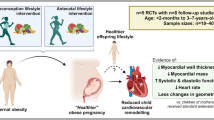
Abstract
Background/Objectives
Obesity in pregnancy has been associated with increased childhood cardiometabolic risk and reduced life expectancy. The UK UPBEAT multicentre randomised control trial was a lifestyle intervention of diet and physical activity in pregnant women with obesity. We hypothesised that the 3-year-old children of women with obesity would have heightened cardiovascular risk compared to children of normal BMI women, and that the UPBEAT intervention would mitigate this risk.
Subjects/Methods
Children were recruited from one UPBEAT trial centre. Cardiovascular measures included blood pressure, echocardiographic assessment of cardiac function and dimensions, carotid intima-media thickness and heart rate variability (HRV) by electrocardiogram.
Results
Compared to offspring of normal BMI women (n = 51), children of women with obesity from the trial standard care arm (n = 39) had evidence of cardiac remodelling including increased interventricular septum (IVS; mean difference 0.04 cm; 95% CI: 0.018 to 0.067), posterior wall (PW; 0.03 cm; 0.006 to 0.062) and relative wall thicknesses (RWT; 0.03 cm; 0.01 to 0.05) following adjustment. Randomisation of women with obesity to the intervention arm (n = 31) prevented this cardiac remodelling (intervention effect; mean difference IVS −0.03 cm (−0.05 to −0.008); PW −0.03 cm (−0.05 to −0.01); RWT −0.02 cm (−0.04 to −0.005)). Children of women with obesity (standard care arm) compared to women of normal BMI also had elevated minimum heart rate (7 bpm; 1.41 to 13.34) evidence of early diastolic dysfunction (e prime) and increased sympathetic nerve activity index by HRV analysis.
Conclusions
Maternal obesity was associated with left ventricular concentric remodelling in 3-year-old offspring. Absence of remodelling following the maternal intervention infers in utero origins of cardiac remodelling.
Clinical trial registry name and registration number
The UPBEAT trial is registered with Current Controlled Trials, ISRCTN89971375.
Similar content being viewed by others
Introduction
Obesity amongst pregnant women has risen in parallel with population trends. In the UK more than half the women attending antenatal care are overweight or obese (BMI ≥25 kg/m2) [1]. Obesity increases the risk of perinatal and maternal morbidity and mortality, and is now the most prevalent risk factor for adverse pregnancy outcome [2], contributing to substantial healthcare costs [3]. Reports of an independent relationship between maternal BMI and body fat mass in their children have raised concern that maternal obesity may contribute directly to the global increase of obesity in childhood [4], prompting the suggestion that preventive strategies for reducing childhood obesity should include interventions targeting maternal BMI [5]. Cardiovascular risk in progeny of mothers with obesity, although less frequently investigated, includes a recent population-based cohort study demonstrating a greater risk of cardiovascular disease amongst 1–25-year-old offspring, which increased incrementally with the severity of maternal obesity, and another reporting an association between maternal obesity and all-cause mortality in adult offspring [6, 7]. Relationships between maternal BMI and childhood blood pressure have also been reported [8]. Using rodent models of maternal obesity, we and others, have demonstrated cardiac hypertrophy, hypertension and altered sympathetic activity in young offspring [9,10,11,12], implicating a direct effect of intrauterine exposures on cardiovascular development. In women and children, these associations could have other origins, including shared genetic traits and family environment [4]. Since randomised controlled trials (RCT) in pregnant women with obesity may offer insight into in utero versus familial origins of cardiovascular risk, this study evaluated the effect of a complex lifestyle intervention of diet and physical activity in obese pregnancy (the UPBEAT intervention) on childhood cardiovascular function.
The UK Pregnancies Better Eating and Activity Trial (UPBEAT) was a complex lifestyle intervention in 1555 pregnant women with obesity [13]. Women were randomised to an intense 8-week behavioural intervention or to standard antenatal care. The intervention had no effect on the primary outcomes, incidence of gestational diabetes and large for gestational age infants. However, there were improvements in several secondary maternal outcomes including; a reduction in total GWG and lower sum of skinfold thicknesses; an improvement in maternal antenatal glycaemic load and saturated fat intake; a modest increase in self-reported physical activity; a healthier metabolic profile across pregnancy [14]; as well as sustained improvements in maternal diet and lower infant adiposity (subscapular skinfold thicknesses) at 6 months postpartum [15]. The present study aimed to assess, in a subgroup of the original cohort, whether the UPBEAT intervention led to improved childhood cardiovascular function at 3 years.
