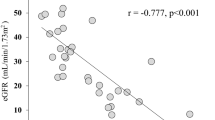
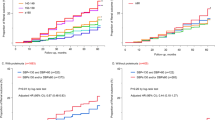
Abstract
Treatment resistant hypertension (TRH) appears of particular relevance in patients with chronic kidney disease (CKD). However, causes and consequences of TRH in CKD patients remain incompletely understood. Therefore, we analyzed the prevalence of apparent TRH (aTRH), and phenotypic characteristics and prognosis associated with aTRH among participants of the German Chronic Kidney Disease (GCKD) study. As insufficient medication adherence has been shown to be a frequent cause of pseudoresistance, we also assessed treatment adherence. Study participants were classified as having aTRH, controlled hypertension and uncontrolled hypertension based on study visit blood pressure and self-reported medication intake. Drug adherence was assessed by comparing self-reported antihypertensive medication with detectable urinary drug metabolites measured by mass spectroscopy. Out of 4901 individuals included in this study, 38% were classified as having aTRH. Male sex, older age, lower estimated glomerular filtration rate (eGFR), higher body mass index (BMI), higher urine albumin-to-creatinine ratio (UACR) and presence of diabetes mellitus were independently associated with higher prevalence of aTRH in a multivariable adjusted regression model. Patients classified as aTRH had higher risk for major adverse cardiovascular events and worsening of kidney disease compared to patients with no aTRH after multivariate adjustment for potential confounders. There was a high agreement between self-reported medication and detectable urinary drug metabolites. In conclusion, in a cohort of Caucasian patients with moderately severe CKD, aTRH was highly prevalent and, in most cases, likely not caused by low medication adherence. Furthermore, aTRH was linked to cardio-renal endpoints, emphasizing the need for improved management.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
GCKD data is not publicly available. The code for this analysis is available from the corresponding author on reasonable request.
References
Bikbov B, Purcell CA, Levey AS, Smith M, Abdoli A, Abebe M, et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2020;395:709–33.
Rossignol P, Massy ZA, Azizi M, Bakris G, Ritz E, Covic A, et al. The double challenge of resistant hypertension and chronic kidney disease. Lancet. 2015;386:1588–98.
Tanner RM, Calhoun DA, Bell EK, Bowling CB, Gutiérrez OM, Irvin MR, et al. Prevalence of Apparent Treatment-Resistant Hypertension among Individuals with CKD. Clin J Am Soc Nephrol. 2013;8:1583–90.
Thomas G, Xie D, Chen HY, Anderson AH, Appel LJ, Bodana S, et al. Prevalence and prognostic significance of apparent treatment resistant hypertension in chronic kidney disease: report from the chronic renal insufficiency cohort study. Hypertension 2016;67:387–96.
Unger T, Borghi C, Charchar F, Khan NA, Poulter NR, Prabhakaran D, et al. 2020 International Society of Hypertension Global Hypertension Practice Guidelines. Hypertension. 2020;75:1334–57.
Calhoun DA, Jones D, Textor S, Goff DC, Murphy TP, Toto RD, et al. Resistant hypertension: diagnosis, evaluation, and treatment: a scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Circulation 2008;117:e510–26.
Carey RM, Calhoun DA, Bakris GL, Brook RD, Daugherty SL, Dennison-Himmelfarb CR, et al. Resistant hypertension: detection, evaluation, and management: a scientific statement from the American heart association. Hypertension 2018;72:e53–e90.
Carey RM, Sakhuja S, Calhoun DA, Whelton PK, Muntner P. Prevalence of apparent treatment-resistant hypertension in the United States. Hypertension 2019;73:424–31.
de Beus E, Bots ML, van Zuilen AD, Wetzels JF, Blankestijn PJ, Group MS. Prevalence of apparent therapy-resistant hypertension and its effect on outcome in patients with chronic kidney disease. Hypertension 2015;66:998–1005.
Hyman DJ, Pavlik V. Medication adherence and resistant hypertension. J Hum Hypertension. 2015;29:213–8.
Schneider MP, Hilgers KF, Schmid M, Hubner S, Nadal J, Seitz D, et al. Blood pressure control in chronic kidney disease: a cross-sectional analysis from the German Chronic Kidney Disease (GCKD) study. PloS one. 2018;13:e0202604.
