Abstract
Objective
To evaluate the relationship between prenatal and postnatal inflammation-related risk factors and severe retinopathy of prematurity (ROP).
Study design
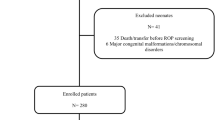
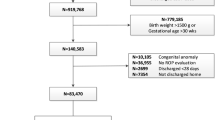
The study included infants born <30 weeks in California from 2007 to 2011. Multivariable log-binomial regression was used to assess the association between prenatal and postnatal inflammation-related exposures and severe ROP, defined as stage 3–5 or surgery for ROP.
Results
Of 14,816 infants, 10.8% developed severe ROP. Though prenatal inflammation-related risk factors were initially associated with severe ROP, after accounting for the effect of these risk factors on gestational age at birth through mediation analysis, the association was non-significant (P = 0.6). Postnatal factors associated with severe ROP included prolonged oxygen exposure, sepsis, intraventricular hemorrhage, and necrotizing enterocolitis.
Conclusion
Postnatal inflammation-related factors were associated with severe ROP more strongly than prenatal factors. The association between prenatal inflammation-related factors and ROP was explained by earlier gestational age in infants exposed to prenatal inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Horbar JD, Edwards EM, Greenberg LT, Morrow KA, Soll RF, Buus-Frank ME, et al. Variation in performance of neonatal Intensive care units in the United States. JAMA Pediatr. 2017;171:e164396.
Early Treatment for Retinopathy of Prematurity Cooperative Group, Good WV, Hardy RJ, Dobson V, Palmer EA, Phelps DL, et al. Final visual acuity results in the early treatment for retinopathy of prematurity study. Arch Ophthalmol. 2010;128:663–71.
Chen J, Smith LEH. Retinopathy of prematurity. Angiogenesis. 2007;10:133–40.
Lee J, Dammann O. Perinatal infection, inflammation, and retinopathy of prematurity. Semin Fetal Neonatal Med. 2012;17:26–9.
Hartnett ME, Penn JS. Mechanisms and management of retinopathy of prematurity. N Engl J Med. 2012;367:2515–26.
Rivera JC, Holm M, Austeng D, Morken TS, Zhou TE, Beaudry-Richard A, et al. Retinopathy of prematurity: inflammation, choroidal degeneration, and novel promising therapeutic strategies. J Neuroinflamm. 2017;14:165.
Lynch AM, Berning AA, Thevarajah TS, Wagner BD, Post MD, McCourt EA, et al. The role of the maternal and fetal inflammatory response in retinopathy of prematurity. Am J Reprod Immunol. 2018;1989:e12986.
Beaudry-Richard A, Nadeau-Vallée M, Prairie É, Maurice N, Heckel É, Nezhady M, et al. Antenatal IL-1-dependent inflammation persists postnatally and causes retinal and sub-retinal vasculopathy in progeny. Sci Rep. 2018;8:11875.
Szade A, Grochot-Przeczek A, Florczyk U, Jozkowicz A, Dulak J. Cellular and molecular mechanisms of inflammation-induced angiogenesis. IUBMB Life. 2015;67:145–59.
Yu H, Yuan L, Zou Y, Peng L, Wang Y, Li T, et al. Serum concentrations of cytokines in infants with retinopathy of prematurity. APMIS Acta Pathol Microbiol Immunol Scand. 2014;122:818–23.
Gardiner TA, Gibson DS, de Gooyer TE, de la Cruz VF, McDonald DM, Stitt AW. Inhibition of tumor necrosis factor-alpha improves physiological angiogenesis and reduces pathological neovascularization in ischemic retinopathy. Am J Pathol. 2005;166:637–44.
Hong HK, Lee HJ, Ko JH, Park JH, Park JY, Choi CW, et al. Neonatal systemic inflammation in rats alters retinal vessel development and simulates pathologic features of retinopathy of prematurity. J Neuroinflamm. 2014;11:87.
Tremblay S, Miloudi K, Chaychi S, Favret S, Binet F, Polosa A, et al. Systemic inflammation perturbs developmental retinal angiogenesis and neuroretinal function. Invest Ophthalmol Vis Sci. 2013;54:8125–39.