The primary aims of this study were to compare detailed transthoracic echocardiographic measures in 3-year-old children from pregnant women with obesity with those from pregnant women of normal BMI (20–25 kg/m2). We hypothesised that children of women with obesity would have evidence of cardiovascular dysfunction compared to children of women with normal BMI, and that the UPBEAT intervention would mitigate this risk. Cardiovascular outcomes prioritised as risk factors for future cardiovascular disease included blood pressure, echocardiographic assessment of cardiac function (e.g., diastolic function) and dimensions (e.g., left ventricular mass (LVM)), and carotid intima-media thickness (CIMT) as a measure of vascular remodelling. Heart rate variability (HRV) by electrocardiogram (ECG) was also assessed as a marker of autonomic dysfunction, which we hypothesised may drive cardiovascular remodelling [16]. If persistent, these changes may contribute to adulthood cardiovascular disease.
Methods
The UPBEAT trial was a multicentre RCT conducted on 1555 pregnant women with a median BMI of 35.1 kg/m2 (IQR 32.8–38.6) [13]. Maternal demographic details were obtained at trial entry. Women with obesity (≥16 years of age; pre-pregnancy BMI ≥30 kg/m2), were recruited in early pregnancy, exclusion criteria included pre-existing disease and multiple pregnancies. Women were randomised, at 15+0 and 18+6 weeks’ gestation, to an intensive 8-week behavioural intervention or to standard antenatal care, as previously reported [13]. In brief, the intervention comprised dietary recommendation to reduce maternal antenatal glycaemic load and saturated fat intake and to increase physical activity over an 8-week period. The intervention, delivered by health trainers, led to a healthier maternal metabolic profile, improved diet and lower weight gain in pregnancy and reduced infant adiposity at 6 months.
Subjects
Mothers and their 3-year-old children attending the UPBEAT RCT follow-up visit at Guy’s and St. Thomas’ NHS Foundation Trust (GSTFT), London, UK, were informed about the additional nested case–control cardiovascular study and invited to return within 2 weeks, when the mother provided consent. The children were studied in the NIHR Clinical Research Facility at St. Thomas’ Hospital. Thirty-nine children were recruited from the standard care arm and 31 from the intervention arm from 228 mother–child pairs attending the 3-year-UPBEAT follow-up visit (Supplementary Fig. 1, Consort diagram).
Comparisons were also made between children from the standard care arm of the UPBEAT cohort, and 51 children of normal BMI mothers (20–25 kg/m2) who gave birth 3–4 years before the study date (i.e., contemporaneously with the UPBEAT cohort) who were identified from the hospital maternity database, GSTFT. The normal BMI women were matched for age and parity with women recruited to the UPBEAT standard care arm and recruited by letter.
Consent and ethical approval
Informed consent was obtained from all subjects. Consent in the UPBEAT trial included an agreement for further contact (UK integrated research application system, reference 09/H0802/5). The follow-up study design and protocol were approved by the NHS Research Ethics Committee (UK Integrated Research Application System; reference 13/LO/1108). Research midwives and research assistants completed data collection between October 2014 and August 2018.
Childhood outcomes
Echocardiography
A transthoracic echocardiographic study and the left common carotid artery image were obtained using the Philips Epiq ultrasound system (Philips Healthcare, Andover, USA) and analysed by one author (HG) blinded to the study group. All echocardiographic views and measurements were performed using standard techniques according to the American Society of Echocardiography [17,18,19] (see Supplementary Material for details).
Pulse wave velocity
Aortic pulse wave velocity (aPWV) was calculated using the time difference between R-wave to onset of pulsed wave Doppler at descending aorta (point of left subclavian artery) and abdominal aorta (point of diaphragm) using the length of the sternum between jugular notch and xiphoid process as a measure of path length.
Carotid intimal-medial thickness (IMT)
IMT was measured using the Philips QLAB vascular automated analysis package (Philips Healthcare, Andover, USA) focusing on the posterior wall over the left common carotid artery.
Blood pressure and heart rate variability
Blood pressure measurements were taken following 5 min of rest in a supine position, using the Welch Allyn 53S00-E4 device, with an appropriately sized arm cuff for the child. An average of three blood pressure readings were taken, and the mean, systolic and diastolic values were recorded. Blood pressure was subsequently converted to percentiles (adjusted for age, sex and height) using the Fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents [20, 21].
Following a further 5 min rest, the ECG was recorded over 20–30 min with the child in the supine position (ECG, lead II electrocardiogram, Medilog AR12 Plus). ECG traces were uploaded to the study database (MedSciNet) and analysed with Medilog Darwin 2 (ver. 2.7.1, SMART Medical, Gloucestershire, UK) and Kubios HRV Premium (ver. 3.3.1, MathWorks Inc. Massachusetts, United States) software [22, 23]. HRV was analysed from at least three 5-min segments of the recording obtained. Recordings were visually inspected, and non-sinusoidal beats and artefacts were excluded from analysis (see Supplementary Material).
