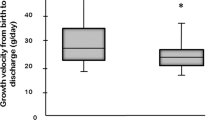
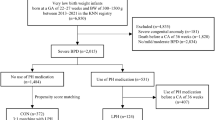
Abstract
Objectives
To identify the relationship between prophylactic indomethacin (PI) administration and (1) mortality and bronchopulmonary dysplasia (BPD) at 36-week postmenstrual age (PMA) (primary outcome), and (2) to evaluate for PI-associated acute kidney injury.
Study design
Retrospective cohort investigation of 22–28 weeks gestation infants (N = 1167) who were admitted to Nationwide Children’s Hospital on postnatal days 0–1 between May 2009 and September 2017 and survived ≥24-h postnatal. The associations of PI treatment with mortality or BPD, and other secondary outcomes, were evaluated via multivariable robust-error-variance Poisson regression.
Results
The adjusted risks of death or BPD (1.02, 95% CI: 0.83, 1.25), BPD (0.97, 95% CI: 0.77, 1.21), and death 1.33 (95% CI: 0.84, 2.10) by 36-week PMA were unchanged following PI treatment after multivariable adjustment. No changes in mean creatinine levels were detected in exposed versus unexposed infants to suggest PI-induced AKI.
Conclusion
Prophylactic indomethacin treatment was unrelated to mortality or BPD outcomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Semberova J, Sirc J, Miletin J, Kucera J, Berka I, Sebkova S, et al. Spontaneous closure of patent ductus arteriosus in infants ≤1500 g. Pediatrics. 2017;140:e20164258.
Slaughter JL, Cua CL, Notestine JL, Rivera BK, Marzec L, Hade EM, et al. Early prediction of spontaneous Patent Ductus Arteriosus (PDA) closure and PDA-associated outcomes: a prospective cohort investigation. BMC Pediatrics. 2019;19:333.
Fowlie PW, Davis PG, McGuire W. Prophylactic intravenous indomethacin for preventing mortality and morbidity in preterm infants. Cochrane Database Syst Rev. 2010;7:CD000174.
Schmidt B, Davis P, Moddemann D, Ohlsson A, Roberts RS, Saigal S, et al. Long-term effects of indomethacin prophylaxis in extremely-low-birth-weight infants. N Engl J Med. 2001;344:1966–1972.
Slaughter JL, Reagan PB, Newman TB, Klebanoff MA. Comparative effectiveness of nonsteroidal anti-inflammatory drug treatment vs no treatment for patent ductus arteriosus in preterm infants. JAMA Pediatr. 2017;171:e164354.
Jensen EA, Dysart KC, Gantz MG, Carper B, Higgins RD, Keszler M, et al. Association between use of prophylactic indomethacin and the risk for bronchopulmonary dysplasia in extremely preterm infants. J Pediatr. 2017;186:34–40.e32.
Fowlie PW. Prophylactic indomethacin: systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed. 1996;74:F81–F87.
Jensen EA, Foglia EE, Schmidt B. Association between prophylactic indomethacin and death or bronchopulmonary dysplasia: A systematic review and meta-analysis of observational studies. Semin Perinatol. 2018;42:228–234.
Liebowitz M, Clyman RI. Prophylactic indomethacin compared with delayed conservative management of the patent ductus arteriosus in extremely preterm infants: effects on neonatal outcomes. J Pediatr. 2017;187:119–126.e111.
Nelin TD, Pena E, Giacomazzi T, Lee S, Logan JW, Moallem M, et al. Outcomes following indomethacin prophylaxis in extremely preterm infants in an all-referral NICU. J Perinatol. 2017;37:932–937.
Papile LA, Burstein J, Burstein R, Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J Pediatr. 1978;92:529–534.
Couser RJ, Ferrara TB, Wright GB, Cabalka AK, Schilling CG, Hoekstra RE, et al. Prophylactic indomethacin therapy in the first twenty-four hours of life for the prevention of patent ductus arteriosus in preterm infants treated prophylactically with surfactant in the delivery room. J Pediatr. 1996;128:631–637.
Ment LR, Oh W, Ehrenkranz RA, Philip AG, Vohr B, Allan W, et al. Low-dose indomethacin and prevention of intraventricular hemorrhage: a multicenter randomized trial. Pediatrics 1994;93:543–550.
Akima S, Kent A, Reynolds G, Gallagher M, Falk M. Indomethacin and renal impairment in neonates. Pediatr Nephrol. 2004;19:490–493.
