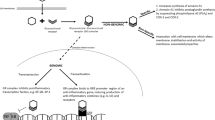
Abstract
Objective
To determine if extremely preterm (EPT) neonates receiving dexamethasone for the prevention of BPD have a higher incidence of presumed adrenal insufficiency (PAI).
Study design
Retrospective cohort study of neonates <28 weeks gestation examining PAI after dexamethasone use and PAI after intratracheal budesonide with surfactant administration.
Result
Of 332 neonates, 38% received dexamethasone. The incidence of PAI was higher in neonates who had received dexamethasone (20.8% vs 2.9%, p < 0.001). However, for intubated babies receiving surfactant, dexamethasone was not independently associated with increased PAI after adjusting for gestational age, birthweight, and race (aOR 2.92, 95% CI: 0.79–10.85). Dexamethasone was independently associated with increased PAI in infants previously receiving budesonide/surfactant treatment (aOR 5.38, 95% CI: 1.38–20.90).
Conclusion
The use of dexamethasone alone was not associated with increased PAI, when adjusted for prematurity-related factors. The combination of budesonide with dexamethasone was significantly associated with increased PAI.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Stoll BJ, Hansen NI, Bell EF, Walsh MC, Carlo WA, Shankaran S, et al. Trends in care practices, morbidity, and mortality of extremely preterm Neonates, 1993–2012. JAMA 2015;314:1039–51.
Foglia EE, Jensen EA, Kirpalani H. Delivery room interventions to prevent bronchopulmonary dysplasia in extremely preterm infants. J Perinatol. 2017;37:1171–9.
Doyle LW, Cheong JL, Ehrenkranz RA, Halliday HL. Early (<8 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants. Cochrane Database Syst Rev. 2017;10:CD001146.
Baud O, Maury L, Lebail F, Ramful D, El Moussawi F, Nicaise C, et al. Effect of early low-dose hydrocortisone on survival without bronchopulmonary dysplasia in extremely preterm infants (PREMILOC): A double-blind, placebo-controlled, multicentre, randomized trial. Lancet (Lond, Engl). 2016;387:1827–36.
Onland W, De Jaegere AP, Offringa M, van Kaam A. Systemic corticosteroid regimens for prevention of bronchopulmonary dysplasia in preterm infants. Cochrane Database Syst Rev. 2017;1:CD010941-CD.
Kothe TB, Sadiq FH, Burleyson N, Williams HL, Anderson C, Hillman NH. Surfactant and budesonide for respiratory distress syndrome: An observational study. Pediatr Res. 2020;87:940–5.
Venkataraman R, Kamaluddeen M, Hasan SU, Robertson HL, Lodha A. Intratracheal administration of budesonide-surfactant in prevention of bronchopulmonary dysplasia in very low birth weight infants: A systematic review and meta-analysis. Pediatr Pulmonol. 2017;52:968–75.
Yeh TF, Chen CM, Wu SY, Husan Z, Li TC, Hsieh WS, et al. Intratracheal administration of budesonide/surfactant to prevent bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2016;193:86–95.
Bassler D, Plavka R, Shinwell ES, Hallman M, Jarreau PH, Carnielli V, et al. Early inhaled budesonide for the prevention of bronchopulmonary dysplasia. N. Engl J Med. 2015;373:1497–506.
Doyle LW, Cheong JL, Ehrenkranz RA, Halliday HL. Late (>7 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants. Cochrane Database Syst Rev. 2017;10:CD001145.
Sauberan JB, Reed EM, Vaucher YV, Katheria A. Oliguria during hydrocortisone dosage wean in very low birth weight infants. Am J Perinatol. 2014;31:673–6.
Walther FJ, Findlay RD, Durand M. Adrenal suppression and extubation rate after moderately early low-dose dexamethasone therapy in very preterm infants. Early Hum Dev. 2003;74:37–45.
Buchman AL. Side effects of corticosteroid therapy. J Clin Gastroenterol. 2001;33:289–94.