Statistics
Maternal and offspring characteristics in the form of numerical and categorical data were compared by t-test and χ2 test, respectively. Using unadjusted and adjusted regression analyses, comparisons were made between normal BMI controls (A) and UPBEAT standard care arms (C), and between UPBEAT intervention (B) and standard care (C) arms. Mean values with standard deviations were calculated for numerical data where appropriate. For the adjusted models, confounders included maternal ethnicity, maternal smoking status at UPBEAT trial baseline, age of the child at follow-up (months), sex and child BMI z-score. Analysis was by intention-to-treat. Analyses were performed using Stata version 15.0 (StataCorp, College Station, TX, USA) and IBM SPSS Statistics, Version 26.0. P ≤ 0.05 was considered significant (see Supplementary Material for sample size calculations).
Owing to the exploratory nature of this study, i.e., to examine childhood cardiovascular parameters potentially modifiable by the UPBEAT intervention in a subgroup of RCT participants, we did not control for multiple testing [24, 25]. This accords with reports of RCTs of exploratory and hypothesis-generating nature, which have adopted similar practice [26, 27].
Results
Subject characteristics
There were no differences in demographic or baseline variables between intervention and standard care arms in the subgroup of 70 women in the present study, as in the main UPBEAT trial (Table 1). Similarly, there was no difference in maternal fasting plasma glucose at 28 weeks gestation between intervention and standard care arms (Table 1). Although lower in the intervention arm, there was no statistical difference in the incidence of GDM between the intervention (23%) and standard care (30%) arms. Women in the normal BMI group, recruited contemporaneously from the general antenatal population were studied in parallel with the UPBEAT participants. UPBEAT participants were less likely to be white and to have a history of smoking than the normal BMI group, and had lower educational attainment, as assessed by years in education (Table 1).
Comparing women in the UPBEAT subgroup to those in the original UPBEAT trial cohort [13, 14], maternal age was slightly higher by approximately 1 year, and ethnicity differed at baseline with more black and less Asian women represented at the London centre (Supplementary Table 1).
There was no difference in the sex ratio, birthweight, and gestational age at birth of offspring between the three study groups. All infants, except one, were born at term (>37 weeks gestation). The children in the normal maternal BMI group were slightly older at 3-year follow-up, by approximately 1 month, than the children in the UPBEAT standard care arm (P < 0.001) (Table 2).
Obesity in pregnancy and child echocardiographic parameters
Compared to children of women with a normal BMI, children in the UPBEAT standard care arm demonstrated increased interventricular septal thickness (IVS) thickness after adjustment for maternal ethnicity and smoking status at baseline, child age, gender and BMI z-score (IVS, mean diff (CI): 0.04 cm; 0.018 to 0.067); they also demonstrated increased posterior wall thickness (PWT, 0.03 cm (0.006 to 0.062) and relative wall thickness after adjustment (RWT 0.03 cm; 0.01 to 0.054). Evidence of LV concentric remodelling included higher LVM index (LVMi g/m2; 3.96; 1.36 to 6.57), and an increase in the ratio of LVM to end-diastolic volume (LVM/EDV; 0.95 g/ml; 0.01 to 0.17, Table 3 and Fig. 1).
Echocardiographic parameters in children showing A maternal obesity effect and B intervention effect. Difference in standard deviation (SD), plotted with 95% confidence intervals are unadjusted (dash line) and adjusted (solid line). A Mean differences associated with the UPBEAT standard care arm (n = 39) versus the normal BMI group [51] are shown on the left and B mean differences associated with the UPBEAT intervention arm (n = 31) versus the UPBEAT standard care arm (n = 39) are shown on the right.
Functional measures suggested early indications of diastolic dysfunction in children in the UPBEAT standard care arm compared to children of women with a normal BMI, as evidenced by a preserved and lower E/A ratio (E/A, −0.23 (−0.42 to −0.05)) that remained significant after adjustment (E/A, −0.24 (−0.47 to −0.0005). A reduction in e prime, also significant after adjustment (e’, −0.08 m/s; −0.14 to −0.02) suggested slower early diastolic filling (Table 3).
UPBEAT intervention in pregnancy prevents LV concentric remodelling
Compared to children of women in the UPBEAT standard care arm, those in the intervention arm did not show evidence of concentric remodelling; rather there was a significant reduction in IVS thickness (−0.02 cm; −0.04 to −0.002), a reduction in PW thickness (−0.02 cm; −0.05 to −0.005), RWT (−0.02; −0.04 to −0.005), and in the ratio of LVM to EDV (−0.11 g/ml; −0.20 to −0.014), all of which remained significant after adjustment (Fig. 1 and Table 3).
Pulse wave velocity and carotid intimal-medial thickness (CIMT)
Across all groups, maternal BMI was not related to offspring PWV or CIMT and there was no apparent effect of the intervention (Table 3).
Heart rate and autonomic function
Children in the UPBEAT standard care arm had significantly increased minimum heart rate (min HR; 7 bpm; 1.4 to 13.3), maximum HR (8 bpm; 0.94 to 15.1) and mean HR (6 bpm; 1.0 to 12.5) compared to children of normal BMI mothers, after adjustment for maternal ethnicity and smoking status at baseline, child age at follow-up, gender and BMI z-score at follow-up (Table 4).
