Abstract
Objective
The aim of this study was to determine in utero fetal-placental growth patterns using in vivo three-dimensional (3D) quantitative magnetic resonance imaging (qMRI).
Study design
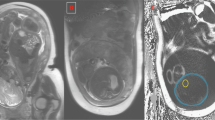
Healthy women with singleton pregnancies underwent fetal MRI to measure fetal body, placenta, and amniotic space volumes. The fetal-placental ratio (FPR) was derived using 3D fetal body and placental volumes (PV). Descriptive statistics were used to describe the association of each measurement with increasing gestational age (GA) at MRI.
Results
Fifty-eight (58) women underwent fetal MRI between 16 and 38 completed weeks gestation (mean = 28.12 ± 6.33). PV and FPR varied linearly with GA at MRI (rPV,GA = 0.83, rFPR,GA = 0.89, p value < 0.001). Fetal volume varied non-linearly with GA (p value < 0.01).
Conclusions
We describe in-utero growth trajectories of fetal-placental volumes in healthy pregnancies using qMRI. Understanding healthy in utero development can establish normative benchmarks where departures from normal may identify early in utero placental failure prior to the onset of fetal harm.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Burton GJ, Fowden AL, Thornburg KL. Placental origins of chronic disease. Physiological Rev. 2016;96:1509–65.
Jansson T, Powell TL. Role of the placenta in fetal programming: underlying mechanisms and potential interventional approaches. Clin Sci. 2007;113:1–13.
Morgan TK. Role of the placenta in preterm birth: a review. Am J Perinatol. 2016;33:258–66.
Salavati N, Smies M, Ganzevoort W, Charles AK, Erwich JJ, Plosch T, et al. The possible role of placental morphometry in the detection of fetal growth restriction. Front Physiol. 2018;9:1884.
Silver RM. Examining the link between placental pathology, growth restriction, and stillbirth. Best Pract Res Clin Obstet Gynaecol. 2018;49:89–102.
Gheorghe CP, Goyal R, Mittal A, Longo LD. Gene expression in the placenta: maternal stress and epigenetic responses. Int J Dev Biol. 2010;54:507–23.
Zur RL, Kingdom JC, Parks WT, Hobson SR. The placental basis of fetal growth restriction. Obstet Gynecol Clin North Am. 2020;47:81–98.
Al-Haddad BJS, Oler E, Armistead B, Elsayed NA, Weinberger DR, Bernier R, et al. The fetal origins of mental illness. Am J Obstet Gynecol. 2019;221:549–62.
Thornburg KL, Kolahi K, Pierce M, Valent A, Drake R, Louey S. Biological features of placental programming. Placenta. 2016;48:S47–S53.
Hayward CE, Lean S, Sibley CP, Jones RL, Wareing M, Greenwood SL, et al. Placental adaptation: what can we learn from birthweight: placental weight ratio? Front Physiol. 2016;7:28.
Salavati N, Gordijn SJ, Sovio U, Zill EHR, Gebril A, Charnock-Jones DS, et al. Birth weight to placenta weight ratio and its relationship to ultrasonic measurements, maternal and neonatal morbidity: a prospective cohort study of nulliparous women. Placenta. 2018;63:45–52.
Fowden AL, Sferruzzi-Perri AN, Coan PM, Constancia M, Burton GJ. Placental efficiency and adaptation: endocrine regulation. J Physiol. 2009;587:3459–72.
Ganer Herman H, Miremberg H, Schreiber L, Bar J, Kovo M. The association between disproportionate birth weight to placental weight ratio, clinical outcome, and placental histopathological lesions. Fetal Diagn Ther. 2017;41:300–6.
Alfirevic Z, Stampalija T, Dowswell T. Fetal and umbilical Doppler ultrasound in high-risk pregnancies. Cochrane Database Syst Rev. 2017;6:CD007529.
Lindqvist PG, Molin J. Does antenatal identification of small-for-gestational age fetuses significantly improve their outcome? Ultrasound Obstet Gynecol. 2005;25:258–64.