Baud O, Trousson C, Biran V, Leroy E, Mohamed D, Alberti C, et al. Association between early low-dose hydrocortisone therapy in extremely preterm neonates and neurodevelopmental outcomes at 2 years of age. JAMA 2017;317:1329–37.
Brouwer MJ, Kersbergen KJ, van Kooij BJM, Benders M, van Haastert IC, Koopman-Esseboom C, et al. Preterm brain injury on term-equivalent age MRI in relation to perinatal factors and neurodevelopmental outcome at two years. PLoS One. 2017;12:e0177128.
Doyle LW, Halliday HL, Ehrenkranz RA, Davis PG, Sinclair JC. An update on the impact of postnatal systemic corticosteroids on mortality and cerebral palsy in preterm infants: effect modification by risk of bronchopulmonary dysplasia. J Pediatr. 2014;165:1258–60.
Laughon MM, Langer JC, Bose CL, Smith PB, Ambalavanan N, Kennedy KA, et al. Prediction of bronchopulmonary dysplasia by postnatal age in extremely premature infants. Am J Respir Crit Care Med. 2011;183:1715–22.
Mesiano S, Jaffe RB. Developmental and functional biology of the primate fetal adrenal cortex. Endocr Rev. 1997;18:378–403.
Fernandez EF, Watterberg KL. Relative adrenal insufficiency in the preterm and term infant. J Perinatol. 2009;29:S44–9.
Prelipcean I, Wynn JL, Thompson L, Burchfield DJ, James-Woodley L, Chase PB, et al. Absence of relationship between serum cortisol and critical illness in premature infants. Arch Dis Child Fetal Neonatal Ed. 2021;106:408–12.
Yeh TF, Lin HC, Chang CH, Wu TS, Su BH, Li TC, et al. Early intratracheal instillation of budesonide using surfactant as a vehicle to prevent chronic lung disease in preterm infants: a pilot study. Pediatrics 2008;121:e1310–8.
Zappitelli M, Ambalavanan N, Askenazi DJ, Moxey-Mims MM, Kimmel PL, Star RA, et al. Developing a neonatal acute kidney injury research definition: A report from the NIDDK neonatal AKI workshop. Pediatr Res. 2017;82:569–73.
Development of audit measures and guidelines for good practice in the management of neonatal respiratory distress syndrome. Report of a joint working group of the British association of perinatal medicine and the research unit of the royal college of physicians. Arch Dis Child. 1992;67:1221–7.
Fenton TR, Kim JH. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pediatr. 2013;13:59.
Halliday HL. Update on Postnatal Steroids. Neonatology 2017;111:415–22.
Virkud YV, Hornik CP, Benjamin DK, Laughon MM, Clark RH, Greenberg RG, et al. Respiratory support for very low birth weight infants receiving Dexamethasone. J Pediatr. 2017;183:26–30.
Harmon HM, Jensen EA, Tan S, Chaudhary AS, Slaughter JL, Bell EF, et al. Timing of postnatal steroids for bronchopulmonary dysplasia: Association with pulmonary and neurodevelopmental outcomes. J Perinatol. 2020;40:616–27.
Esmailpour N, Hogger P, Rohdewald P. Binding kinetics of budesonide to the human glucocorticoid receptor. Eur J Pharm Sci. 1998;6:219–23.
Ng PC, Lee CH, Lam CW, Ma KC, Fok TF, Chan IH, et al. Transient adrenocortical insufficiency of prematurity and systemic hypotension in very low birthweight infants. Arch Dis Child Fetal Neonatal Ed. 2004;89:F119–26.
Huysman MW, Hokken-Koelega AC, De Ridder MA, Sauer PJ. Adrenal function in sick very preterm infants. Pediatr Res. 2000;48:629–33.
Fernandez EF, Montman R, Watterberg KL. ACTH and cortisol response to critical illness in term and late preterm newborns. J Perinatol. 2008;28:797–802.
Baud O, Watterberg KL. Prophylactic postnatal corticosteroids: Early hydrocortisone. Semin Fetal Neonatal Med. 2019;24:202–6.