Thomas W, Speer CP. Chorioamnionitis: important risk factor or innocent bystander for neonatal outcome? Neonatology. 2011;99:177–87.
Combs CA, Gravett M, Garite TJ, Hickok DE, Lapidus J, Porreco R, et al. Amniotic fluid infection, inflammation, and colonization in preterm labor with intact membranes. Am J Obstet Gynecol. 2014;210:125.e1–15.
Yoon BH, Romero R, Park JS, Kim CJ, Kim SH, Choi JH, et al. Fetal exposure to an intra-amniotic inflammation and the development of cerebral palsy at the age of three years. Am J Obstet Gynecol. 2000;182:675–81.
Nelson KB, Dambrosia JM, Grether JK, Phillips TM. Neonatal cytokines and coagulation factors in children with cerebral palsy. Ann Neurol. 1998;44:665–75.
Fortes Filho JB, Costa MC, Eckert GU, Santos PGB, Silveira RC, Procianoy RS. Maternal preeclampsia protects preterm infants against severe retinopathy of prematurity. J Pediatr. 2011;158:372–6.
Hendson L, Russell L, Robertson CMT, Liang Y, Chen Y, Abdalla A, et al. Neonatal and neurodevelopmental outcomes of very low birth weight infants with histologic chorioamnionitis. J Pediatr. 2011;158:397–402.
Mitra S, Aune D, Speer CP, Saugstad OD. Chorioamnionitis as a risk factor for retinopathy of prematurity: a systematic review and meta-analysis. Neonatology. 2014;105:189–99.
Kim SJ, Port AD, Swan R, Campbell JP, Chan RVP, Chiang MF. Retinopathy of prematurity: a review of risk factors and their clinical significance. Surv Ophthalmol. 2018;63:618–37.
Slidsborg C, Jensen A, Forman JL, Rasmussen S, Bangsgaard R, Fledelius HC, et al. Neonatal risk factors for treatment-Demandingretinopathy of prematurity: a Danish National Study. Ophthalmology. 2016;123:796–803.
Hutchinson AK, Melia M, Yang MB, VanderVeen DK, Wilson LB, Lambert SR. Clinical models and algorithms for the prediction of retinopathy of prematurity: a report by the American Academy of Ophthalmology. Ophthalmology. 2016;123:804–16.
California Perinatal Quality Care Collaborative |. Available at: https://www.cpqcc.org/.
Vermont Oxford Network – Improving care for infants and their families. Available at: https://public.vtoxford.org/.
Fierson WM, American Academy of Pediatric Section on Ophthalmology, American Academy of Ophthalmology, American Association for Pediatric Ophthalmology and Strabismus, American Association of Certified Orthoptists. Screening Examination of Premature Infants for Retinopathy of Prematurity. Pediatrics. 2013; 131:189–95.
Segovia SA, Vickers MH, Reynolds CM. The impact of maternal obesity on inflammatory processes and consequences for later offspring health outcomes. J Dev Orig Health Dis. 2017;8:529–40.
Kim SM, Romero R, Park JW, Oh KJ, Jun JK, Yoon BH. The relationship between the intensity of intra-amniotic inflammation and the presence and severity of acute histologic chorioamnionitis in preterm gestation. J Matern Fetal Neonatal Med. 2015;28:1500–9.
Boisen AB, Knorr S, Hansen TK, Vlachova Z, Bytoft B, Damm P, et al. Signs of low-grade systemic inflammation in female offspring of women with type 1 diabetes: the EPICOM study. Diabetes Metab. 2016;42:462–5.
Sharma K, Singh R, Kumar M, Gupta U, Rohil V, Bhattacharjee J. First-trimester inflammatory markers for risk evaluation of pregnancy hypertension. J Obstet Gynaecol India. 2018;68:27–32.
Kalagiri RR, Carder T, Choudhury S, Vora N, Ballard AR, Govande V, et al. Inflammation in complicated pregnancy and its outcome. Am J Perinatol. 2016;33:1337–56.