Time domain analysis revealed a decrease in the standard deviation of the normal to normal beat variation (−7.13 ms; −13.3 to −0.95) indicating reduced HRV; a reduction in the root mean square of successive differences (−8.9; −17.3 to −0.65) and the proportion of successive NN intervals that differ by more than 50 ms (pNN50, −7.41; −14.4 to −0.35) suggest reduced parasympathetic activity in the UPBEAT standard care arm compared to the normal BMI group, after adjustment (Table 4).
Children in the UPBEAT standard care arm showed an increase in sympathetic nervous system (SNS) activity and a reduction in parasympathetic nervous system (PNS) activity relative to the normal BMI group, after adjustment (SNS index: 0.98; 0.19 to 1.76: PNS index: −0.48; −0.90 to −0.06, Table 4).
Whilst there was a trend towards improvement of all parameters of HR and HRV in the UPBEAT intervention arm compared to the UPBEAT standard care arm, these differences were not significant.
Maternal obesity in pregnancy and child blood pressure
There was no association between maternal BMI and child systolic or diastolic blood pressure percentiles across the groups (Table 5).
Discussion
Children born to women with obesity in pregnancy versus women of normal BMI
Despite population-based evidence for increased cardiovascular risk in adults born to women with obesity [7], few investigators have addressed associations between maternal obesity and childhood cardiovascular outcomes [8, 28,29,30], with only one reporting on children of pre-school age [31]. One has addressed the effect of a prenatal intervention with metformin [32]. Our observation that children born to women with obesity have an elevated heart rate and evidence for concentric remodelling of the heart, independent of childhood BMI, contrasts with observations in older children from Generation R, a large cohort in the Netherlands in which associations between maternal BMI and LVM, LVMi and concentric LVH in the 6-year-old children became non-significant after adjustment for child’s BMI [33]. However, most women in that study were of normal BMI, only 8% being obese.
Although we found that some differences observed in left ventricular function (LVM/EDV ratio and SV, EF, LAV and LAVi) between children of women with obesity versus mothers of normal BMI did not persist after adjustment (including childhood BMI) others were maintained, including a slower E wave and E/A ratio, suggesting that the LV concentric remodelling observed was associated with slower LV filling, and an early indication of diastolic dysfunction, respectively.
Our study also contrasts with Litwin et al. [30] who, in a cohort of older children of women with obesity from the Finnish Gestational Diabetes Prevention Study (RADIEL), found that LVM, LVMi and LVM z-score were not associated with pre-pregnancy BMI, or gestational diabetes, or child body fat percentage, but rather with lean body mass at 6 years of age [30]. Whilst we found no apparent difference in BP or CIMT in our 3-year olds born to women with obesity, the RADIEL study also reported increased BP and increased carotid intima thickness in 6-year-old children of mothers with obesity compared to a reference population [8]. Arterial dimensions were again mainly predicted by child lean body mass, and not associated with maternal or child adiposity, or GDM [8].
Effects of a lifestyle intervention in women with obesity in pregnancy on offspring cardiovascular development
The association between maternal obesity and childhood cardiac structure and function might be attributed to common genetic cardiovascular traits in mother and child or to a shared family environment. However, this and other recent studies might argue in favour of a direct influence of maternal obesity during critical windows of development in utero. We report here that the UPBEAT lifestyle intervention in pregnancy complicated by obesity, attenuated the observed cardiac remodelling in the children with some improvement in cardiac function. As this was a representative subgroup from a well-conducted randomised control trial this should mitigate against confounding variables. We cannot discount a persisting influence of the UPBEAT pregnancy lifestyle intervention on mothers, influencing the family environment and childhood cardiovascular risk. However, data from the UPBEAT 6-month and 3-year follow-ups suggest no difference in maternal infant feeding practices [34] or the child’s dietary patterns and/or eating behaviours at 3 years of age [35]. Moreover, in the current UPBEAT subgroup, there were no differences in the mode of infant feeding on hospital discharge, mode of feeding at 6 months of age, or dietary patterns at 3 years of age between randomisation groups.
A direct in utero effect of maternal obesity becomes more likely in the context of parallel observations in foetuses and neonates of women with obesity. Abnormalities in the cardiac structure of offspring of women with obesity have been reported as early as 14 weeks’ gestation and are consistent with animal studies of maternal obesity-induced fibrosis in fetus myocardium [36]. Previous studies have reported increased foetal LV and RV global strain at 14 weeks’ gestation in women with obesity, compared to lean mothers, as assessed by ultrasound, a defect that persisted at 20 and 32 weeks’ gestation [36] and persists after birth. Moreover, compared to offspring of women of normal BMI, newborns of women with obesity have been shown to have larger and thicker left ventricles and an increased SV and EDV at 12 months of age [37]. Our study would suggest that these subclinical changes persist into pre-school years and there is evidence they may well track through childhood [38, 39]. Subclinical structural changes in children may be predictive of future cardiovascular events in adulthood [40].
