Abstract
Objective
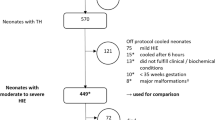
Determine whether blanket temperatures during therapeutic hypothermia (TH) are associated with 18–22 month outcomes for infants with hypoxic ischemic encephalopathy (HIE).
Study design
Retrospective cohort study of 181 infants with HIE who received TH in two randomized trials within the Neonatal Research Network. We defined summative blanket temperature constructs and evaluated for association with a primary composite outcome of death or moderate/ severe disability at 18–22 months.
Results
Each 0.5 °C above 33.5 °C in the mean of the highest quartile blanket temperature was associated with a 52% increase in the adjusted odds of death/ disability (aOR 1.52, 95% CI 1.09–2.11). Having >8 consecutive blanket temperatures above 33.5 °C rendered an aOR of death/disability of 5.04 in the first 24 h (95% CI 1.54–16.6) and 6.92 in the first 48 h (95% CI 2.20–21.8) of TH.
Conclusions
Higher blanket temperature during TH may be an early, clinically useful biomarker of HIE outcome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Shankaran S, Laptook AR, Ehrenkranz RA, Tyson JE, McDonald SA, Donovan EF, et al. Whole-body hypothermia for neonates with hypoxic-ischemic encephalopathy. N. Engl J Med. 2005;353:1574–84.
Gluckman PD, Wyatt JS, Azzopardi D, Ballard R, Edwards AD, Ferriero DM, et al. Selective head cooling with mild systemic hypothermia after neonatal encephalopathy: multicentre randomised trial. Lancet. 2005;365:663–70.
Azzopardi DV, Strohm B, Edwards AD, Dyet L, Halliday HL, Juszczak E, et al. Moderate hypothermia to treat perinatal asphyxial encephalopathy. N. Engl J Med. 2009;361:1349–58.
Simbruner G, Mittal RA, Rohlmann F, Muche R.neo.n EnTP Systemic hypothermia after neonatal encephalopathy: outcomes of neo.nEURO.network RCT. Pediatrics. 2010;126:e771–778.
Jacobs SE. Whole-body hypothermia for term and near-term newborns with hypoxic-ischemic encephalopathy. Arch pediatrics Adolesc Med. 2011;165:692.
Shankaran S, Laptook AR, Pappas A, McDonald SA, Das A, Tyson JE, et al. Effect of depth and duration of cooling on death or disability at age 18 months among neonates with hypoxic-ischemic encephalopathy: a randomized clinical trial. JAMA. 2017;318:57–67.
Ambalavanan N, Carlo WA, Shankaran S, Bann CM, Emrich SL, Higgins RD, et al. Predicting outcomes of neonates diagnosed with hypoxemic-ischemic encephalopathy. Pediatrics. 2006;118:2084–93.
Ambalavanan N, Shankaran S, Laptook AR, Carper BA, Das A, Carlo WA, et al. Early determination of prognosis in neonatal moderate or severe hypoxic-ischemic encephalopathy. Pediatrics. 2021;147:e2020048678.
Peeples ES, Rao R, Dizon MLV, Johnson YR, Joe P, Flibotte J, et al. Predictive models of neurodevelopmental outcomes after neonatal hypoxic-ischemic encephalopathy. Pediatrics. 2020;147:e2020022962.
Río RD, Ochoa C, Alarcon A, Arnáez J, Blanco D, García-Alix A. Amplitude integrated electroencephalogram as a prognostic tool in neonates with hypoxic-ischemic encephalopathy: a systematic review. PloS ONE. 2016;11:e0165744.
Shankaran S, Laptook AR, Tyson JE, Ehrenkranz RA, Bann CM, Das A, et al. Evolution of encephalopathy during whole body hypothermia for neonatal hypoxic-ischemic encephalopathy. J Pediatrics. 2012;160:567–72.
Laerhoven HV, Haan TRD, Offringa M, Post B, Lee JHVD. Prognostic tests in term neonates with hypoxic-ischemic encephalopathy: a systematic review. Pediatrics. 2013;131:88–98.
Shankaran S, McDonald SA, Laptook AR, Hintz SR, Barnes PD, Das A, et al. Neonatal magnetic resonance imaging pattern of brain injury as a biomarker of childhood outcomes following a trial of hypothermia for neonatal hypoxic-ischemic encephalopathy. J Pediatrics. 2015;167:987–93.
Lally PJ, Montaldo P, Oliveira V, Soe A, Swamy R, Bassett P, et al. Magnetic resonance spectroscopy assessment of brain injury after moderate hypothermia in neonatal encephalopathy: a prospective multicentre cohort study. Lancet Neurol. 2019;18:35–45.
Cheong JLY, Coleman L, Hunt RW, Lee KJ, Doyle LW, Inder TE, et al. Prognostic utility of magnetic resonance imaging in neonatal hypoxic-ischemic encephalopathy. Arch Pedia Adol Med. 2012;166:634–40.
Rutherford M, Ramenghi LA, Edwards AD, Brocklehurst P, Halliday H, Levene M, et al. Assessment of brain tissue injury after moderate hypothermia in neonates with hypoxic–ischaemic encephalopathy: a nested substudy of a randomised controlled trial. Lancet Neurol. 2010;9:39–45.
