Abstract
Background/Objective
There are limited real-world studies assessing semaglutide weight loss and associated comorbidity and metabolic outcomes over periods ≥ 6 months. We aim to assess weight loss, metabolic, and cardiovascular outcomes of 12 months of semaglutide.
Subject/Methods
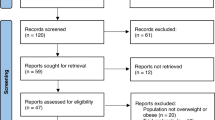
We conducted a multicentered retrospective cohort study on semaglutide use. We included patients with a body-mass index (BMI) ≥ 27 kg/m2 who were prescribed weekly semaglutide subcutaneous injections. We excluded patients with bariatric surgeries, taking other anti-obesity medications, and with active malignancy or pregnancy. A total of 1023 patients had semaglutide prescription for obesity.
Intervention/Methods
We assessed weight loss outcomes of subcutaneous semaglutide for 12 months. The primary endpoint was total body weight loss percentage (TBWL%) at 12 months. Secondary endpoints included proportion of patients achieving ≥5%, ≥10%, ≥15%, and ≥20% weight loss, and improvements in metabolic, cardiovascular, and comorbidities after 12 months of follow-up.
Results
We included 304 patients (73% female, 93% White, mean age 48.8 [12.4] years, BMI 40.9 [9.6] kg/m2) in the analysis. Patients achieved a TBWL of 13.4 (8.0)% at 12 months (p < 0.001 from baseline). Patients without T2DM achieved a TBWL of 16.9 (6.9)% compared to 9.9 (8.4)% in patients without T2DM at 12 months on the higher doses of semaglutide (p < 0.001 from baseline). In this cohort, 81% achieved ≥5%, 64% achieved ≥10%, 41% achieved ≥15%, and 22% achieved ≥20% TBWL at 12 months. Patients with overweight or obesity experienced significant improvements in metabolic, lipid profile, blood pressure, liver function tests, and cardiovascular disease risk outcomes.
Conclusions
Semaglutide demonstrated notable improvement in obesity, metabolic, and cardiovascular disease risk outcomes in a clinical setting.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
Deidentified individual participant data can be shared upon request: Data can be sent to journal if needed. No limited timeframe.
References
Sørensen TIA, Martinez AR, Jørgensen TSH. Epidemiology of Obesity. Handb Exp Pharmacol. 2022;274:3–27.
Abdelaal M, le Roux CW, Docherty NG. Morbidity and mortality associated with obesity. Ann Transl Med. 2017;5:161.
Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384:989–1002.
Gadde KM, Allison DB, Ryan DH, Peterson CA, Troupin B, Schwiers ML, et al. Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER): a randomised, placebo-controlled, phase 3 trial. Lancet. 2011;377:1341–52.
Tchang BG, Aras M, Kumar RB, Aronne LJ. Pharmacologic treatment of overweight and obesity in adults. In: Feingold KR, Anawalt B, Blackman MR, et al., editors. Endotext. South Dartmouth (MA): MDText.com, Inc.; 2000. Available from: https://www.ncbi.nlm.nih.gov/books/NBK279038/. 2015.
Ghusn W, Hurtado MD, Acosta A. Weight-centric treatment of type 2 diabetes mellitus. Obes Pillars. 2022;4:100045.
Ghusn W, De la Rosa A, Sacoto D, Cifuentes L, Campos A, Feris F, et al. Weight Loss Outcomes Associated With Semaglutide Treatment for Patients With Overweight or Obesity. JAMA Network Open. 2022;5:e2231982.
Rubino DM, Greenway FL, Khalid U, O’Neil PM, Rosenstock J, Sørrig R, et al. Effect of Weekly Subcutaneous Semaglutide vs Daily Liraglutide on Body Weight in Adults With Overweight or Obesity Without Diabetes: The STEP 8 Randomized Clinical Trial. JAMA. 2022;327:138–50.
Davies M, Færch L, Jeppesen OK, Pakseresht A, Pedersen SD, Perreault L, et al. Semaglutide 2·4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): a randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial. Lancet. 2021;397:971–84.
Calderon G, Gonzalez-Izundegui D, Shan KL, Garcia-Valencia OA, Cifuentes L, Campos A, et al. Effectiveness of anti-obesity medications approved for long-term use in a multidisciplinary weight management program: a multi-center clinical experience. Int J Obes. 2022;46:555–63.
De la Rosa A, Ghusn W, Sacoto D, Campos A, Cifuentes L, Feris F, et al. A comparison between weight loss outcomes with anti-obesity medications before and during Covid-19 pandemic at a tertiary weight management center. Obes Pillars. 2022;4:100046.
Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jódar E, Leiter LA, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N Engl J Med. 2016;375:1834–44.
Orsi E, Solini A, Bonora E, Vitale M, Garofolo M, Fondelli C, et al. Risk of all-cause mortality according to the European Society of Cardiology risk categories in individuals with type 2 diabetes: the Renal Insufficiency And Cardiovascular Events (RIACE) Italian Multicenter Study. Acta Diabetol. 2022;59:1369–81.
Söderberg C, Stål P, Askling J, Glaumann H, Lindberg G, Marmur J, et al. Decreased survival of subjects with elevated liver function tests during a 28-year follow-up. Hepatology. 2010;51:595–602.
Ghusn W, Bouchard C, Frye MA, Acosta A. Weight-centric treatment of depression and chronic pain. Obes Pillars. 2022;3:100025.
Wilding JPH, Batterham RL, Davies M, Van Gaal LF, Kandler K, Konakli K, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes Obes Metabol. 2022;24:1553–64.
Funding
AA is supported by NIH (K23-DK114460), and is a stockholder in Gila Therapeutics, Phenomix Sciences; he served as a consultant for Rhythm Pharmaceuticals, General Mills, Currax, Nestle, Amgen, Structure and Boehringer Ingelheim.
Author information
Authors and Affiliations
Contributions
All authors had full access to all the data and statistical analyses. AA had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. WG wrote the first draft of the manuscript. WG, SF, DA, ET, BN, KG, AR, DS, LC, AC, FF, and MH were responsible of reviewing the manuscript. WG performed the analysis. AA and DH supervised the manuscript writing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghusn, W., Fansa, S., Anazco, D. et al. Weight loss and cardiovascular disease risk outcomes of semaglutide: a one-year multicentered study. Int J Obes 48, 662–667 (2024). https://doi.org/10.1038/s41366-023-01456-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-023-01456-5