Cifuentes RF, Olley PM, Balfe JW, Radde IC, Soldin SJ. Indomethacin and renal function in premature infants with persistent patent ductus arteriosus. J Pediatr. 1979;95:583–587.
Gersony WM, Peckham GJ, Ellison RC, Miettinen OS, Nadas AS. Effects of indomethacin in premature infants with patent ductus arteriosus: results of a national collaborative study. J Pediatr. 1983;102:895–906.
Shaffer CL, Gal P, Ransom JL, Carlos RQ, Smith MS, Davey AM, et al. Effect of age and birth weight on indomethacin pharmacodynamics in neonates treated for patent ductus arteriosus. Crit Care Med. 2002;30:343–348.
Bell MJ, Ternberg JL, Feigin RD, Keating JP, Marshall R, Barton L, et al. Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann Surg. 1978;187:1–7.
Alexander GR, Himes JH, Kaufman RB, Mor J, Kogan M. A United States national reference for fetal growth. Obstet Gynecol. 1996;87:163–168.
Pishevar N, Fathi O, Backes CH, Shepherd EG, Nelin LD. Predicting survival in infants born at <27 weeks gestation admitted to an all referral neonatal intensive care unit: a pilot study. J Perinatol. 2020;40:750–757.
Altit G, Basso O, Grandi SM, Yang S. Letter to the editor. J Perinatol. 2018;38:767.
Rolnitsky A, Lee SK, Piedbouf B, Harrison A, Shah PS, Canadian Neonatal N. Prophylactic interventions in neonatology: how do they fare in real life? Am J Perinatol. 2015;32:1098–1104.
Schmidt B, Roberts RS, Fanaroff A, Davis P, Kirpalani HM, Nwaesei C, et al. Indomethacin prophylaxis, patent ductus arteriosus, and the risk of bronchopulmonary dysplasia: Further analyses from the Trial of Indomethacin Prophylaxis in Preterms (TIPP). The. J Pediatr. 2006;148:730–734.e731.
Clyman RI. Recommendations for the postnatal use of indomethacin: an analysis of four separate treatment strategies. J Pediatr. 1996;128:601–607.
Nada A, Bonachea EM, Askenazi D. Acute kidney injury in the fetus and neonate. Semin Fetal Neonatal Med. 2017;22:90–97.
Selewski DT, Charlton JR, Jetton JG, Guillet R, Mhanna MJ, Askenazi DJ, et al. Neonatal acute kidney injury. Pediatrics 2015;136:e463–e473.
Hanna MH, Askenazi DJ, Selewski DT. Drug induced acute kidney injury in neonates. Curr Opin Pediatr. 2016;28:180–187.
Bateman DA, Thomas W, Parravicini E, Polesana E, Locatelli C, Lorenz JM. Serum creatinine concentration in very-low-birth-weight infants from birth to 34-36 wk postmenstrual age. Pediatr Res. 2015;77:696–702.
Mirza H, Oh W, Laptook A, Vohr B, Tucker R, Stonestreet BS. Indomethacin prophylaxis to prevent intraventricular hemorrhage: association between incidence and timing of drug administration. J Pediatr. 2013;163:706–710.e701.
Mirza H, Laptook AR, Oh W, Vohr BR, Stoll BJ, Kandefer S, et al. Effects of indomethacin prophylaxis timing on IVH and PDA in extremely low birth weight (ELBW) infants. Arch Dis Child Fetal Neonatal Ed. 2016;101:F418–F422.
Suissa S. Immortal time bias in pharmaco-epidemiology. Am J Epidemiol. 2007;167:492–499.
Acknowledgements
This work was supported by the National Institutes of Health [R01HL145032] and Nationwide Children’s Hospital.
Author information
Authors and Affiliations
Contributions
HHA and JLS designed, analyzed, interpreted, drafted, revised, and approved the final version of the manuscript. CHB, MKB, MMT, MAK, SRJ, and THM interpreted, revised, and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Abdi, H.H., Backes, C.H., Ball, M.K. et al. Prophylactic Indomethacin in extremely preterm infants: association with death or BPD and observed early serum creatinine levels. J Perinatol 41, 749–755 (2021). https://doi.org/10.1038/s41372-021-00995-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-021-00995-x
This article is cited by
-
Prophylactic indomethacin and the risk of serious pulmonary hemorrhages in preterm infants less than 28 weeks’ gestation
Journal of Perinatology (2024)
-
Effects of prophylactic indomethacin on intraventricular hemorrhage and adverse outcomes in neonatal intensive care unit
Journal of Perinatology (2022)