Abstract
Objective
To examine the association between probiotic use and antimicrobial utilization.
Study design
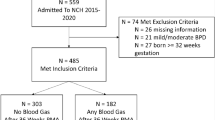
We retrospectively evaluated very-low-birth-weight (VLBW) infants admitted to tertiary neonatal intensive care units in Canada between 2014 and 2019. Our outcome was antimicrobial utilization rate (AUR) defined as number of days of antimicrobial exposure per 1000 patient-days.
Result
Of 16,223 eligible infants, 7279 (45%) received probiotics. Probiotic use rate increased from 10% in 2014 to 68% in 2019. The AUR was significantly lower in infants who received probiotics vs those who did not (107 vs 129 per 1000 patient-days, aRR = 0.89, 95% CI [0.81, 0.98]). Among 13,305 infants without culture-proven sepsis or necrotizing enterocolitis ≥Stage 2, 5931 (45%) received probiotics. Median AUR was significantly lower in the probiotic vs the no-probiotic group (78 vs 97 per 1000 patient-days, aRR = 0.85, 95% CI [0.74, 0.97]).
Conclusion
Probiotic use was associated with a significant reduction in AUR among VLBW infants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Osowicki J, Gwee A, Noronha J, Britton PN, Isaacs D, Lai TB, et al. Australia-wide point prevalence survey of antimicrobial prescribing in neonatal units: how much and how good? Pediatr Infect Dis J. 2015;34:e185–190.
Patel SJ, Oshodi A, Prasad P, Delamora P, Larson E, Zaoutis T, et al. Antibiotic use in neonatal intensive care units and adherence with Centers for Disease Control and Prevention 12 Step Campaign to Prevent Antimicrobial Resistance. Pediatr Infect Dis J. 2009;28:1047–51.
Pammi M, Hollister E, Neu J. Gut injury and the microbiome in neonates. Clin Perinatol. 2020;47:369–82.
Schwartz DJ, Langdon AE, Dantas G. Understanding the impact of antibiotic perturbation on the human microbiome. Genome Med. 2020;12:82. https://doi.org/10.1186/s13073-020-00782-x.
Ting JY, Synnes A, Roberts A, Deshpandey A, Dow K, Yoon E, et al. Association of antibiotic utilization and neonatal outcomes in very-low-birth-weight infants without proven sepsis. JAMA Pediatrics. 2016;170:1181–7.
Ting JY, Synnes A, Roberts A, Deshpandey AC, Dow K, Yang J, et al. Association of antibiotic utilization and neurodevelopmental outcomes among extremely low gestational age neonates without proven sepsis or necrotizing enterocolitis. Am J Perinatol. 2018;35:972–8.
Sharif S, Meader N, Oddie SJ, Rojas-Reyes MX, McGuire W. Probiotics to prevent necrotising enterocolitis in very preterm or very low birth weight infants. Cochrane Database Syst Rev. 2020;10:CD005496.
Principi N, Gnocchi M, Gagliardi M, Argentiero A, Neglia C, Esposito S. Prevention of clostridium difficile infection and associated diarrhea: an unsolved problem. Microorganisms. 2020;8:1640.
King S, Tancredi D, Lenoir-Wijnkoop I, Gould K, Vann H, Connors G, et al. Does probiotic consumption reduce antibiotic utilization for common acute infections? A systematic review and meta-analysis. Eur J Public Health. 2019;29:494–9.
Shah PS, Seidlitz W, Chan P, Yeh S, Musrap N, Lee SK, et al. Internal audit of the Canadian Neonatal Network data collection system. Am J Perinatol. 2017;34:1241–9.
Singh B, Shah PS, Afifi J, Simpson CD, Mitra S, Dow K, et al. Probiotics for preterm infants: a national retrospective cohort study. J Perinatol. 2019;39:533–9.
Health Canada Government of Canada. Licensed Natural Health Products Database (LNHPD). (Accessed 24th May 2021). https://health-products.canada.ca/lnhpd-bdpsnh/index-eng.jsp.
Canadian Neonatal Network. CNN Abstractor’s Mannual. (Accessed May 24, 2021) http://www.canadianneonatalnetwork.org/portal/Portals/0/CNN%20Manual_19Sep2011.pdf.
Richardson DK, Corcoran JD, Escobar GJ, Lee SK. SNAP-II and SNAPPE-II: simplified newborn illness severity and mortality risk scores. J Pediatr. 2001;138:92–100.
Beltempo M, Shah PS, Ye XY, Afifi J, Lee S, McMillan DD, et al. SNAP-II for prediction of mortality and morbidity in extremely preterm infants. J Matern Fetal Neonatal Med. 2019;32:2694–701.
Bell MJ, Ternberg JL, Feigin RD, Keating JP, Marshall R, Barton L, et al. Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann Surg. 1978;187:1–7.
Lui K, Vento M, Modi N, Kusuda S, Lehtonen L, Hakansson S, et al. Inter-center variability in neonatal outcomes of preterm infants: a longitudinal evaluation of 298 neonatal units in 11 countries. Semin Fetal Neonatal Med. 2021;26:101196.
Morgan RL, Preidis GA, Kashyap PC, Weizman AV, Sadeghirad B, McMaster Probiotic P, et al. Probiotics reduce mortality and morbidity in preterm, low-birth-weight infants: a systematic review and network meta-analysis of randomized trials. Gastroenterology. 2020;159:467–80.
Suez J, Zmora N, Segal E, Elinav E. The pros, cons, and many unknowns of probiotics. Nat Med. 2019;25:716–29.