Potential role of autonomic dysfunction in infant cardiac remodelling
In association with increased LVM and cardiac wall thickening, we also report altered autonomic activity in the 3-year olds as evidenced by increased heart rate in the children of women with obesity vs women of normal BMI and an increase in SNS index and a decrease in PNS index of the HRV. Although in this subgroup there was no significant effect of the UPBEAT intervention on the children’s heart rate, we have observed in the follow-up study of 514 UPBEAT 3-year olds a lower resting pulse rate (−5 bpm; −8.41, −1.07) in children from the UPBEAT intervention arm versus those from the UPBEAT standard care arm [35]. Several studies also report a higher heart rate and/or reduced HRV in foetuses of women with obesity [16, 41, 42].
These observations of altered autonomic nervous system function are paralleled in experimental animals. In young offspring of obese rodents, we have found, in association with hypertension, a shift in the sympathetic to parasympathetic ratio of HRV, indicative of heightened sympathetic efferent tone [43, 44] which persists to adulthood, and is mimicked by exposure to hyperleptinemia in new-born pups [43]. Hence, we have proposed that hyperleptinemia exposure, at the level of the developing hypothalamus, may play a permissive role in increased sympathetic efferent activity, leading to offspring cardiac dysfunction and hypertrophy.
Although animal models have provided some insight into potential mechanisms, any extrapolation to the human condition of maternal obesity must remain conjectural. An absence of differences in cord blood leptin in the original UPBEAT cohort (n = 698) between intervention and standard care arms would not support the leptin hypothesis [34]. Although the UPBEAT subgroup did not demonstrate significant differences in HR or parameters of HRV between randomisation arms, despite trends towards improvements, as mentioned above the larger cohort did demonstrate a significant reduction in resting pulse rate in the intervention group [35]. This could infer a role for sympathetic hyperactivity in the cardiac dysfunction observed, and its prevention in the intervention arm.
Several reports propose that maternal insulin resistance and/or diabetes may play a role in both altered foetal and neonatal HRV and offspring LVH [45,46,47] but we found no difference in the incidence of gestational diabetes or in fasting glucose at the time of the oral glucose tolerance test (24–28 weeks’ gestation) in pregnant women with obesity between standard care and intervention arms, suggesting independence from glucose status in the differences observed.
Other potential mediators of the effect of maternal obesity on cardiac remodelling, that may be mitigated by the UPBEAT intervention include lipotoxity in the placenta [48], leading to placental inflammation, oxidative stress, cardiac inflammation [49] and foetal myocyte hypertrophy [50].
Conclusions
Maternal obesity was associated with LV concentric remodelling in 3-year-old offspring that was prevented by the UPBEAT diet and exercise intervention in pregnancy suggesting the in utero developmental origins of cardiac remodelling and implicating an underlying elevated heart rate and increased sympathetic drive in the aetiology. Despite the relatively small cohort size in this exploratory study, the principal strength lies in the detailed phenotyping of the mother and children in the UPBEAT trial, and the breadth of cardiovascular measures undertaken in the child.
Subclinical cardiac hypertrophy in pre-school-age children secondary to maternal obesity may be a surrogate endpoint for the prediction of future cardiovascular risk [51] but could also be an antecedent to hypertension and cardiovascular disease [52, 53]. Evaluation of the same parameters in longitudinal prospective studies of cardiovascular risk in young people would be of interest. A recent systematic review identified consistent evidence of associations between maternal obesity and offspring cardiovascular dysfunction throughout the lifecourse, supporting targeted maternal obesity interventions for the promotion of offspring cardiovascular health [54].
A possible limitation is the sample size of those eligible from the original RCT, which may have resulted in selection bias, apparent in some minor differences by randomised group in maternal characteristics between those lost to follow-up and those included in the analyses (Supplementary Table 1). The unique findings of the effect of a pregnancy intervention (RCT) on the long-term cardiovascular health of the progeny warrant follow-up of the children in all the UPBEAT trial centres as they grow to maturity and funding has now been secured from the British Heart Foundation for a full cardiovascular assessment of the UPBEAT children at 10 years of age.
Data availability
All data generated or analysed during this study are included in this published article [and its Supplementary information files]. Data collected for this study, including individual participant data and a data dictionary defining each field in the set, will be made available to others, upon request following publication. Proposals to use data from the UPBEAT RCT are considered by the UPBEAT Scientific Committee. In the first instance, scientists interested in using these data should contact the UPBEAT principal investigator LP at lucilla.poston@kcl.ac.uk.
References
HQIP. National Maternity and Perinatal Audit (NMPA) clinical report. 2019.
Poston L, Caleyachetty R, Cnattingius S, Corvalan C, Uauy R, Herring S, et al. Preconceptional and maternal obesity: epidemiology and health consequences. Lancet Diabetes Endocrinol. 2016;4:1025–36.