Ng SM, Ogundiya A, Didi M, Turner MA. Adrenal function of extremely premature infants in the first 5 days after birth. J Pediatr Endocrinol Metab. 2019;32:363–7.
Kawai M. Late-onset circulatory collapse of prematurity. Pediatr Int. 2017;59:391–6.
Yasuoka K, Inoue H, Egami N, Ochiai M, Tanaka K, Sawano T, et al. Late-onset circulatory collapse and risk of cerebral palsy in extremely preterm infants. J Pediatr. 2019;212:117–23.e4.
Lee WJ, Kim MY, Cho HJ, Lee JS, Son DW. Clinical features of late-onset circulatory collapse in preterm infants. Korean. J Perinatol. 2013;24:148–57.
Marinelli KC, Lyden ER, Peeples ES. Clinical risk factors for the development of late-onset circulatory collapse in premature infants. Pediatr Res. 2021;89:968–73.
Suzuki Y, Kono Y, Hayakawa T, Shimozawa H, Matano M, Yada Y. Neonatal factors related to center variation in the incidence of late-onset circulatory collapse in extremely preterm infants. PLoS One. 2018;13:1–11.
Doyle LW, Ehrenkranz RA, Halliday HL. Dexamethasone treatment after the first week of life for bronchopulmonary dysplasia in preterm infants: A systematic review. Neonatology 2010;98;289–96.
Onland W, Cools F, Kroon A, Rademaker K, Merkus MP, Dijk PH, et al. Effect of hydrocortisone therapy initiated 7 to 14 days after birth on mortality or bronchopulmonary dysplasia among very preterm infants receiving mechanical ventilation. JAMA 2019;321:354.
Watterberg KL, Scott SM. Evidence of early adrenal insufficiency in babies who develop bronchopulmonary dysplasia. Pediatrics 1995;95:120–5.
Stanley JS. Pharmacodynamics and pharmacokinetics of budesonide: A new nebulized corticosteroid. J Allergy Clin Immunol. 1999;104:S175–S83.
Hillman NH, Abugisisa L, Royse E, Fee E, Kemp MW, Kramer BW, et al. Dose of budesonide with surfactant affects lung and systemic inflammation after normal and injurious ventilation in preterm lambs. Pediatr Res. 2020;88:726–32.
McEvoy CT, Ballard PL, Ward RM, Rower JE, Wadhawan R, Hudak ML, et al. Dose-escalation trial of budesonide in surfactant for prevention of bronchopulmonary dysplasia in extremely low gestational age high-risk newborns (SASSIE). Pediatr Res. 2020;88:629–36.
Masumoto K, Kusuda S, Aoyagi H, Tamura Y, Obonai T, Yamasaki C, et al. Comparison of serum cortisol concentrations in preterm infants with or without late-onset circulatory collapse due to adrenal insufficiency of prematurity. Pediatr Res. 2008;63:686–90.
Author information
Authors and Affiliations
Contributions
JJ conceptualized the study, wrote and reviewed the draft as submitted. NH conceptualized the study, provided interpretation of the results, and reviewed the draft as submitted. LL was engaged in the study design and interpretation of the results and reviewed the final draft as submitted. BH and BH wrote early drafts of the manuscript, conceptualized the study, and approved the final draft as submitted. HW extracted the data from the EMR and reviewed the final manuscript as submitted. CA ensured the integrity of the data extracted from the EMR, provided interpretation of the results, and reviewed the final document as submitted. PB analyzed and provided interpretation of the data and edited the final manuscript as submitted.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study (#28487) was approved by the Saint Louis University Institutional Review Board with a waiver of HIPAA authorization. The study was performed in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Josephsen, J.B., Hemmann, B.M., Anderson, C.D. et al. Presumed adrenal insufficiency in neonates treated with corticosteroids for the prevention of bronchopulmonary dysplasia. J Perinatol 42, 65–71 (2022). https://doi.org/10.1038/s41372-021-01251-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-021-01251-y