Narendran N, Yusuf K. Fetal doppler assessment in neonatal care: analysis of fetal doppler abnormalities and neonatal outcomes. Neoreviews. 2019;20:e575–e583.
Allen BC, Leyendecker JR. Placental evaluation with magnetic resonance. Radio Clin North Am. 2013;51:955–66.
Andescavage N, Dahdouh S, Jacobs M, Yewale S, Bulas D, Iqbal S, et al. In vivo textural and morphometric analysis of placental development in healthy & growth-restricted pregnancies using magnetic resonance imaging. Pediatr Res. 2019;85:974–81.
Andescavage N, duPlessis A, Metzler M, Bulas D, Vezina G, Jacobs M, et al. In vivo assessment of placental and brain volumes in growth-restricted fetuses with and without fetal Doppler changes using quantitative 3D MRI. J Perinatol. 2017;37:1278–84.
Andescavage N, Yarish A, Donofrio M, Bulas D, Evangelou I, Vezina G, et al. 3-D volumetric MRI evaluation of the placenta in fetuses with complex congenital heart disease. Placenta. 2015;36:1024–30.
Andescavage NN, du Plessis A, Limperopoulos C. Advanced MR imaging of the placenta: exploring the in utero placenta-brain connection. Semin Perinatol. 2015;39:113–23.
Dahdouh S, Andescavage N, Yewale S, Yarish A, Lanham D, Bulas D, et al. In vivo placental MRI shape and textural features predict fetal growth restriction and postnatal outcome. J Magn Reson Imaging. 2018;47:449–58.
Gudmundsson S, Dubiel M, Sladkevicius P. Placental morphologic and functional imaging in high-risk pregnancies. Semin Perinatol. 2009;33:270–80.
Sohn YS, Kim MJ, Kwon JY, Kim YH, Park YW. The usefulness of fetal MRI for prenatal diagnosis. Yonsei Med J. 2007;48:671–7.
Yushkevich PA, Piven J, Hazlett HC, Smith RG, Ho S, Gee JC, et al. User-guided 3D active contour segmentation of anatomical structures: significantly improved efficiency and reliability. Neuroimage. 2006;31:1116–28.
Zun Z, Zaharchuk G, Andescavage NN, Donofrio MT, Limperopoulos C. Non-invasive placental perfusion imaging in pregnancies complicated by fetal heart disease using velocity-selective arterial spin labeled MRI. Sci Rep. 2017;7:16126.
Yushkevich PA, Piven J, Hazlett, HC, Smith RG, Ho S, Gee JC, et al. User-guided 3D active contour segmentation of anatomical structures: Significantly improved efficiency and reliability. Neuroimage 2006;31:1116–28.
Zou KH, Wells WM 3rd, Kikinis R, Warfield SK. Three validation metrics for automated probabilistic image segmentation of brain tumours. Stat Med. 2004;23:1259–82.
Duncan KR, Sahota DS, Gowland PA, Moore R, Chang A, Baker PN, et al. Multilevel modeling of fetal and placental growth using echo-planar magnetic resonance imaging. J Soc Gynecol Investig. 2001;8:285–90.
Fenton TR, Kim JH. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pediatr. 2013;13:59.
Team RC. R: A language and environment for statistical computing. 2020. https://www.R-project.org/.
Baschat AA. Fetal responses to placental insufficiency: an update. BJOG. 2004;111:1031–41.
Langhoff L, Gronbeck L, von Huth S, Axelsson A, Jorgensen C, Thomsen C, et al. Placental growth during normal pregnancy—a magnetic resonance imaging study. Gynecol Obstet Investig. 2017;82:462–7.
Leon RL, Li KT, Brown BP. A retrospective segmentation analysis of placental volume by magnetic resonance imaging from first trimester to term gestation. Pediatr Radio. 2018;48:1936–44.
Carlin A, Kadji C, De Angelis R, Cannie MM, Jani JC. Prenatal prediction of small-for-gestational age neonates using MR imaging: comparison with conventional 2D ultrasound. J Matern Fetal Neonatal Med. 2019;32:1673–81.