Heslehurst N. Identifying ‘at risk’ women and the impact of maternal obesity on National Health Service maternity services. Proc Nutr Soc. 2011;70:439–49.
Godfrey KM, Reynolds RM, Prescott SL, Nyirenda M, Jaddoe VW, Eriksson JG, et al. Influence of maternal obesity on the long-term health of offspring. Lancet Diabetes Endocrinol. 2017;5:53–64.
Patro Golab B, Santos S, Voerman E, Lawlor DA, Jaddoe VWV, Gaillard R, et al. Influence of maternal obesity on the association between common pregnancy complications and risk of childhood obesity: an individual participant data meta-analysis. Lancet Child Adolesc Health. 2018;2:812–21.
Razaz N, Villamor E, Muraca GM, Bonamy AE, Cnattingius S. Maternal obesity and risk of cardiovascular diseases in offspring: a population-based cohort and sibling-controlled study. Lancet Diabetes Endocrinol. 2020;8:572–81.
Reynolds RM, Allan KM, Raja EA, Bhattacharya S, McNeill G, Hannaford PC, et al. Maternal obesity during pregnancy and premature mortality from cardiovascular event in adult offspring: follow-up of 1 323 275 person years. BMJ. 2013;347:f4539.
Sundholm JKM, Litwin L, Rono K, Koivusalo SB, Eriksson JG, Sarkola T. Maternal obesity and gestational diabetes: Impact on arterial wall layer thickness and stiffness in early childhood – RADIEL study six-year follow-up. Atherosclerosis. 2019;284:237–44.
Samuelsson AM, Matthews PA, Argenton M, Christie MR, McConnell JM, Jansen EH, et al. Diet-induced obesity in female mice leads to offspring hyperphagia, adiposity, hypertension, and insulin resistance: a novel murine model of developmental programming. Hypertension. 2008;51:383–92.
Samuelsson AM, Morris A, Igosheva N, Kirk SL, Pombo JMC, Coen CW, et al. Evidence for sympathetic origins of hypertension in juvenile offspring of obese rats. Hypertension. 2010;55:76–U129.
Fernandez-Twinn DS, Blackmore HL, Siggens L, Giussani DA, Cross CM, Foo R, et al. The programming of cardiac hypertrophy in the offspring by maternal obesity is associated with hyperinsulinemia, AKT, ERK, and mTOR activation. Endocrinology. 2012;153:5961–71.
Samuelsson AS, Mullier A, Maicas N, Oosterhuis NR, Eun Bae S, Novoselova TV, et al. Central role for melanocortin-4 receptors in offspring hypertension arising from maternal obesity. Proc Natl Acad Sci USA. 2016;113:12298–303.
Poston L, Bell R, Croker H, Flynn AC, Godfrey KM, Goff L, et al. Effect of a behavioural intervention in obese pregnant women (the UPBEAT study): a multicentre, randomised controlled trial. Lancet Diabetes Endocrinol. 2015;3:767–77.
Mills HL, Patel N, White SL, Pasupathy D, Briley AL, Santos Ferreira DL, et al. The effect of a lifestyle intervention in obese pregnant women on gestational metabolic profiles: findings from the UK Pregnancies Better Eating and Activity Trial (UPBEAT) randomised controlled trial. BMC Med. 2019;17:15.
Patel N, Godfrey KM, Pasupathy D, Levin J, Flynn AC, Hayes L, et al. Infant adiposity following a randomised controlled trial of a behavioural intervention in obese pregnancy. Int J Obes (Lond). 2017;41:1018–26.
Mat Husin H, Schleger F, Bauer I, Fehlert E, Kiefer-Schmidt I, Weiss M, et al. Maternal weight, weight gain, and metabolism are associated with changes in fetal heart rate and variability. Obesity (Silver Spring). 2020;28:114–21.
Daniels SR, Kimball TR, Morrison JA, Khoury P, Meyer RA. Indexing left ventricular mass to account for differences in body size in children and adolescents without cardiovascular disease. Am J Cardiol. 1995;76:699–701.
Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2015;16:233–70.
de Simone G, Daniels SR, Devereux RB, Meyer RA, Roman MJ, de Divitiis O, et al. Left ventricular mass and body size in normotensive children and adults: assessment of allometric relations and impact of overweight. J Am Coll Cardiol. 1992;20:1251–60.
National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents. The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics. 2004;114:555–76.
Nunan D, Sandercock GR, Brodie DA. A quantitative systematic review of normal values for short-term heart rate variability in healthy adults. Pacing Clin Electrophysiol. 2010;33:1407–17.
Tarvainen MP, Niskanen JP, Lipponen JA, Ranta-Aho PO, Karjalainen PA. Kubios HRV-heart rate variability analysis software. Comput Methods Programs Biomed. 2014;113:210–20.
