born 22–26 weeks gestation who received iNO for HRF at a single institution. Infants were categorized as positive, negative, or no responders based on change in FiO2 or OI. Underlying physiology was determined using Echocardiography/Radiography/Biochemistry.
Results
63% of infants had a positive response; they received iNO earlier and were more likely to have acute pulmonary hypertension(PH). Positive response correlated with decreased incidence of death or grade 3 BPD at 36 weeks postmenstrual age, as compared to a negative response.
Conclusions
Extremely premature infants have a positive response rate to iNO comparable to term infants when used for PH in the transitional period. Infants with a negative response to iNO had worse outcomes, necessitating the determination of the underlying physiology of HRF prior to iNO initiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
Requests to view the included data can be made to the corresponding author.
References
Dani C, Corsini I, Cangemi J, Vangi V, Pratesi S. Nitric oxide for the treatment of preterm infants with severe RDS and pulmonary hypertension. Pediatr Pulmonol. 2017;52:1461–8.
Pandya S, Baser O, Wan GJ, Lovelace G, Potenziano J, Pham At, et al. The burden of hypoxic respiratory failure in preterm and term/near-term infants in the United States 2011-2015. J Health Econ Outcomes Res. 2019;6:130–41.
Giesinger RE, More K, Odame J, Jain A, Jankov RP, McNamara PJ. Controversies in the identification and management of acute pulmonary hypertension in preterm neonates. Pediatr Res. 2017;82:901–14.
Seth SA, Soraisham AS, Harabor A. Risk factors and outcomes of early pulmonary hypertension in preterm infants. J Matern Fetal Neonatal Med, 2017;31:1–17.
Nakanishi H, Suenaga H, Uchiyama A, Kusuda S. Neonatal research network Japan, Persistent pulmonary hypertension of the newborn in extremely preterm infants: a Japanese cohort study. Arch Dis Child Fetal Neonatal Ed. 2018;103:F554–61.
Kumari S, Braun RK, Tetri LH, Barton GP, Hacker TA, Goss KN. Bimodal right ventricular dysfunction after postnatal hyperoxia exposure: implications for the preterm heart. Am J Physiol Heart Circ Physiol. 2019;317:H1272–81.
Carey WA, Weaver AL, Mara KC, Clark RH. Inhaled nitric oxide in extremely premature neonates with respiratory distress syndrome. Pediatrics, 2018;141:1–9.
Gaddam Bhoomaiah S, Rasiah SV. Outcomes of inhaled nitric oxide in preterm neonates—a five-year experience in a tertiary neonatal centre. Acta Paediatr. 2015;104:880–2.
Barrington KJ, Finer N, Pennaforte T. Inhaled nitric oxide for respiratory failure in preterm infants. Cochrane Database Syst Rev. 2017;1:Cd000509.
Baczynski M, Ginty S, Weisz DE, McNamara PJ, Kelly E, Shah P, et al. Short-term and long-term outcomes of preterm neonates with acute severe pulmonary hypertension following rescue treatment with inhaled nitric oxide. Arch Dis Child Fetal Neonatal Ed. 2017;102:F508–14.
Ahmed MS, Giesinger RE, Ibrahim M, Baczynski M, Louis D, McNamara KP, et al. Clinical and echocardiography predictors of response to inhaled nitric oxide in hypoxic preterm neonates. J Paediatr Child Health. 2019;55:753–61.
Dani C, Bertini G, Pezzati M, Fillipi L, Cecchi A, Rubaltelli FF. Inhaled nitric oxide in very preterm infants with severe respiratory distress syndrome. Acta Paediatr. 2006;95:1116–23.
Van Meurs KP, Wright LL, Ehrenkranz RA, Lemons JA, Ball MB, Poole WK, et al. Inhaled nitric oxide for premature infants with severe respiratory failure. N. Engl J Med. 2005;353:13–22.
Wernovsky G, Wypij D, Jonas RA, Mayer JE Jr, Hanley FL, Hickey PR, et al. Postoperative course and hemodynamic profile after the arterial switch operation in neonates and infants. A comparison of low-flow cardiopulmonary bypass and circulatory arrest. Circulation. 1995;92:2226–35.