Shankaran S, Laptook AR, McDonald SA, Higgins RD, Tyson JE, Ehrenkranz RA, et al. Temperature profile and outcomes of neonates undergoing whole body hypothermia for neonatal hypoxic-ischemic encephalopathy. Pediatr Crit Care Med. 2012;13:53–9.
Laptook A, Tyson J, Shankaran S, McDonald S, Ehrenkranz R, Fanaroff A, et al. Elevated temperature after hypoxic-ischemic encephalopathy: risk factor for adverse outcomes. Pediatrics. 2008;122:491–9.
Laptook AR, McDonald SA, Shankaran S, Stephens BE, Vohr BR, Guillet R, et al. Elevated temperature and 6‐ to 7‐year outcome of neonatal encephalopathy. Ann Neurol. 2013;73:520–8.
Mietzsch U, Radhakrishnan R, Boyle FA, Juul S, Wood TR. Active cooling temperature required to achieve therapeutic hypothermia correlates with short‐term outcome in neonatal hypoxic‐ischaemic encephalopathy. J Physiol. 2020;598:415–24.
Burnard ED, Cross KW. Rectal temperature in the newborn after birth asphyxia. Bmj. 1958;2:1197–9.
Sahni R, Schulze K. Temperature control in newborn infants. In: Polin RA, Fox WW, Abman SH (eds). Fetal and Neonatal Physiology, 4th edn. Elsevier: Philadelphia, PA, 2011, pp 624–46.
Shankaran S, Laptook AR, Pappas A, McDonald SA, Das A, Tyson JE, et al. Effect of depth and duration of cooling on deaths in the NICU among neonates with hypoxic ischemic encephalopathy: a randomized clinical trial. JAMA. 2014;312:2629–39.
Bayley N Bayley Scales of Infant Development–II., 2nd edn. Psychological Corporation: San Antonio, TX, 1993.
Bayley N Manual for the Bayley Scales of Infant and Toddler Development-III, 3rd edn. Hardcourt Assessment: San Antonio, TX, 2006.
Palisano R, Rosenbaum P, Walter S, Russell D, Wood E, Galuppi B. Development and reliability of a system to classify gross motor function in children with cerebral palsy. Dev Med Child Neurol. 1997;39:214–23.
Bonifacio SL, McDonald SA, Chock VY, Wusthoff CJ, Hintz SR, Laptook AR, et al. Differences in patient characteristics and care practices between two trials of therapeutic hypothermia. Pediatr Res. 2019;85:1008–15.
Enweronu‐Laryea C, Martinello KA, Rose M, Manu S, Tann CJ, Meek J, et al. Core temperature after birth in babies with neonatal encephalopathy in a sub‐Saharan African hospital setting. J Physiol. 2019;597:4013–24.
Tsuda K, Iwata S, Mukai T, Shibasaki J, Takeuchi A, Ioroi T, et al. Body temperature, heart rate, and short-term outcome of cooled infants. Ther Hypothermia Tem. 2019;9:76–85.
Sant’Anna G, Laptook AR, Shankaran S, Bara R, McDonald SA, Higgins RD, et al. Phenobarbital and temperature profile during hypothermia for hypoxic-ischemic encephalopathy. J Child Neurol. 2011;27:451–7.
Bourgoin P, Barrault V, Loron G, Roger A, Bataille E, Leclair-Visonneau L, et al. Interrater agreement between critical care providers for background classification and seizure detection after implementation of amplitude-integrated electroencephalography in neonates, infants, and children. J Clin Neurophysiol Off Publ Am Electroencephalogr Soc. 2020;37:259–62.
Shankaran S, Pappas A, McDonald SA, Laptook AR, Bara R, Ehrenkranz RA, et al. Predictive value of an early amplitude integrated electroencephalogram and neurologic examination. Pediatrics. 2011;128:e112–120.
Acknowledgements
This work was supported and funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) and the Neonatal Research Network (NRN). Participating NRN sites collected data and transmitted it to RTI International, the data coordinating center (DCC) for the network, which stored, managed and analyzed the data for this study. On behalf of the NRN, RTI International had full access to all of the data in the study, and with the NRN Center Principal Investigators, takes responsibility for the integrity of the data and accuracy of the data analysis. We are indebted to our medical and nursing colleagues and the infants and their parents who agreed to take part in the original trials informing this study.
Author information
Authors and Affiliations
Consortia
Contributions
JF and KD conceived of this study and proposed initial design. ARL, SS, MCB, EFB, CMC, SBD, TLD, ECE, RH, EAJ, KPV critically reviewed the design and offered revisions. SAM and AD designed approach to and performed statistical analysis. JF drafted the manuscript and all authors critically reviewed and approved the final content.
Corresponding author
Ethics declarations
Competing interests
For the Induced Hypothermia trial: Dr. Cotten reports having served on the data and safety monitoring board for the Inhibitex phase 3 study of Vernonate for the prevention of infections in preterm infants.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Flibotte, J., Laptook, A.R., Shankaran, S. et al. Blanket temperature during therapeutic hypothermia and outcomes in hypoxic ischemic encephalopathy. J Perinatol 42, 348–353 (2022). https://doi.org/10.1038/s41372-021-01302-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-021-01302-4