Tarvainen MP, Ranta-Aho PO, Karjalainen PA. An advanced detrending method with application to HRV analysis. IEEE Trans Biomed Eng. 2002;49:172–5.
Rothman KJ. No adjustments are needed for multiple comparisons. Epidemiology. 1990;1:43–6.
Althouse AD. Adjust for multiple comparisons? It’s not that simple. Ann Thorac Surg. 2016;101:1644–5.
Hershman DL, Unger JM, Greenlee H, Capodice JL, Lew DL, Darke AK, et al. Effect of acupuncture vs sham acupuncture or waitlist control on joint pain related to aromatase inhibitors among women with early-stage breast cancer: a randomized clinical trial. JAMA. 2018;320:167–76.
Qian X, Loo BRY, Castellanos FX, Liu S, Koh HL, Poh XWW, et al. Brain-computer-interface-based intervention re-normalizes brain functional network topology in children with attention deficit/hyperactivity disorder. Transl Psychiatry. 2018;8:149.
Gademan MG, van Eijsden M, Roseboom TJ, van der Post JA, Stronks K, Vrijkotte TG. Maternal prepregnancy body mass index and their children’s blood pressure and resting cardiac autonomic balance at age 5 to 6 years. Hypertension. 2013;62:641–7.
Gaillard R, Welten M, Oddy WH, Beilin LJ, Mori TA, Jaddoe VW, et al. Associations of maternal prepregnancy body mass index and gestational weight gain with cardio-metabolic risk factors in adolescent offspring: a prospective cohort study. BJOG. 2016;123:207–16.
Litwin L, Sundholm JKM, Rono K, Koivusalo SB, Eriksson JG, Sarkola T. Transgenerational effects of maternal obesity and gestational diabetes on offspring body composition and left ventricle mass: the Finnish Gestational Diabetes Prevention Study (RADIEL) 6-year follow-up. Diabet Med. 2020;37:147–56.
Geelhoed JJ, VAN Osch-Gevers L, Verburg BO, Steegers EA, Hofman A, Helbing W, et al. Maternal anthropometrics in pregnancy are associated with left ventricular mass in infancy. The generation R study. Pediatr Res. 2008;63:62–6.
Panagiotopoulou O, Syngelaki A, Georgiopoulos G, Simpson J, Akolekar R, Shehata H, et al. Metformin use in obese mothers is associated with improved cardiovascular profile in the offspring. Am J Obstet Gynecol. 2020;223:246 e1–e10.
Toemen L, Gishti O, van Osch-Gevers L, Steegers EA, Helbing WA, Felix JF, et al. Maternal obesity, gestational weight gain and childhood cardiac outcomes: role of childhood body mass index. Int J Obes (Lond). 2016;40:1070–8.
Patel N, Dalrymple KV, Briley AL, Pasupathy D, Seed PT, Flynn AC, et al. Mode of infant feeding, eating behaviour and anthropometry in infants at 6-months of age born to obese women – a secondary analysis of the UPBEAT trial. BMC Pregnancy Childbirth. 2018;18:355.
Dalrymple KV, Tydeman FAS, Taylor PD, Flynn AC, O'Keeffe M, Briley AL, et al. Adiposity and cardiovascular outcomes in three-year-old children of participants in UPBEAT, an RCT of a complex intervention in pregnant women with obesity. Pediatr Obes. 2021;16:e12725.
Ingul CB, Loras L, Tegnander E, Eik-Nes SH, Brantberg A. Maternal obesity affects fetal myocardial function as early as in the first trimester. Ultrasound Obstet Gynecol. 2016;47:433–42.
Guzzardi MA, Liistro T, Gargani L, Ait Ali L, D’Angelo G, Rocchiccioli S, et al. Maternal obesity and cardiac development in the offspring: study in human neonates and minipigs. JACC Cardiovasc Imaging. 2018;11:1750–5.
Schieken RM, Schwartz PF, Goble MM. Tracking of left ventricular mass in children: race and sex comparisons: the MCV Twin Study. Medical College of Virginia. Circulation. 1998;97:1901–6.
Toemen L, Gaillard R, van Osch-Gevers L, Helbing WA, Hofman A, Jaddoe VW. Tracking of structural and functional cardiac measures from infancy into school-age. Eur J Prev Cardiol. 2017;24:1408–15.
Armstrong AC, Liu K, Lewis CE, Sidney S, Colangelo LA, Kishi S, et al. Left atrial dimension and traditional cardiovascular risk factors predict 20-year clinical cardiovascular events in young healthy adults: the CARDIA study. Eur Heart J Cardiovasc Imaging. 2014;15:893–9.
Voegtline KM, Costigan KA, Henderson JL, DiPietro JA. Fetal heart rate and motor development in overweight and obese pregnant women. Int J Gynaecol Obstet. 2016;133:103–7.
Ojala T, Aaltonen J, Siira S, Jalonen J, Ekholm E, Ekblad U, et al. Fetal cardiac sympathetic activation is linked with maternal body mass index. Early Hum Dev. 2009;85:557–60.