Perri A, Riccardi R, Iannotta R, Di Molfetta DV, Arena R, Vento G, et al. Lung ultrasonography score versus chest X-ray score to predict surfactant administration in newborns with respiratory distress syndrome. Pediatr Pulmonol. 2018;53:1231–6.
Rios DR, de Freitas Martins F, El-Khuffash A, Weisz DE, Giesinger RE, McNamara PJ. Early role of the atrial-level communication in premature infants with patent ductus arteriosus. J Am Soc Echocardiogr. 2021;34:423–e1.
Teixeira LS, Shivananda SP, Stephens D, Van Arsdell GV, McNamara PJ. Postoperative cardiorespiratory instability following ligation of the preterm ductus arteriosus is related to early need for intervention. J Perinatol. 2008;28:803–10.
Jensen EA, Dysart K, Gantz MG, McDonald S, Bamat NA, Keszler M, et al. The diagnosis of bronchopulmonary dysplasia in very preterm infants. an evidence-based approach. Am J Respir Crit Care Med. 2019;200:751–9.
Backes CH, Rivera BK, Pavlek L, Beer LJ, Ball MK, Zettler ET, et al. Proactive neonatal treatment at 22 weeks of gestation: a systematic review and meta-analysis. Am J Obstet Gynecol. 2021;224:158–74.
Cole FS, Alleyne C, Barks JDE, Boyle RJ, Carroll JL, Dokken D, et al. NIH consensus development conference statement: inhaled nitric-oxide therapy for premature infants. Pediatrics. 2011;127:363–9.
Kumar P. Committee on fetus and newborn american academy of pediatrics, use of inhaled nitric oxide in preterm infants. Pediatrics. 2014;133:164–70.
Abman SH, Hansmann G, Archer SL, Ivy DD, Adatia I, Chung WK, et al. Pediatric pulmonary hypertension: guidelines from the American Heart Association and American Thoracic Society. Circulation. 2015;132:2037–99.
Ellsworth MA, Harris MN, Carey WA, Spitzer AR, Clark RH. Off-label use of inhaled nitric oxide after release of NIH consensus statement. Pediatrics. 2015;135:643–8.
Manja V, Guyatt G, Lakshminrusimha S, Jack S, Kirpalani H, Zupancic JAF, et al. Factors influencing decision making in neonatology: inhaled nitric oxide in preterm infants. J Perinatol. 2019;39:86–94.
Kumar VH, Hutchison AA, Lakshminrusimha S, Morin FC, Wynn RJ, Ryan RM. Characteristics of pulmonary hypertension in preterm neonates. J Perinatol. 2007;27:214–9.
Early compared with delayed inhaled nitric oxide in moderately hypoxaemic neonates with respiratory failure: a randomised controlled trial. The Franco-Belgium Collaborative NO Trial Group. Lancet, 1999;354:1066–71.
Uga N, Ishii T, Kawase Y, Arai H, Tada H. Nitric oxide inhalation therapy in very low-birthweight infants with hypoplastic lung due to oligohydramnios. Pediatr Int. 2004;46:10–4.
Chock VY, Van Meurs KP, Hintz SR, Ehrenkranz RA, Lemons JA, Kendrick DE, et al. Inhaled nitric oxide for preterm premature rupture of membranes, oligohydramnios, and pulmonary hypoplasia. Am J Perinatol. 2009;26:317–22.
Su PH, Chen JY. Inhaled nitric oxide in the management of preterm infants with severe respiratory failure. J Perinatol. 2008;28:112–6.
Field D, Elbourne D, Truesdale A, Grieve R, Hardy P, Fenton AC, et al. Neonatal ventilation with inhaled nitric oxide versus ventilatory support without inhaled nitric oxide for preterm infants with severe respiratory failure: the INNOVO multicentre randomised controlled trial (ISRCTN 17821339). Pediatrics. 2005;115:926–36.
Shaul PW. Ontogeny of nitric oxide in the pulmonary vasculature. Semin Perinatol. 1997;21:381–92.
Liu YA, Theis JG, Coceani F. Contractile and relaxing mechanisms in pulmonary resistance arteries of the preterm fetal lamb. Biol Neonate. 2000;77:253–60.