De Nicola L, Gabbai FB, Agarwal R, Chiodini P, Borrelli S, Bellizzi V, et al. Prevalence and prognostic role of resistant hypertension in chronic kidney disease patients. J Am Coll Cardiol. 2013;61:2461–7.
Eckardt KU, Bärthlein B, Baid-Agrawal S, Beck A, Busch M, Eitner F, et al. The German Chronic Kidney Disease (GCKD) study: design and methods. Nephrol, Dialysis, Transplant: Off Publ Eur Dialysis Transpl Assoc - Eur Ren Assoc. 2012;27:1454–60.
Titze S, Schmid M, Köttgen A, Busch M, Floege J, Wanner C, et al. Disease burden and risk profile in referred patients with moderate chronic kidney disease: composition of the German Chronic Kidney Disease (GCKD) cohort. Nephrol, Dialysis, Transplant: Off Publ Eur Dialysis Transpl Assoc - Eur Ren Assoc. 2015;30:441–51.
Evans AM, Bridgewater BR, Liu Q, Mitchell MW, Robinson RJ, Dai H, et al. High resolution mass spectrometry improves data quantity and quality as compared to unit mass resolution mass spectrometry in high-throughput profiling metabolomics. Metabolomics 2014;04:132.
Schlosser P, Li Y, Sekula P, Raffler J, Grundner-Culemann F, Pietzner M, et al. Genetic studies of urinary metabolites illuminate mechanisms of detoxification and excretion in humans. Nat Genet. 2020;52:167–76.
Kotsis F, Schultheiss U, Wuttke M, Schlosser P, Mielke J, Becker MS, et al. Self-reported medication use and urinary drug metabolites in the German chronic kidney disease (GCKD) study. J of the Am Soc of Nephrol. 2021;32:2315–29.
Dieterle F, Ross A, Schlotterbeck G, Senn H. Probabilistic quotient normalization as robust method to account for dilution of complex biological mixtures. application in1H NMR metabonomics. Anal Chem. 2006;78:4281–90.
Gray RJ. A class of K-sample tests for comparing the cumulative incidence of a competing risk. The Ann Stat. 1988;16:1141–54.
Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509.
Schoenfeld D. Partial residuals for the proportional hazards regression model. Biometrika 1982;69:239–41.
McHugh ML. Interrater reliability: the kappa statistic. Biochem Med (Zagreb). 2012;22:276–82.
R Core Team. R: A language and environment for statistical computing. Vienna. Austria: R foundation for statistical computing.; 2021. https://www.R-project.org/
Thernau T. A package for survival analysis in R. R package version 3.1-12 2020. https://CRAN.R-project.org/package=survival.
Kolde R. pheatmap: Pretty Heatmaps. R package version 1.0.12. 2019. https://CRAN.R-project.org/package=pheatmap.
Wickham H. tidyverse: Easily Install and Load the “Tidyverse”. R package version 1.2.1. 2017. https://CRAN.R-project.org/package=tidyverse.
Kassambara A, Kosinski M, Biecek P survminer: Drawing Survival Curves using “ggplot2”. R package version 0.4.6. 2019. https://CRAN.R-project.org/package=survminer.
Gray B cmprsk: Subdistribution Analysis of Competing Risks. R package version 2.2.-10. 2020. https://CRAN.R-project.org/package=cmprsk.
Revelle W. psych: Procedures for Personality and Psychological Research. Evanston, Illinois, USA: Northwerstern University; 2019. https://CRAN.R-project.org/package=psych
Hothorn T, Bretz F, Westfall P. Simultaneous inference in general parametric models. Biometrical J. 2008;50:346–63.
Viazzi F, Greco E, Ceriello A, Fioretto P, Giorda C, Guida P, et al. Apparent treatment resistant hypertension, blood pressure control and the progression of chronic kidney disease in patients with Type 2 diabetes. Kidney blood Press Res. 2018;43:422–38.
Daugherty SL, Powers JD, Magid DJ, Tavel HM, Masoudi FA, Margolis KL, et al. Incidence and prognosis of resistant hypertension in hypertensive patients. Circulation 2012;125:1635–42.
Akinyelure OP, Sakhuja S, Colvin CL, Clark D 3rd, Jaeger BC, Hardy ST, et al. Cardiovascular health and transition from controlled blood pressure to apparent treatment resistant hypertension: The Jackson Heart Study and the REGARDS Study. Hypertension 2020;76:1953–61.