Musilova I, Andrys C, Drahosova M, Soucek O, Stepan M, Bestvina T, et al. Intraamniotic inflammation and umbilical cord blood interleukin-6 concentrations in pregnancies complicated by preterm prelabor rupture of membranes. J Matern Fetal Neonatal Med. 2017;30:900–10.
Cardenas I, Means RE, Aldo P, Koga K, Lang SM, Booth CJ, et al. Viral infection of the placenta leads to fetal inflammation and sensitization to bacterial products predisposing to preterm labor. J Immunol. 2010;185:1248–57.
WHO:: Global Database on Body Mass Index. Available at: http://apps.who.int/bmi/index.jsp?introPage=intro_3.html.
Kennedy KA, Wrage LA, Higgins RD, Finer NN, Carlo WA, Walsh MC, et al. Evaluating retinopathy of prematurity screening guidelines for 24- to 27-week gestational age infants. J Perinatol. 2014;34:311–8.
Al-Abdi SY, Al-Aamri MA. A systematic review and meta-analysis of the timing of early intraventricular hemorrhage in preterm neonates: clinical and research implications. J Clin Neonatol. 2014;3:76–88.
Yee WH, Soraisham AS, Shah VS, Aziz K, Yoon W, Lee SK, et al. Incidence and timing of presentation of necrotizing enterocolitis in preterm infants. Pediatrics. 2012;129:e298–304.
Lange T, Vansteelandt S, Bekaert M. A simple unified approach for estimating natural direct and indirect effects. Am J Epidemiol. 2012;176:190–5.
Vanderweele TJ. Explanation in causal inference: methods for mediation and interaction. New York, NY, US: Oxford University Press; 2015.
R: The R Project for Statistical Computing. Available at: https://www.r-project.org/.
Villamor-Martinez E, Cavallaro G, Raffaeli G, Mohammed Rahim OMM, Gulden S, Ghazi AMT, et al. Chorioamnionitis as a risk factor for retinopathy of prematurity: An updated systematic review and meta-analysis. PLoS ONE. 2018;13:e0205838.
Wilcox AJ, Weinberg CR, Basso O. On the pitfalls of adjusting for gestational age at birth. Am J Epidemiol. 2011;174:1062–8.
York JR, Landers S, Kirby RS, Arbogast PG, Penn JS. Arterial oxygen fluctuation and retinopathy of prematurity in very-low-birth-weight infants. J Perinatol. 2004;24:82–7.
Wu C, Löfqvist C, Smith LEH, VanderVeen DK, Hellström A.WINROP Consortium Importance of early postnatal weight gain for normal retinal angiogenesis in very preterm infants: a multicenter study analyzing weight velocity deviations for the prediction of retinopathy of prematurity. Arch Ophthalmol. 2012;130:992–9.
Acknowledgements
G.P.G. is supported in part by the Stanford Maternal & Child Health Research Institute. S.A.L. is supported in part by postdoctoral fellowship F32HD091945 from the Eunice Kennedy Shriver National Institute of Child Health and Development and a postdoctoral fellowship from the Stanford Maternal & Child Health Research Institute.
Author information
Authors and Affiliations
Contributions
All authors contributed substantially to the study and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Goldstein, G.P., Leonard, S.A., Kan, P. et al. Prenatal and postnatal inflammation-related risk factors for retinopathy of prematurity. J Perinatol 39, 964–973 (2019). https://doi.org/10.1038/s41372-019-0357-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-019-0357-2
This article is cited by
-
Retinopathy of prematurity screening: prevalence and risk factors of ophthalmic complications in non-treated preterm infants
Eye (2024)
-
Dysregulation of histone deacetylases in ocular diseases
Archives of Pharmacal Research (2024)
-
Risk factors for bronchopulmonary dysplasia infants with respiratory score greater than four: a multi-center, prospective, longitudinal cohort study in China
Scientific Reports (2023)
-
Association of inflammatory and angiogenic biomarkers in maternal plasma with retinopathy of prematurity in preterm infants
Eye (2023)
-
Identification of clinical factors associated with timing and duration of spontaneous regression of retinopathy of prematurity not requiring treatment
Journal of Perinatology (2023)