Kadji C, Cannie MM, Resta S, Guez D, Abi-Khalil F, De Angelis R, et al. Magnetic resonance imaging for prenatal estimation of birthweight in pregnancy: review of available data, techniques, and future perspectives. Am J Obstet Gynecol. 2019;220:428–39.
Malin GL, Bugg GJ, Takwoingi Y, Thornton JG, Jones NW. Antenatal magnetic resonance imaging versus ultrasound for predicting neonatal macrosomia: a systematic review and meta-analysis. BJOG: Int J Obstet Gynaecol. 2016;123:77–88.
Baker PN, Johnson IR, Gowland PA, Hykin J, Harvey PR, Freeman A, et al. Fetal weight estimation by echo-planar magnetic resonance imaging. Lancet. 1994;343:644–5.
Uotila J, Dastidar P, Heinonen T, Ryymin P, Punnonen R, Laasonen E. Magnetic resonance imaging compared to ultrasonography in fetal weight and volume estimation in diabetic and normal pregnancy. Acta Obstet Gynecol Scand. 2000;79:255–9.
Kubik-Huch RA, Wildermuth S, Cettuzzi L, Rake A, Seifert B, Chaoui R, et al. Fetus and uteroplacental unit: fast MR imaging with three-dimensional reconstruction and volumetry-feasibility study. Radiology. 2001;219:567–73.
Kacem Y, Cannie MM, Kadji C, Dobrescu O, Lo Zito L, Ziane S, et al. Fetal weight estimation: comparison of two-dimensional US and MR imaging assessments. Radiology. 2013;267:902–10.
Kadji C, De Groof M, Camus MF, De Angelis R, Fellas S, Klass M, et al. The use of a software-assisted method to estimate fetal weight at and near term using magnetic resonance imaging. Fetal Diagn Ther. 2017;41:307–13.
Kadji C, Camus MF, Bevilacqua E, Cannie MM, Sanchez TC, Jani JC. Repeatability of estimated fetal weight: comparison between MR imaging versus 2D ultrasound in at- and near-term patients. Eur J Radio. 2017;91:35–40.
Kadji C, Cannie MM, De Angelis R, Camus M, Klass M, Fellas S, et al. Prenatal prediction of postnatal large-for-dates neonates using a simplified MRI method: comparison with conventional 2D ultrasound estimates. Ultrasound Obstet Gynecol. 2018;52:250–7.
Kadji C, Cannie MM, Van Wettere M, Bevilacqua E, Dutemeyer V, Strizek B, et al. A longitudinal study on fetal weight estimation at third trimester of pregnancy: comparison of magnetic resonance imaging and 2-D ultrasound predictions. Fetal Diagn Ther. 2017;42:181–8.
Kadji C, Bevilacqua E, Hurtado I, Carlin A, Cannie MM, Jani JC. Comparison of conventional 2D ultrasound to magnetic resonance imaging for prenatal estimation of birthweight in twin pregnancy. Am J Obstet Gynecol. 2018;218:128 e121–128 e111.
Gilbert WM, Danielsen B. Pregnancy outcomes associated with intrauterine growth restriction. Am J Obstet Gynecol. 2003;188:1596–9.
Chauhan SP, Rice MM, Grobman WA, Bailit J, Reddy UM, Wapner RJ, et al. Neonatal morbidity of small- and large-for-gestational-age neonates born at term in uncomplicated pregnancies. Obstet Gynecol. 2017;130:511–9.
Doty MS, Chen HY, Sibai BM, Chauhan SP. Maternal and neonatal morbidity associated with early term delivery of large-for-gestational-age but nonmacrosomic neonates. Obstet Gynecol. 2019;133:1160–6.
Rustogi D, Synnes A, Alshaikh B, Hasan S, Drolet C, Masse E, et al. Neurodevelopmental outcomes of singleton large for gestational age infants <29 weeks’ gestation: a retrospective cohort study. J Perinatol. 2021;41:1313–21.