Sim JJ, Bhandari SK, Shi J, Reynolds K, Calhoun DA, Kalantar-Zadeh K, et al. Comparative risk of renal, cardiovascular, and mortality outcomes in controlled, uncontrolled resistant, and nonresistant hypertension. Kidney Int. 2015;88:622–32.
van der Sande NGC, de Beus E, Bots ML, Voskuil M, Blankestijn PJ, Visseren F, et al. Apparent resistant hypertension and the risk of vascular events and mortality in patients with manifest vascular disease. J Hypertens. 2018;36:143–50.
Smith SM, Gong Y, Handberg E, Messerli FH, Bakris GL, Ahmed A, et al. Predictors and outcomes of resistant hypertension among patients with coronary artery disease and hypertension. J Hypertens. 2014;32:635–43.
Williams B, MacDonald TM, Morant S, Webb DJ, Sever P, McInnes G, et al. Spironolactone versus placebo, bisoprolol, and doxazosin to determine the optimal treatment for drug-resistant hypertension (PATHWAY-2): a randomised, double-blind, crossover trial. Lancet. 2015;386:2059–68.
Maiolino G, Azzolini M, Rossi GP. Mineralocorticoid Receptor Antagonists Therapy in Resistant Hypertension: Time to Implement Guidelines! Front cardiovascular Med. 2015;2:3.
Jung O, Gechter JL, Wunder C, Paulke A, Bartel C, Geiger H, et al. Resistant hypertension? Assessment of adherence by toxicological urine analysis. J Hypertens. 2013;31:766–74.
Schmieder RE, Ott C, Schmid A, Friedrich S, Kistner I, Ditting T, et al. Adherence to Antihypertensive Medication in Treatment-Resistant Hypertension Undergoing Renal Denervation. J Am Heart Assoc. 2016;5:e002343.
Tomaszewski M, White C, Patel P, Masca N, Damani R, Hepworth J, et al. High rates of non-adherence to antihypertensive treatment revealed by high-performance liquid chromatography-tandem mass spectrometry (HP LC-MS/MS) urine analysis. Heart 2014;100:855–61.
Scheppach JB, Raff U, Toncar S, Ritter C, Klink T, Stork S, et al. Blood Pressure Pattern and Target Organ Damage in Patients With Chronic Kidney Disease. Hypertension 2018;72:929–36.
Acknowledgements
We are grateful for the willingness of the patients to participate in the GCKD study. The enormous effort of the study personnel of the various regional centers is highly appreciated. We thank the large number of nephrologists who provide routine care for the patients and collaborate with the GCKD study. The work of AK was supported by grant 3598/5-1 from the German Research Foundation (DFG). FK was supported by the Else Kroener Fresenius Foundation (NAKSYS) and by the German Federal Ministry of Education and Research (BMBF) within the framework of the e:Med research and funding concept (grant 01ZX1912B). The GCKD study was/is funded by grants from the Federal Ministry of Education and Research (BMBF, grant number 01ER0804) and the KfH Foundation for Preventive Medicine and corporate sponsors.
Author information
Authors and Affiliations
Consortia
Contributions
Design of this study: JM, MSB. Recruitment, management and conduct of cohort study: FK, AK, MPS, KUE. Bioinformatics and statistical analysis: JM, CS. Wrote the manuscript: JM, MSB, HT-J. Edited, critically read and approved the manuscript: JM, HT-J, CS, FK, AK, MPS, KUE, DFF, FE, MSB.
Corresponding author
Ethics declarations
Competing interests
JM, HT-J, CS, DFF, FE, MSB are employees from Bayer AG. For the remaining authors no conflicts of interest were declared.
Ethical approval
The study was approved by the local ethics committee, registered in the German national registry for clinical studies (DRKS00003971) and was conducted according to the principles of the Declaration of Helsinki and Good Clinical Practice. Informed consent was obtained from all patients prior to enrollment.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Mielke, J., Trucks-Jansen, H., Schurmann, C. et al. Prevalence, phenotypic characteristics and prognostic role of apparent treatment resistant hypertension in the German Chronic Kidney Disease (GCKD) study. J Hum Hypertens 37, 345–353 (2023). https://doi.org/10.1038/s41371-022-00701-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41371-022-00701-0