Nguyen M, Holdbrooks H, Mishra P, Abrantes MA, Eskew S, Garma M, et al. Impact of probiotic B. infantis EVC001 feeding in premature infants on the gut microbiome, nosocomially acquired antibiotic resistance, and enteric inflammation. Front Pediatr. 2021;9:618009.
Stiemsma LT, Michels KB. The role of the microbiome in the developmental origins of health and disease. Pediatrics. 2018;141:e20172437.
Tamburini S, Shen N, Wu HC, Clemente JC. The microbiome in early life: implications for health outcomes. Nat Med. 2016;22:713–22.
Russell JT, Lauren Ruoss J, de la Cruz D, Li N, Bazacliu C, Patton L, et al. Antibiotics and the developing intestinal microbiome, metabolome and inflammatory environment in a randomized trial of preterm infants. Sci Rep. 2021;11:1943.
Alcon-Giner C, Dalby MJ, Caim S, Ketskemety J, Shaw A, Sim K, et al. Microbiota supplementation with bifidobacterium and lactobacillus modifies the preterm infant gut microbiota and metabolome: an observational study. Cell Rep Med. 2020;1:100077.
Barrett E, Kerr C, Murphy K, O’Sullivan O, Ryan CA, Dempsey EM, et al. The individual-specific and diverse nature of the preterm infant microbiota. Arch Dis Child Fetal Neonatal Ed. 2013;98:F334–340.
Baldassarre ME, Di Mauro A, Capozza M, Rizzo V, Schettini F, Panza R, et al. Dysbiosis and prematurity: is there a role for probiotics? Nutrients. 2019;11:1273.
Henrick BM, Chew S, Casaburi G, Brown HK, Frese SA, Zhou Y, et al. Colonization by B. infantis EVC001 modulates enteric inflammation in exclusively breastfed infants. Pediatr Res. 2019;86:749–57.
Casaburi G, Duar RM, Vance DP, Mitchell R, Contreras L, Frese SA, et al. Early-life gut microbiome modulation reduces the abundance of antibiotic-resistant bacteria. Antimicrob Resist Infect Control. 2019;8:131.
Underwood MA, Sohn K. The microbiota of the extremely preterm infant. Clin Perinatol. 2017;44:407–27.
Mueller NT, Differding MK, Ostbye T, Hoyo C, Benjamin-Neelon SE. Association of birth mode of delivery with infant faecal microbiota, potential pathobionts, and short chain fatty acids: a longitudinal study over the first year of life. BJOG. 2020;128:1293–303.
Arshad M, Seed PC. Urinary tract infections in the infant. Clin Perinatol. 2015;42:17–28. vii
Pramanik AK, Rangaswamy N, Gates T. Neonatal respiratory distress: a practical approach to its diagnosis and management. Pediatr Clin North Am. 2015;62:453–69.
Beltempo M, Shah P, Yoon E, Chan P, Members of the Annual Report Committee. The Canadian Neonatal Network Annual Report 2019. (Accessed May 20, 2021). http://www.canadianneonatalnetwork.org/portal/Portals/0/Annual%20Reports/2019%20CNN%20report%20final_links.pdf.
Shah P, Yoon E, Chan P, Members of the Annual Report Committee. The Canadian Neonatal Network Annual Report 2016. (Accessed May 20, 2021). http://www.canadianneonatalnetwork.org/portal/Portals/0/Annual%20Reports/2016%20CNN%20report_final_amendment1.pdf.
Schuller SS, Kramer BW, Villamor E, Spittler A, Berger A, Levy O. Immunomodulation to prevent or treat neonatal sepsis: past, present, and future. Front Pediatr. 2018;6:199.
Underwood MA. Arguments for routine administration of probiotics for NEC prevention. Curr Opin Pediatr. 2019;31:188–94.
Acknowledgements
The authors thank all site investigators and data abstractors of the Canadian Neonatal Network (CNN). A list of participating CNN site investigators and their affiliations is presented in the Supplementary Infromation. We thank the staff at the Maternal-infant Care Research Centre (MiCare) at Mount Sinai Hospital in Toronto, Ontario, Canada, for organizational support of the CNN. In particular, we thank Heather McDonald Kinkaid, PhD, a scientific writer at MiCare, for editorial support in preparing this paper.
Funding
Although no specific funding was received for this study, organizational support for the Canadian Neonatal Network was provided by the Maternal-infant Care Research Centre (MiCare) at Mount Sinai Hospital in Toronto, Ontario, Canada. MiCare is supported by the Canadian Institutes of Health Research (CTP 87518), the Ontario Ministry of Health and Long-Term Care, and Mount Sinai Hospital. Dr JYT receives salary support from the Investigator Grant Award Program of the British Columbia Children’s Hospital Research Institute.
Author information
Authors and Affiliations
Consortia
Contributions
JYT conceptualized and designed the study, interpreted the data, drafted the paper, and critically revised the paper for important intellectual content. EWY conducted the statistical analysis, interpreted the data, and critically revised the paper. PSS conceptualized and designed the study, contributed to the statistical analysis and interpreted the data, critically revised the paper for important intellectual content, obtained funding, and provided technical and material support as well as overall study supervision. CAF, TD, and VB interpreted the study data and critically reviewed and revised the paper for important intellectual content. All authors approved the final version of this paper as submitted, and all agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ting, J.Y., Yoon, E.W., Fajardo, C.A. et al. Antimicrobial utilization in very-low-birth-weight infants: association with probiotic use. J Perinatol 42, 947–952 (2022). https://doi.org/10.1038/s41372-022-01382-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-022-01382-w