Christians JK, Grynspan D, Greenwood SL, Dilworth MR. The problem with using the birthweight: placental weight ratio as a measure of placental efficiency. Placenta. 2018;68:52–8.
Didier RA, Khrichenko D, Barrera CA, Maya C, Johnson AM, Oliver ER, et al. Novel computerized analytic technique for quantification of amniotic fluid volume in fetal MRI. AJR Am J Roentgenol. 2019;213:W149–W152.
Hilliard NJ, Hawkes R, Patterson AJ, Graves MJ, Priest AN, Hunter S, et al. Amniotic fluid volume: rapid MR-based assessment at 28-32 weeks gestation. Eur Radio. 2016;26:3752–9.
Kehl S, Schelkle A, Thomas A, Puhl A, Meqdad K, Tuschy B, et al. Single deepest vertical pocket or amniotic fluid index as evaluation test for predicting adverse pregnancy outcome (SAFE trial): a multicenter, open-label, randomized controlled trial. Ultrasound Obstet Gynecol. 2016;47:674–9.
Magann EF, Sandlin AT, Ounpraseuth ST. Amniotic fluid and the clinical relevance of the sonographically estimated amniotic fluid volume: oligohydramnios. J Ultrasound Med. 2011;30:1573–85.
Dubil EA, Magann EF. Amniotic fluid as a vital sign for fetal wellbeing. Australas J Ultrasound Med. 2013;16:62–70.
DeFreitas MJ, Katsoufis CP, Infante JC, Granda ML, Abitbol CL, Fornoni A. The old becomes new: advances in imaging techniques to assess nephron mass in children. Pediatr Nephrol. 2021;36:517–25.
Sun L, Guo C, Yao L, Zhang T, Wang J, Wang L, et al. Quantitative diagnostic advantages of three-dimensional ultrasound volume imaging for fetal posterior fossa anomalies: preliminary establishment of a prediction model. Prenat Diagn. 2019;39:1086–95.
Guerriero S, Alcazar JL, Pascual MA, Ajossa S, Perniciano M, Piras A, et al. Deep infiltrating endometriosis: comparison between 2-dimensional ultrasonography (US), 3-dimensional US, and magnetic resonance imaging. J Ultrasound Med. 2018;37:1511–21.
Goncalves LF, Lee W, Mody S, Shetty A, Sangi-Haghpeykar H, Romero R. Diagnostic accuracy of ultrasonography and magnetic resonance imaging for the detection of fetal anomalies: a blinded case-control study. Ultrasound Obstet Gynecol. 2016;48:185–92.
Acknowledgements
We would like to acknowledge our funding sources: Supported by National Institutes of Health (1U54HD090257, R01-HL116585, K23HD092585-01A1).
Author information
Authors and Affiliations
Contributions
AA was responsible for the manual data collection and providing original draft and final review of the manuscript. KK and NRA were responsible for MRI data collection, critical review and editing of the manuscript along with final review. DK was responsible for the semi-automated process in MR image analysis, critical review and editing of the manuscript along with final review. RI was responsible for statistical analysis and creating figures, critical review and editing of the manuscript along with final review. AB and CL were responsible for study design and objectives, interpreting results, critical review and editing of the manuscript along with final review. JQ was responsible for screening, approaching and enrolling eligible subjects, critical review and editing of the manuscript along with final review. ACG and HKA were responsible for identifying and arbitrating potentially eligible subjects, interpreting results, critical review and editing of the manuscript along with final review. NNA was responsible for study design and objectives, oversight of data collection and analysis, interpreting results, critical review and editing of the manuscript along with final review.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Amgalan, A., Kapse, K., Krishnamurthy, D. et al. Measuring intrauterine growth in healthy pregnancies using quantitative magnetic resonance imaging. J Perinatol 42, 860–865 (2022). https://doi.org/10.1038/s41372-022-01340-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-022-01340-6
This article is cited by
-
Fetal weight estimation based on deep neural network: a retrospective observational study
BMC Pregnancy and Childbirth (2023)