Skimming JW, DeMarco VG, Cassin S. The effects of nitric oxide inhalation on the pulmonary circulation of preterm lambs. Pediatr Res. 1995;37:35–40.
Rallis D, DeierlA, Atreja G, Chaban B, Banerjee J. The efficacy of inhaled nitric oxide treatment in premature infants with acute pulmonary hypertension. Early Hum Dev. 2018;127:1–5.
Barrington KJ, Finer N, Pennaforte T, Altit G. Nitric oxide for respiratory failure in infants born at or near term. Cochrane Database Syst Rev. 2017;1:Cd000399.
Bussmann N, El-Khuffash A, Breatnach CR, McCallion N, Franklin O, Singh GK, et al. Left ventricular diastolic function influences right ventricular—Pulmonary vascular coupling in premature infants. Early Hum Dev. 2019;128:35–40.
Hasan SU, Potenziano J, Konduri GG, Perez JA, Van Meurs KP, Walker MW, et al. Effect of inhaled nitric oxide on survival without bronchopulmonary dysplasia in preterm infants: a randomized clinical trial. JAMA Pediatr. 2017;171:1081–9.
Mercier JC, Hummler H, Durrmeyer X, Sanchez-Luna M, Carnielli V, Field D, et al. Inhaled nitric oxide for prevention of bronchopulmonary dysplasia in premature babies (EUNO): a randomised controlled trial. Lancet. 2010;376:346–54.
Jiang Q, Gao X, Liu C, Chen D, Lin X, Xia S, et al. Early inhaled nitric oxide in preterm infants <34 weeks with evolving bronchopulmonary dysplasia. J Perinatol. 2016;36:883–9.
Ballard RA, Truog WE, Cnaan A, Martin RJ, Ballard PL, Merrill JD, et al. Inhaled nitric oxide in preterm infants undergoing mechanical ventilation. N. Engl J Med. 2006;355:343–53.
Kinsella JP, Cutter GR, Walsh WF, Gerstmann DR, Bose CL, Hart C, et al. Early inhaled nitric oxide therapy in premature newborns with respiratory failure. N. Engl J Med. 2006;355:354–64.
ter Horst SA, Walther FJ, Poorthuis BJHM, Hiemstra PS, Wagenaar GTM. Inhaled nitric oxide attenuates pulmonary inflammation and fibrin deposition and prolongs survival in neonatal hyperoxic lung injury. Am J Physiol Lung Cell Mol Physiol. 2007;293:L35–44.
Tourneux P, Markham N, Seedorf G, Balasubramaniam V, Abman SH. Inhaled nitric oxide improves lung structure and pulmonary hypertension in a model of bleomycin-induced bronchopulmonary dysplasia in neonatal rats. Am J Physiol Lung Cell Mol Physiol. 2009;297:L1103–11.
Laube M, Amann E, Uhlig U, Yang Y, Fuchs HW, Zemlin M, et al. Inflammatory mediators in tracheal aspirates of preterm infants participating in a randomized trial of inhaled nitric oxide. PLoS One. 2017;12:e0169352.
Watkins PL, Dagle JM, Bell EF, Colaizy TT. Outcomes at 18 to 22 months of corrected age for infants born at 22 to 25 weeks of gestation in a center practicing active management. J Pediatr. 2020;217:52–58.e1.
Author information
Authors and Affiliations
Contributions
TJB designed the study, acquired data, drafted the initial manuscript, revised the manuscript, and approved the final version of the manuscript. JMD acquired data, revised the manuscript, and approved the final version of the manuscript. JMK acquired data, revised the manuscript, and approved the final version of the manuscript. DRR acquired data, revised the manuscript, and approved the final version of the manuscript. PMJ contributed to the design of the study, acquired data, revised the manuscript, and approved the final version of the manuscript. REG conceptualized and designed the study, analyzed and interpreted data, revised the manuscript, and approved the final version of the manuscript. All authors agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Boly, T.J., Dagle, J.M., Klein, J.M. et al. Response categorization and outcomes in extremely premature infants born at 22–26 weeks gestation that received inhaled nitric oxide for hypoxic respiratory failure. J Perinatol 43, 324–331 (2023). https://doi.org/10.1038/s41372-022-01582-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-022-01582-4