Samuelsson AM, Clark J, Rudyk O, Shattock MJ, Bae SE, South T, et al. Experimental hyperleptinemia in neonatal rats leads to selective leptin responsiveness, hypertension, and altered myocardial function. Hypertension. 2013;62:627–33.
Samuelsson AM, Morris A, Igosheva N, Kirk SL, Pombo JM, Coen CW, et al. Evidence for sympathetic origins of hypertension in juvenile offspring of obese rats. Hypertension. 2010;55:76–82.
Krishnaveni GV, Veena SR, Hill JC, Kehoe S, Karat SC, Fall CH. Intrauterine exposure to maternal diabetes is associated with higher adiposity and insulin resistance and clustering of cardiovascular risk markers in Indian children. Diabetes Care. 2010;33:402–4.
Yli BM, Kallen K, Stray-Pedersen B, Amer-Wahlin I. Intrapartum fetal ECG and diabetes. J Matern Fetal Neonatal Med. 2008;21:231–8.
Marco LJ, McCloskey K, Vuillermin PJ, Burgner D, Said J, Ponsonby AL. Cardiovascular disease risk in the offspring of diabetic women: the impact of the intrauterine environment. Exp Diabetes Res. 2012;2012:565160.
Saben J, Lindsey F, Zhong Y, Thakali K, Badger TM, Andres A, et al. Maternal obesity is associated with a lipotoxic placental environment. Placenta. 2014;35:171–7.
Kandadi MR, Hua Y, Zhu M, Turdi S, Nathanielsz PW, Ford SP, et al. Influence of gestational overfeeding on myocardial proinflammatory mediators in fetal sheep heart. J Nutr Biochem. 2013;24:1982–90.
Thornburg KL, O’Tierney PF, Louey S. Review: The placenta is a programming agent for cardiovascular disease. Placenta. 2010;31(Suppl):S54–9.
Du T, Fernandez C, Barshop R, Chen W, Urbina EM, Bazzano LA. Pediatric Hypertension guidelines improve prediction of adult cardiovascular outcomes. Hypertension. 2019;73:1217–23.
Grossman C, Grossman A, Koren-Morag N, Azaria B, Goldstein L, Grossman E. Interventricular septum thickness predicts future systolic hypertension in young healthy pilots. Hypertens Res. 2008;31:15–20.
Park SK, Jung JY, Kang JG, Chung PW, Oh CM. Left ventricular geometry and risk of incident hypertension. Heart. 2019;105:1402–7.
Kankowski L, Ardissino M, McCracken C, Lewandowski AJ, Leeson P, Neubauer S, et al. The impact of maternal obesity on offspring cardiovascular health: a systematic literature review. Front Endocrinol (Lausanne). 2022;13:868441.
Acknowledgements
We thank all staff in the UPBEAT consortium, the research midwives and research assistants, and we are most grateful to all the women and their children who took part in the UPBEAT study.
Funding
British Heart Foundation (PG/13/38/30289); European Union’s 7th Framework Programme (FP7/2007–2013), project EarlyNutrition; grant agreement no. 289346 and the National Institute for Health Research (NIHR, UK) Programme Grants for Applied Research Programme (RP-0407-10452). LP is supported by Tommy’s charity and KVD by the BHF (FS/17/71/32953). This study also received support from the NIHR Biomedical Research Centre based at Guy’s and St Thomas’ NHS Foundation Trust and King’s College London. The views expressed in this publication are those of the author and not necessarily those of the NHS, the NIHR, or the Department of Health.
Author information
Authors and Affiliations
Consortia
Contributions
PDT and LP were the principal investigators and DP, AMG and PJC were the co-principal investigators. PDT, MCV, CS, AB, PTS, DP, AMG, and LP conceived and/or designed the work that led to the submission; PDT, HG, HG, HS, FF, KVD, PS, LY, FM, BJ, MCV, CS, AB, KVD, DP and PJS acquired data and/or played an important role in interpreting the results; PT and HG drafted the manuscript and all authors contributed to the revision of the manuscript and approved the final version. KVD and PTS were responsible for statistical analysis. LP, MCV, AB, CS and DP contributed to the study supervision. FM, BJ, MCV, AB and CS led and participated in the site work, including recruitment, follow-up and data collection. CS and AB monitored the trial and database. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Taylor, P.D., Gu, H., Saunders, H. et al. Lifestyle intervention in obese pregnancy and cardiac remodelling in 3-year olds: children of the UPBEAT RCT. Int J Obes 46, 2145–2155 (2022). https://doi.org/10.1038/s41366-022-01210-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-022-01210-3
This article is cited by
-
Maternal obesity and offspring cardiovascular remodelling — the effect of preconception and antenatal lifestyle interventions: a systematic review
International Journal of Obesity (2024)
-
Adipositas und Diabetes in der Schwangerschaft
Die Diabetologie (2023)